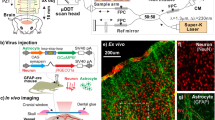
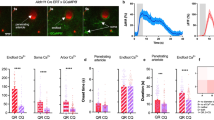
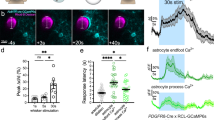
Abstract
Cerebral blood flow (CBF) is coupled to neuronal activity and is imaged in vivo to map brain activation1. CBF is also modified by afferent projection fibres that release vasoactive neurotransmitters2,3 in the perivascular region, principally on the astrocyte endfeet4,5 that outline cerebral blood vessels6. However, the role of astrocytes in the regulation of cerebrovascular tone remains uncertain. Here we determine the impact of intracellular Ca2+ concentrations ([Ca2+]i) in astrocytes on the diameter of small arterioles by using two-photon Ca2+ uncaging7,8 to increase [Ca2+]i. Vascular constrictions occurred when Ca2+ waves evoked by uncaging propagated into the astrocyte endfeet and caused large increases in [Ca2+]i. The vasoactive neurotransmitter noradrenaline2,3 increased [Ca2+]i in the astrocyte endfeet, the peak of which preceded the onset of arteriole constriction. Depressing increases in astrocyte [Ca2+]i with BAPTA inhibited the vascular constrictions in noradrenaline. We find that constrictions induced in the cerebrovasculature by increased [Ca2+]i in astrocyte endfeet are generated through the phospholipase A2–arachidonic acid pathway and 20-hydroxyeicosatetraenoic acid production. Vasoconstriction by astrocytes is a previously unknown mechanism for the regulation of CBF.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chaigneau, E., Oheim, M., Audinat, E. & Charpak, S. Two-photon imaging of capillary blood flow in olfactory bulb glomeruli. Proc. Natl Acad. Sci. USA 100, 13081–13086 (2003)
Raichle, M. E., Hartman, B. K., Eichling, J. O. & Sharpe, L. G. Central noradrenergic regulation of cerebral blood flow and vascular permeability. Proc. Natl Acad. Sci. USA 72, 3726–3730 (1975)
Goadsby, P. J. & Edvinsson, L. in Cerebral Blood Flow and Metabolism (ed. Krause, D. N.) 172–188 (Lippincott Williams & Wilkins, Philadelphia, 2002)
Cohen, Z., Molinatti, G. & Hamel, E. Astroglial and vascular interactions of noradrenaline terminals in the rat cerebral cortex. J. Cereb. Blood Flow Metab. 17, 894–904 (1997)
Paspalas, C. D. & Papadopoulos, G. C. Ultrastructural relationships between noradrenergic nerve fibers and non-neuronal elements in the rat cerebral cortex. Glia 17, 133–146 (1996)
Simard, M., Arcuino, G., Takano, T., Liu, Q. S. & Nedergaard, M. Signaling at the gliovascular interface. J. Neurosci. 23, 9254–9262 (2003)
Soeller, C. & Cannell, M. B. Two-photon microscopy: imaging in scattering samples and three-dimensionally resolved flash photolysis. Microsc. Res. Tech. 47, 182–195 (1999)
Brown, E. B., Shear, J. B., Adams, S. R., Tsien, R. Y. & Webb, W. W. Photolysis of caged calcium in femtoliter volumes using two-photon excitation. Biophys. J. 76, 489–499 (1999)
Zhuo, L. et al. Live astrocytes visualized by green fluorescent protein in transgenic mice. Dev. Biol. 187, 36–42 (1997)
Kang, J. & Nedergaard, M. in Imaging Neurons: A Laboratory Manual (ed. Konnerth, A.) 42.1–42.11 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 2000)
Walz, W. & MacVicar, B. Electrophysiological properties of glial cells: comparison of brain slices with primary cultures. Brain Res. 443, 321–324 (1988)
Duffy, S. & MacVicar, B. A. Adrenergic calcium signaling in astrocyte networks within the hippocampal slice. J. Neurosci. 15, 5535–5550 (1995)
Farooqui, A. A., Yang, H. C., Rosenberger, T. A. & Horrocks, L. A. Phospholipase A2 and its role in brain tissue. J. Neurochem. 69, 889–901 (1997)
Katsuki, H. & Okuda, S. Arachidonic acid as a neurotoxic and neurotrophic substance. Prog. Neurobiol. 46, 607–636 (1995)
Street, I. P. et al. Slow- and tight-binding inhibitors of the 85-kDa human phospholipase A2. Biochemistry 32, 5935–5940 (1993)
Roman, R. J. P-450 metabolites of arachidonic acid in the control of cardiovascular function. Physiol. Rev. 82, 131–185 (2002)
Gebremedhin, D. et al. Production of 20-HETE and its role in autoregulation of cerebral blood flow. Circ. Res. 87, 60–65 (2000)
Miyata, N. et al. HET0016, a potent and selective inhibitor of 20-HETE synthesizing enzyme. Br. J. Pharmacol. 133, 325–329 (2001)
Zonta, M. et al. Neuron-to-astrocyte signaling is central to the dynamic control of brain microcirculation. Nature Neurosci. 6, 43–50 (2003)
Chillon, J. M. & Baumbach, G. L. in Cerebral Blood Flow and Metabolism (ed. Krause, D. N.) 395–412 (Lippincott Williams & Wilkins, Philadelphia, 2002)
Basarsky, T. A., Duffy, S. N., Andrew, R. D. & MacVicar, B. A. Imaging spreading depression and associated intracellular calcium waves in brain slices. J. Neurosci. 18, 7189–7199 (1998)
Dreier, J. P. et al. Ischaemia triggered by spreading neuronal activation is inhibited by vasodilators in rats. J. Physiol. (Lond.) 531, 515–526 (2001)
Duffy, S. & MacVicar, B. A. In vitro ischemia promotes calcium influx and intracellular calcium release in hippocampal astrocytes. J. Neurosci. 16, 71–81 (1996)
Xu, C., Zipfel, W., Shear, J. B., Williams, R. M. & Webb, W. W. Multiphoton fluorescence excitation: new spectral windows for biological nonlinear microscopy. Proc. Natl Acad. Sci. USA 93, 10763–10768 (1996)
Acknowledgements
We thank A. G. Phillips, Y. T. Wang and T. Murphy for comments on the manuscript, and D. Feighan for technical assistance. S.J.M. was supported by a Canadian Heart and Stroke Fellowship. B.A.M. is a Canada Research Chair in Neuroscience and Michael Smith Distinguished Scholar. Work was supported by Canadian Institutes of Health Research and the Canadian Stroke Network.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Mulligan, S., MacVicar, B. Calcium transients in astrocyte endfeet cause cerebrovascular constrictions. Nature 431, 195–199 (2004). https://doi.org/10.1038/nature02827
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02827
This article is cited by
-
Astrocytes modulate cerebral blood flow and neuronal response to cocaine in prefrontal cortex
Molecular Psychiatry (2024)
-
Encephalitis-like episodes with cortical edema and enhancement in patients with neuronal intranuclear inclusion disease
Neurological Sciences (2024)
-
Brain Maturation as a Fundamental Factor in Immune-Neurovascular Interactions in Stroke
Translational Stroke Research (2024)
-
Astrocytes display ultrastructural alterations and heterogeneity in the hippocampus of aged APP-PS1 mice and human post-mortem brain samples
Journal of Neuroinflammation (2023)
-
Aging reduces calreticulin expression and alters spontaneous calcium signals in astrocytic endfeet of the mouse dorsolateral striatum
npj Aging (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.