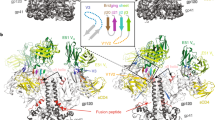
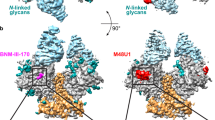
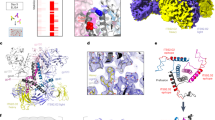
Abstract
Envelope glycoproteins of human and simian immunodeficiency virus (HIV and SIV) undergo a series of conformational changes when they interact with receptor (CD4) and co-receptor on the surface of a potential host cell, leading ultimately to fusion of viral and cellular membranes. Structures of fragments of gp120 and gp41 from the envelope protein are known, in conformations corresponding to their post-attachment and postfusion states, respectively. We report the crystal structure, at 4 Å resolution, of a fully glycosylated SIV gp120 core, in a conformation representing its prefusion state, before interaction with CD4. Parts of the protein have a markedly different organization than they do in the CD4-bound state. Comparison of the unliganded and CD4-bound structures leads to a model for events that accompany receptor engagement of an envelope glycoprotein trimer. The two conformations of gp120 also present distinct antigenic surfaces. We identify the binding site for a compound that inhibits viral entry.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Wyatt, R. & Sodroski, J. The HIV-1 envelope glycoproteins: Fusogens, antigens, and immunogens. Science 280, 1884–1888 (1998)
Allan, J. S. et al. Major glycoprotein antigens that induce antibodies in AIDS patients are encoded by HTLV-III. Science 228, 1091–1094 (1985)
Veronese, F. D. et al. Characterization of gp41 as the transmembrane protein coded by the HTLV-III/LAV envelope gene. Science 229, 1402–1405 (1985)
Center, R. J. et al. Oligomeric structure of the human immunodeficiency virus type 1 envelope protein on the virion surface. J. Virol. 76, 7863–7867 (2002)
Dalgleish, A. G. et al. The CD4 (T4) antigen is an essential component of the receptor for the AIDS retrovirus. Nature 312, 763–767 (1984)
Feng, Y., Broder, C. C., Kennedy, P. E. & Berger, E. A. HIV-1 entry cofactor: functional cDNA cloning of a seven-transmembrane, G protein-coupled receptor. Science 272, 872–877 (1996)
Trkola, A. et al. CD4-dependent, antibody-sensitive interactions between HIV-1 and its co-receptor CCR-5. Nature 384, 184–187 (1996)
Wu, L. et al. CD4-induced interaction of primary HIV-1 gp120 glycoproteins with the chemokine receptor CCR-5. Nature 384, 179–183 (1996)
Sattentau, Q. J. & Moore, J. P. Conformational changes induced in the human immunodeficiency virus envelope glycoprotein by soluble CD4 binding. J. Exp. Med. 174, 407–415 (1991)
Sattentau, Q. J., Moore, J. P., Vignaux, F., Traincard, F. & Poignard, P. Conformational changes induced in the envelope glycoproteins of the human and simian immunodeficiency viruses by soluble receptor binding. J. Virol. 67, 7383–7393 (1993)
Rizzuto, C. D. et al. A conserved HIV gp120 glycoprotein structure involved in chemokine receptor binding. Science 280, 1949–1953 (1998)
Chan, D. C., Fass, D., Berger, J. M. & Kim, P. S. Core structure of gp41 from the HIV envelope glycoprotein. Cell 89, 263–273 (1997)
Weissenhorn, W., Dessen, A., Harrison, S. C., Skehel, J. J. & Wiley, D. C. Atomic structure of the ectodomain from HIV-1 gp41. Nature 387, 426–430 (1997)
Kwong, P. D. et al. Structure of an HIV gp120 envelope glycoprotein in complex with the CD4 receptor and a neutralizing human antibody. Nature 393, 648–659 (1998)
Chan, D. C., Chutkowski, C. T. & Kim, P. S. Evidence that a prominent cavity in the coiled coil of HIV type 1 gp41 is an attractive drug target. Proc. Natl Acad. Sci. USA 95, 15613–15617 (1998)
Rimsky, L. T., Shugars, D. C. & Matthews, T. J. Determinants of human immunodeficiency virus type 1 resistance to gp41-derived inhibitory peptides. J. Virol. 72, 986–993 (1998)
