Abstract
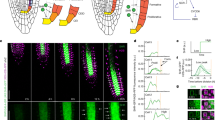
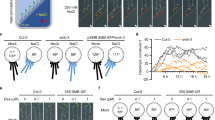
Root hairs are cellular protuberances extending from the root surface into the soil; there they provide access to immobile inorganic ions such as phosphate, which are essential for growth1. Their cylindrical shape results from a polarized mechanism of cell expansion called tip growth in which elongation is restricted to a small area at the surface of the hair-forming cell (trichoblast) tip2,3,4. Here we identify proteins that spatially control the sites at which cell growth occurs by isolating Arabidopsis mutants (scn1) that develop ectopic sites of growth on trichoblasts. We cloned SCN1 and showed that SCN1 is a RhoGTPase GDP dissociation inhibitor (RhoGDI) that spatially restricts the sites of growth to a single point on the trichoblast. We showed previously that localized production of reactive oxygen species by RHD2/AtrbohC NADPH oxidase is required for hair growth5; here we show that SCN1/AtrhoGDI1 is a component of the mechanism that focuses RHD2/AtrbohC-catalysed production of reactive oxygen species to hair tips during wild-type development. We propose that the spatial organization of growth in plant cells requires the local RhoGDI-regulated activation of the RHD2/AtrbohC NADPH oxidase.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gahoonia, T. S., Care, D. & Nielsen, N. E. Root hairs and phosphorus acquisition of wheat and barley cultivars. Plant Soil 191, 181–188 (1997)
Dolan, L. et al. Clonal relationships and cell patterning in the root epidermis of Arabidopsis. Development 120, 2465–2475 (1994)
Shaw, S. L., Dumais, J. & Long, S. R. Cell surface expansion in polarly growing root hairs of Medicago truncatula. Plant Physiol. 124, 959–970 (2000)
Dumais, J., Long, S. R. & Shaw, S. L. The mechanics of surface expansion anisotropy in Medicago truncatula root hairs. Plant Physiol. 136, 3266–3275 (2004)
Foreman, J. et al. Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature 422, 442–446 (2003)
Parker, J. S., Cavell, A. C., Dolan, L., Roberts, K. & Grierson, C. S. Genetic interactions during root hair morphogenesis in Arabidopsis. Plant Cell 12, 1961–1974 (2000)
Fryer, M. J., Oxborough, K., Mullineaux, P. M. & Baker, N. R. Imaging of photo-oxidative stress responses in leaves. J. Exp. Bot. 53, 1249–1254 (2002)
Barcelo, A. R. Some properties of the H2O2/O2-generating system from the lignifying xylem of Zinnia elegans. Free Radical Res. 31 (Suppl.), S147–S154 (1999)
Bischoff, F., Vahlkamp, L., Molendijk, A. & Palme, K. Localization of AtROP4 and AtROP6 and interaction with the guanine nucleotide dissociation inhibitor AtRhoGDI1 from Arabidopsis. Plant Mol. Biol. 42, 515–530 (2000)
Vernoud, V., Horton, A. C., Yang, Z. & Nielsen, E. Analysis of the small GTPase gene superfamily of Arabidopsis. Plant Physiol. 131, 1191–1208 (2003)
Jones, M. A. et al. The Arabidopsis Rop2 GTPase is a positive regulator of both root hair initiation and tip growth. Plant Cell 14, 763–776 (2002)
Molendijk, A. J. et al. Arabidopsis thaliana Rop GTPases are localized to tips of root hairs and control polar growth. EMBO J. 20, 2779–2788 (2001)
Grizot, S. et al. Crystal structure of the Rac1–RhoGDI complex involved in NADPH oxidase activation. Biochemistry 40, 10007–10013 (2001)
Rivero, F. et al. Defects in cytokinesis, actin reorganization and the contractile vacuole in cells deficient in RhoGDI. EMBO J. 21, 4539–4549 (2002)
Xu, J. & Scheres, B. Dissection of Arabidopsis ADP-RIBOSYLATION FACTOR 1 function in epidermal cell polarity. Plant Cell 17, 525–536 (2005)
Fu, Y., Wu, G. & Yang, Z. Rop GTPase-dependent dynamics of tip-localized F-actin controls tip growth in pollen tubes. J. Cell Biol. 152, 1019–1032 (2001)
Segal, A. W. & Abo, A. The biochemical basis of the NADPH oxidase of phagocytes. Trends Biochem. Sci. 18, 43–47 (1993)
Abo, A. et al. Activation of the NADPH oxidase involves the small GTP-binding protein p21rac1. Nature 353, 668–670 (1991)
Favery, B. et al. KOJAK encodes a cellulose synthase-like protein required for root hair cell morphogenesis in Arabidopsis. Genes Dev. 15, 79–89 (2001)
Cowling, R. J., Kamiya, Y., Seto, H. & Harberd, N. P. Gibberellin dose–response regulation of GA4 gene transcript levels in Arabidopsis. Plant Physiol. 117, 1195–1203 (1998)
Coen, E. S. et al. floricaula: a homeotic gene required for flower development in Antirrhinum majus. Cell 63, 1311–1322 (1990)
Berman, H. M. et al. The Protein Data Bank. Nucleic Acids Res. 28, 235–242 (2000)
Acknowledgements
We thank F. Cvrckova for the anti-Rop antibody, C. S. Grierson for providing scn1-1 seeds, and B. Scheres and J. Xu for providing the ROP2::YFP fusion construct and ROP2::YFP-transformed seed. S.T. is supported by a grant from the BBSRC to L.D., who is also supported by EU TIPNET. R.C., L.D. and M.D. are funded by a grant-in-aid to the John Innes Centre by the BBSRC. P.D. and S.D. were supported by grants to L.D., P. Shaw and J. Doonan and from Syngenta and the BBSRC. V.Z. and H.K. are funded by EU TIPNET and GACR (Grant Agency of Czech Republic).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Figure 1
NBT-stained growing and non-growing root hairs. (JPG 114 kb)
Supplementary Figure 2
Cloning, sequence analysis and expression of SCN1/AtRhoGDI1. (PDF 5957 kb)
Supplementary Figure 3
Interaction between scn1-2 and ROP proteins is defective in vitro. (PDF 1484 kb)
Supplementary Figure 4
SCN1 regulates the subcellular localisation of ROP2 protein. (PDF 18138 kb)
Supplementary Figure 5
Primers used in this study. (DOC 25 kb)
Supplementary Figure Legends
Text to accompany the above Supplementary Figures. (DOC 27 kb)
Supplementary Methods
Additional descriptions of the methods used in this study. (DOC 32 kb)
Rights and permissions
About this article
Cite this article
Carol, R., Takeda, S., Linstead, P. et al. A RhoGDP dissociation inhibitor spatially regulates growth in root hair cells. Nature 438, 1013–1016 (2005). https://doi.org/10.1038/nature04198
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04198
This article is cited by
-
Two subtypes of GTPase-activating proteins coordinate tip growth and cell size regulation in Physcomitrium patens
Nature Communications (2023)
-
Temporal expression of defense-related genes in Ganoderma-infected oil palm roots
Trees (2022)
-
Rice Carbohydrate-Binding Malectin-Like Protein, OsCBM1, Contributes to Drought-Stress Tolerance by Participating in NADPH Oxidase-Mediated ROS Production
Rice (2021)
-
ARMADILLO REPEAT ONLY proteins confine Rho GTPase signalling to polar growth sites
Nature Plants (2020)
-
A nucleoside diphosphate kinase gene OsNDPK4 is involved in root development and defense responses in rice (Oryza sativa L.)
Planta (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.