Abstract
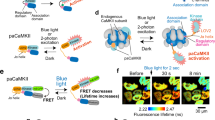
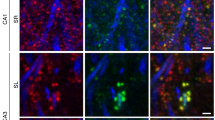
Calcium/calmodulin-dependent kinase II (CaMKII) plays a central part in long-term potentiation (LTP), which underlies some forms of learning and memory. Here we monitored the spatiotemporal dynamics of CaMKII activation in individual dendritic spines during LTP using two-photon fluorescence lifetime imaging microscopy, in combination with two-photon glutamate uncaging. Induction of LTP and associated spine enlargement in single spines triggered transient (∼1 min) CaMKII activation restricted to the stimulated spines. CaMKII in spines was specifically activated by NMDA receptors and L-type voltage-sensitive calcium channels, presumably by nanodomain Ca2+ near the channels, in response to glutamate uncaging and depolarization, respectively. The high degree of compartmentalization and channel specificity of CaMKII signalling allow stimuli-specific spatiotemporal patterns of CaMKII signalling and may be important for synapse-specificity of synaptic plasticity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rosenberg, O. S. et al. Oligomerization states of the association domain and the holoenyzme of Ca2+/CaM kinase II. FEBS J. 273, 682–694 (2006)
Lisman, J. E. & Zhabotinsky, A. M. A model of synaptic memory: a CaMKII/PP1 switch that potentiates transmission by organizing an AMPA receptor anchoring assembly. Neuron 31, 191–201 (2001)
Merrill, M. A., Chen, Y., Strack, S. & Hell, J. W. Activity-driven postsynaptic translocation of CaMKII. Trends Pharmacol. Sci. 26, 645–653 (2005)
Hudmon, A. et al. CaMKII tethers to L-type Ca2+ channels, establishing a local and dedicated integrator of Ca2+ signals for facilitation. J. Cell Biol. 171, 537–547 (2005)
Takao, K. et al. Visualization of synaptic Ca2+/calmodulin-dependent protein kinase II activity in living neurons. J. Neurosci. 25, 3107–3112 (2005)
Zacharias, D. A., Violin, J. D., Newton, A. C. & Tsien, R. Y. Partitioning of lipid-modified monomeric GFPs into membrane microdomains of live cells. Science 296, 913–916 (2002)
Ganesan, S., Ameer-Beg, S. M., Ng, T. T., Vojnovic, B. & Wouters, F. S. A dark yellow fluorescent protein (YFP)-based Resonance Energy-Accepting Chromoprotein (REACh) for Förster resonance energy transfer with GFP. Proc. Natl Acad. Sci. USA 103, 4089–4094 (2006)
Yasuda, R. Imaging spatiotemporal dynamics of neuronal signaling using fluorescence resonance energy transfer and fluorescence lifetime imaging microscopy. Curr. Opin. Neurobiol. 16, 551–561 (2006)
Yasuda, R. et al. Super-sensitive Ras activation in dendrites and spines revealed by 2-photon fluorescence lifetime imaging. Nature Neurosci. 9, 283–291 (2006)
Harvey, C. D., Yasuda, R., Zhong, H. & Svoboda, K. The spread of Ras activity triggered by activation of a single dendritic spine. Science 321, 136–140 (2008)
McAllister, A. K. Biolistic transfection of neurons. Sci. STKE 2000, pl1 (2000)
Matsuzaki, M. et al. Dendritic spine geometry is critical for AMPA receptor expression in hippocampal CA1 pyramidal neurons. Nature Neurosci. 4, 1086–1092 (2001)
Matsuzaki, M., Honkura, N., Ellis-Davies, G. C. & Kasai, H. Structural basis of long-term potentiation in single dendritic spines. Nature 429, 761–766 (2004)
Harvey, C. D. & Svoboda, K. Locally dynamic synaptic learning rules in pyramidal neuron dendrites. Nature 450, 1195–1200 (2007)
Otmakhov, N. et al. Persistent accumulation of calcium/calmodulin-dependent protein kinase II in dendritic spines after induction of NMDA receptor-dependent chemical long-term potentiation. J. Neurosci. 24, 9324–9331 (2004)
Ohno, M., Frankland, P. W. & Silva, A. J. A pharmacogenetic inducible approach to the study of NMDA/αCaMKII signaling in synaptic plasticity. Curr. Biol. 12, 654–656 (2002)