Wyatt, R. et al. The antigenic structure of the HIV gp120 envelope glycoprotein. Nature 393, 705–711 (1998)
Pollard, S. R., Meier, W., Chow, P., Rosa, J. J. & Wiley, D. C. CD4-binding regions of human immunodeficiency virus envelope glycoprotein gp120 defined by proteolytic digestion. Proc. Natl Acad. Sci. USA 88, 11320–11324 (1991)
Wyatt, R. et al. Functional and immunologic characterization of human immunodeficiency virus type 1 envelope glycoproteins containing deletions of the major variable regions. J. Virol. 67, 4557–4565 (1993)
Rud, E. W. et al. in Vaccines 92: Modern Approaches to New Vaccines Including Prevention of AIDS (eds Brown, F., Chanock, R. M., Ginsberg, H. S. & Lerner, R. A.) 229–235 (Cold Spring Harbor Laboratory, New York, 1992)
Chen, B., Vogan, E., Gong, H. Y., Wiley, D. C. & Harrison, S. C. Determining the structure of an unliganded and fully-glycosylated SIV gp120 envelope glycoprotein. Structure (Camb.) (in the press)
Kwong, P. D. et al. Probability analysis of variational crystallization and its application to gp120, the exterior envelope glycoprotein of type 1 human immunodeficiency virus (HIV-1). J. Biol. Chem. 274, 4115–4123 (1999)
Thali, M. et al. Characterization of conserved human immunodeficiency virus type 1 gp120 neutralization epitopes exposed upon gp120–CD4 binding. J. Virol. 67, 3978–3988 (1993)
Lin, P. F. et al. A small molecule HIV-1 inhibitor that targets the HIV-1 envelope and inhibits CD4 receptor binding. Proc. Natl Acad. Sci. USA 100, 11013–11018 (2003)
Moore, J. P. & Sodroski, J. Antibody cross-competition analysis of the human immunodeficiency virus type 1 gp120 exterior envelope glycoprotein. J. Virol. 70, 1863–1872 (1996)
Myszka, D. G. et al. Energetics of the HIV gp120–CD4 binding reaction. Proc. Natl Acad. Sci. USA 97, 9026–9031 (2000)
Kwong, P. D. et al. HIV-1 evades antibody-mediated neutralization through conformational masking of receptor-binding sites. Nature 420, 678–682 (2002)
Burton, D. R. et al. Efficient neutralization of primary isolates of HIV-1 by a recombinant human monoclonal antibody. Science 266, 1024–1027 (1994)
Pantophlet, R. et al. Fine mapping of the interaction of neutralizing and nonneutralizing monoclonal antibodies with the CD4 binding site of human immunodeficiency virus type 1 gp120. J. Virol. 77, 642–658 (2003)
Etemad-Moghadam, B. et al. Characterization of simian-human immunodeficiency virus envelope glycoprotein epitopes recognized by neutralizing antibodies from infected monkeys. J. Virol. 72, 8437–8445 (1998)
Zwick, M. B. et al. A novel human antibody against human immunodeficiency virus type 1 gp120 is V1, V2, and V3 loop dependent and helps delimit the epitope of the broadly neutralizing antibody immunoglobulin G1 b12. J. Virol. 77, 6965–6978 (2003)
Guo, Q. et al. Biochemical and genetic characterizations of a novel human immunodeficiency virus type 1 inhibitor that blocks gp120–CD4 interactions. J. Virol. 77, 10528–10536 (2003)
Madani, N. et al. Localized changes in the gp120 envelope glycoprotein confer resistance to human immunodeficiency virus entry inhibitors BMS-806 and #155. J. Virol. 78, 3742–3752 (2004)
Ferrer, M. & Harrison, S. C. Peptide ligands to human immunodeficiency virus type 1 gp120 identified from phage display libraries. J. Virol. 73, 5795–5802 (1999)
Chen, B. et al. A chimeric protein of simian immunodeficiency virus envelope glycoprotein gp140 and Escherichia coli aspartate transcarbamoylase. J. Virol. 78, 4508–4516 (2004)
Moore, J. P., McKeating, J. A., Weiss, R. A. & Sattentau, Q. J. Dissociation of gp120 from HIV-1 virions induced by soluble CD4. Science 250, 1139–1142 (1990)
Pugach, P. et al. The prolonged culture of human immunodeficiency virus type 1 in primary lymphocytes increases its sensitivity to neutralization by soluble CD4. Virology 321, 8–22 (2004)
Puffer, B. A., Altamura, L. A., Pierson, T. C. & Doms, R. W. Determinants within gp120 and gp41 contribute to CD4 independence of SIV Envs. Virology 327, 16–25 (2004)
Kolchinsky, P., Kiprilov, E. & Sodroski, J. Increased neutralization sensitivity of CD4-independent human immunodeficiency virus variants. J. Virol. 75, 2041–2050 (2001)
Puffer, B. A. et al. CD4 independence of simian immunodeficiency virus Envs is associated with macrophage tropism, neutralization sensitivity, and attenuated pathogenicity. J. Virol. 76, 2595–2605 (2002)
Turner, S. et al. Resistance of primary isolates of human immunodeficiency virus type 1 to neutralization by soluble CD4 is not due to lower affinity with the viral envelope glycoprotein gp120. Proc. Natl Acad. Sci. USA 89, 1335–1339 (1992)
Kwong, P. D., Wyatt, R., Sattentau, Q. J., Sodroski, J. & Hendrickson, W. A. Oligomeric modeling and electrostatic analysis of the gp120 envelope glycoprotein of human immunodeficiency virus. J. Virol. 74, 1961–1972 (2000)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
Terwilliger, T. C. & Berendzen, J. Automated MAD and MIR structure solution. Acta Crystallogr. D Biol. Crystallogr. 55, 849–861 (1999)
de la Fortelle, E. & Bricogne, G. Maximum-likelihood heavy-atom parameter refinement for multiple isomorphous replacement and multiwavelength anomalous diffraction methods. Methods Enzymol. 276, 472–493 (1997)
Collaborative Computational Project, N. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Brunger, A. T. et al. Crystallography & NMR system: A new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998)
Jones, T. A., Zou, J. Y., Cowan, S. W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991)
McDonald, I. K. & Thornton, J. M. Satisfying hydrogen bonding potential in proteins. J. Mol. Biol. 238, 777–793 (1994)
Wallace, A. C., Laskowski, R. A. & Thornton, J. M. LIGPLOT: a program to generate schematic diagrams of protein-ligand interactions. Protein Eng. 8, 127–134 (1995)
Acknowledgements
We thank staff at CHESS beamline F1 and APS beamline 19ID for assistance, J. Hoxie of University of Pennsylvania, for hybridomas, and members of the Harrison/Wiley laboratory for discussion. The research was supported by a Scholar Award from the American Foundation for AIDS Research (to B.C.), by the NIH Innovation Grant Program for Approaches in HIV Vaccine Research (to S.C.H. and D.C.W.), and by an NIH HIVRAD grant (to Ellis Reinherz). S.C.H. is, and D.C.W. was, an Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure S1
Analysis by native polyacrylamide gel eletrophoresis of complexes of SIV gp120 core and Fab fragments from a panel of neutralizing monoclonal antibodies. (DOC 87 kb)
Rights and permissions
About this article
Cite this article
Chen, B., Vogan, E., Gong, H. et al. Structure of an unliganded simian immunodeficiency virus gp120 core. Nature 433, 834–841 (2005). https://doi.org/10.1038/nature03327
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03327
This article is cited by
-
Impact of adjuvants on the biophysical and functional characteristics of HIV vaccine-elicited antibodies in humans
npj Vaccines (2022)
-
Entry Inhibitors: Efficient Means to Block Viral Infection
The Journal of Membrane Biology (2020)
-
Predominant envelope variable loop 2-specific and gp120-specific antibody-dependent cellular cytotoxicity antibody responses in acutely SIV-infected African green monkeys
Retrovirology (2018)
-
HIV-1 clade C escapes broadly neutralizing autologous antibodies with N332 glycan specificity by distinct mechanisms
Retrovirology (2016)
-
Glycan shield and epitope masking of a coronavirus spike protein observed by cryo-electron microscopy
Nature Structural & Molecular Biology (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.