Sabatini, B. L., Oertner, T. G. & Svoboda, K. The life cycle of Ca2+ ions in dendritic spines. Neuron 33, 439–452 (2002)
Malenka, R. C. & Bear, M. F. LTP and LTD: an embarrassment of riches. Neuron 44, 5–21 (2004)
Conti, R. & Lisman, J. A large sustained Ca2+ elevation occurs in unstimulated spines during the LTP pairing protocol but does not change synaptic strength. Hippocampus 12, 667–679 (2002)
Magee, J. C. & Johnston, D. Characterization of single voltage-gated Na+ and Ca2+ channels in apical dendrites of rat CA1 pyramidal neurons. J. Physiol. (Lond.) 487, 67–90 (1995)
Noguchi, J., Matsuzaki, M., Ellis-Davies, G. C. & Kasai, H. Spine-neck geometry determines NMDA receptor-dependent Ca2+ signaling in dendrites. Neuron 46, 609–622 (2005)
Sobczyk, A., Scheuss, V. & Svoboda, K. NMDA receptor subunit-dependent [Ca2+] signaling in individual hippocampal dendritic spines. J. Neurosci. 25, 6037–6046 (2005)
Dan, Y. & Poo, M. M. Spike timing-dependent plasticity of neural circuits. Neuron 44, 23–30 (2004)
Grover, L. M. & Teyler, T. J. Two components of long-term potentiation induced by different patterns of afferent activation. Nature 347, 477–479 (1990)
Magee, J. C. & Johnston, D. A synaptically controlled, associative signal for Hebbian plasticity in hippocampal neurons. Science 275, 209–213 (1997)
Remy, S. & Spruston, N. Dendritic spikes induce single-burst long-term potentiation. Proc. Natl Acad. Sci. USA 104, 17192–17197 (2007)
Yasuda, R., Sabatini, B. L. & Svoboda, K. Plasticity of calcium channels in dendritic spines. Nature Neurosci. 6, 948–955 (2003)
Wheeler, D. G., Barrett, C. F., Groth, R. D., Safa, P. & Tsien, R. W. CaMKII locally encodes L-type channel activity to signal to nuclear CREB in excitation-transcription coupling. J. Cell Biol. 183, 849–863 (2008)
Neher, E. Usefulness and limitations of linear approximations to the understanding of Ca++ signals. Cell Calcium 24, 345–357 (1998)
Zhu, J. J., Qin, Y., Zhao, M., Van Aelst, L. & Malinow, R. Ras and Rap control AMPA receptor trafficking during synaptic plasticity. Cell 110, 443–455 (2002)
Okamoto, K., Narayanan, R., Lee, S. H., Murata, K. & Hayashi, Y. The role of CaMKII as an F-actin-bundling protein crucial for maintenance of dendritic spine structure. Proc. Natl Acad. Sci. USA 104, 6418–6423 (2007)
Malinow, R., Schulman, H. & Tsien, R. W. Inhibition of postsynaptic PKC or CaMKII blocks induction but not expression of LTP. Science 245, 862–866 (1989)
Chen, H. X., Otmakhov, N., Strack, S., Colbran, R. J. & Lisman, J. E. Is persistent activity of calcium/calmodulin-dependent kinase required for the maintenance of LTP? J. Neurophysiol. 85, 1368–1376 (2001)
Fukunaga, K., Muller, D. & Miyamoto, E. Increased phosphorylation of Ca2+/calmodulin-dependent protein kinase II and its endogenous substrates in the induction of long-term potentiation. J. Biol. Chem. 270, 6119–6124 (1995)
Barria, A., Muller, D., Derkach, V., Griffith, L. C. & Soderling, T. R. Regulatory phosphorylation of AMPA-type glutamate receptors by CaM-KII during long-term potentiation. Science 276, 2042–2045 (1997)
Lisman, J., Schulman, H. & Cline, H. The molecular basis of CaMKII function in synaptic and behavioural memory. Nature Rev. Neurosci. 3, 175–190 (2002)
Cheng, D. et al. Relative and absolute quantification of postsynaptic density proteome isolated from rat forebrain and cerebellum. Mol. Cell. Proteomics 5, 1158–1170 (2006)
Kullmann, D. M., Perkel, D. J., Manabe, T. & Nicoll, R. A. Ca2+ entry via postsynaptic voltage-sensitive Ca2+ channels can transiently potentiate excitatory synaptic transmission in the hippocampus. Neuron 9, 1175–1183 (1992)
Murakoshi, H., Lee, S.-J. & Yasuda, R. Highly sensitive and quantitative FRET-FLIM imaging in single dendritic spines using improved non-radiative YFP. Brain Cell Biol. 36, 31–42 (2008)
Stoppini, L., Buchs, P. A. & Muller, D. A simple method for organotypic cultures of nervous tissue. J. Neurosci. Methods 37, 173–182 (1991)
Nagai, T. et al. A variant of yellow fluorescent protein with fast and efficient maturation for cell-biological applications. Nature Biotechnol. 20, 87–90 (2002)
Kwok, S. et al. Genetically encoded probe for fluorescence lifetime imaging of CaMKII activity. Biochem. Biophys. Res. Commun. 369, 519–525 (2008)
Yasuda, R. et al. Imaging calcium concentration dynamics in small neuronal compartments. Sci. STKE 2004, pl5 (2004)
Tsien, R. Y. The green fluorescent protein. Annu. Rev. Biochem. 67, 509–544 (1998)
Schwille, P., Haupts, U., Maiti, S. & Webb, W. W. Molecular dynamics in living cells observed by fluorescence correlation spectroscopy with one- and two-photon excitation. Biophys. J. 77, 2251–2265 (1999)
Acknowledgements
We thank H. Murakoshi for purified mEGFP and technical advice, Y. Hayashi for complementary DNAs and shRNA, A. Wang for cultured slices, T. Zimmerman and D. Kloetzer for laboratory management, and M. Ehlers, C. Harvey, J. Lisman, M. Patterson, S. Raghavachari, K. Svoboda, R. Weinberg and H. Zhong for comments on the manuscript. This study was funded by Burroughs Wellcome Fund, Dana Foundation, Alfred P. Sloan Foundation, Autism Speaks, National Alliance of Autism Research, Whitehall Foundation, Alzheimer’s Association, National Institute of Mental Health (R01MH08004), National Science Foundation (0642000), Duke University Undergraduate Research Support Grants (S.-J.R.L.), Howard Hughes Neuroscience Forum Fellowship (S.-J.R.L.), and Ruth K. Broad Biomedical Research Foundation (E.M.S.).
Author Contributions S.-J.R.L. and R.Y. designed the experiments. R.Y. built the microscope and developed the green-Camuiα construct. S.-J.R.L. performed most of the experiments. S.-J.R.L. and Y.E.-R. performed calcium imaging. E.M.S. performed immunostaining and pull-down assay. S.-J.R.L. and R.Y. analysed the data. R.Y. wrote the paper. All authors discussed and commented on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Notes, Supplementary References and Supplementary Figures 1-11 with Legends. (PDF 639 kb)
Supplementary Movie 1
This movie shows CaMKII activation during structural plasticity induced by 2-photon glutamate uncaging in the absence of extracellular Mg2+. The white arrowhead indicates the location of 2-photon glutamate uncaging. (AVI 6124 kb)
Supplementary Movie 2
This movie shows CaMKII activation during LTP induced by 2-photon glutamate uncaging paired with postsynaptic depolarization. The white box at the left top corner indicates the time the neuron is depolarized, and the white arrowhead indicates the location of 2-photon uncaging. (AVI 5225 kb)
Rights and permissions
About this article
Cite this article
Lee, SJ., Escobedo-Lozoya, Y., Szatmari, E. et al. Activation of CaMKII in single dendritic spines during long-term potentiation. Nature 458, 299–304 (2009). https://doi.org/10.1038/nature07842
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature07842
This article is cited by
-
Myeloid deficiency of the intrinsic clock protein BMAL1 accelerates cognitive aging by disrupting microglial synaptic pruning
Journal of Neuroinflammation (2023)
-
Transport and localization on dendrite-inspired flat band linear photonic lattices
Scientific Reports (2023)
-
Excitation–transcription coupling, neuronal gene expression and synaptic plasticity
Nature Reviews Neuroscience (2023)
-
Increased spine PIP3 is sequestered from dendritic shafts
Molecular Brain (2022)
-
CaMKII: a central molecular organizer of synaptic plasticity, learning and memory
Nature Reviews Neuroscience (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.