Abstract
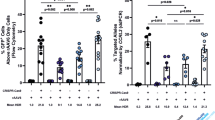
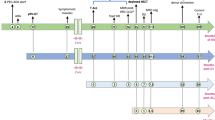
Severe combined immunodeficiency (SCID) patients with an inactivating mutation in recombination activation gene 1 (RAG1) lack B and T cells due to the inability to rearrange immunoglobulin (Ig) and T-cell receptor (TCR) genes. Gene therapy is a valid treatment option for RAG-SCID patients, especially for patients lacking a suitable bone marrow donor, but developing such therapy has proven challenging. As a preclinical model for RAG-SCID, we used Rag1−/− mice and lentiviral self-inactivating (SIN) vectors harboring different internal elements to deliver native or codon-optimized human RAG1 sequences. Treatment resulted in the appearance of B and T cells in peripheral blood and developing B and T cells were detected in central lymphoid organs. Serum Ig levels and Ig and TCR Vβ gene segment usage was comparable to wild-type (WT) controls, indicating that RAG-mediated rearrangement took place. Remarkably, relatively low frequencies of B cells produced WT levels of serum immunoglobulins. Upon stimulation of the TCR, corrected spleen cells proliferated and produced cytokines. In vivo challenge resulted in production of antigen-specific antibodies. No leukemia development as consequence of insertional mutagenesis was observed. The functional reconstitution of the B- as well as the T-cell compartment provides proof-of-principle for therapeutic RAG1 gene transfer in Rag1−/− mice using lentiviral SIN vectors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Notarangelo L, Casanova JL, Conley ME, Chapel H, Fischer A, Puck J et al. Primary immunodeficiency diseases: an update from the International Union of Immunological Societies Primary Immunodeficiency Diseases Classification Committee Meeting in Budapest, 2005. J Allergy Clin Immunol 2006; 117: 883–896.
Recio MJ, Moreno-Pelayo MA, Kilic SS, Guardo AC, Sanal O, Allende LM et al. Differential biological role of CD3 chains revealed by human immunodeficiencies. J Immunol 2007; 178: 2556–2564.
Roberts JL, Lauritsen JP, Cooney M, Parrott RE, Sajaroff EO, Win CM et al. T-B+NK+ severe combined immunodeficiency caused by complete deficiency of the CD3zeta subunit of the T-cell antigen receptor complex. Blood 2007; 109: 3198–3206.
van der Burg M, van Veelen LR, Verkaik NS, Wiegant WW, Hartwig NG, Barendregt BH et al. A new type of radiosensitive T-B-NK+ severe combined immunodeficiency caused by a LIG4 mutation. J Clin Invest 2006; 116: 137–145.
van der Burg M, Ijspeert H, Verkaik NS, Turul T, Wiegant WW, Morotomi-Yano K et al. A DNA-PKcs mutation in a radiosensitive T-B- SCID patient inhibits Artemis activation and nonhomologous end-joining. J Clin Invest 2009; 119: 91–98.
Corneo B, Moshous D, Gungor T, Wulffraat N, Philippet P, Le Deist FL et al. Identical mutations in RAG1 or RAG2 genes leading to defective V(D)J recombinase activity can cause either T-B-severe combined immune deficiency or Omenn syndrome. Blood 2001; 97: 2772–2776.
Noordzij JG, de Bruin-Versteeg S, Verkaik NS, Vossen JM, de Groot R, Bernatowska E et al. The immunophenotypic and immunogenotypic B-cell differentiation arrest in bone marrow of RAG-deficient SCID patients corresponds to residual recombination activities of mutated RAG proteins. Blood 2002; 100: 2145–2152.
Schwarz K, Gauss GH, Ludwig L, Pannicke U, Li Z, Lindner D et al. RAG mutations in human B cell-negative SCID. Science 1996; 274: 97–99.
Villa A, Sobacchi C, Notarangelo LD, Bozzi F, Abinun M, Abrahamsen TG et al. V(D)J recombination defects in lymphocytes due to RAG mutations: severe immunodeficiency with a spectrum of clinical presentations. Blood 2001; 97: 81–88.
Gennery AR, Slatter MA, Grandin L, Taupin P, Cant AJ, Veys P et al. Transplantation of hematopoietic stem cells and long-term survival for primary immunodeficiencies in Europe: entering a new century, do we do better? J Allergy Clin Immunol 2010; 126: 602–610, e1-11.
Aiuti A, Slavin S, Aker M, Ficara F, Deola S, Mortellaro A et al. Correction of ADA-SCID by stem cell gene therapy combined with nonmyeloablative conditioning. Science 2002; 296: 2410–2413.
Gaspar HB, Parsley KL, Howe S, King D, Gilmour KC, Sinclair J et al. Gene therapy of X-linked severe combined immunodeficiency by use of a pseudotyped gammaretroviral vector. Lancet 2004; 364: 2181–2187.
Hacein-Bey-Abina S, Le Deist F, Carlier F, Bouneaud C, Hue C, De Villartay JP et al. Sustained correction of X-linked severe combined immunodeficiency by ex vivo gene therapy. N Engl J Med 2002; 346: 1185–1193.
Jiang Q, Li WQ, Aiello FB, Klarmann KD, Keller JR, Durum SK . Retroviral transduction of IL-7Ralpha into IL-7Ralpha-/- bone marrow progenitors: correction of lymphoid deficiency and induction of neutrophilia. Gene Ther 2005; 12: 1761–1768.
Mostoslavsky G, Fabian AJ, Rooney S, Alt FW, Mulligan RC . Complete correction of murine Artemis immunodeficiency by lentiviral vector-mediated gene transfer. Proc Natl Acad Sci USA 2006; 103: 16406–16411.
Yates F, Malassis-Seris M, Stockholm D, Bouneaud C, Larousserie F, Noguiez-Hellin P et al. Gene therapy of RAG-2-/- mice: sustained correction of the immunodeficiency. Blood 2002; 100: 3942–3949.
Lagresle-Peyrou C, Yates F, Malassis-Seris M, Hue C, Morillon E, Garrigue A et al. Long-term immune reconstitution in RAG-1-deficient mice treated by retroviral gene therapy: a balance between efficiency and toxicity. Blood 2006; 107: 63–72.
Hacein-Bey-Abina S, Garrigue A, Wang GP, Soulier J, Lim A, Morillon E et al. Insertional oncogenesis in 4 patients after retrovirus-mediated gene therapy of SCID-X1. J Clin Invest 2008; 118: 3132–3142.
Hacein-Bey-Abina S, von Kalle C, Schmidt M, Le Deist F, Wulffraat N, McIntyre E et al. A serious adverse event after successful gene therapy for X-linked severe combined immunodeficiency. N Engl J Med 2003; 348: 255–256.
Staal FJ, Pike-Overzet K, Ng YY, van Dongen JJ . Sola dosis facit venenum. Leukemia in gene therapy trials: a question of vectors, inserts and dosage? Leukemia 2008; 22: 1849–1852.
Bukovsky AA, Song JP, Naldini L . Interaction of human immunodeficiency virus-derived vectors with wild-type virus in transduced cells. J Virol 1999; 73: 7087–7092.
Dull T, Zufferey R, Kelly M, Mandel RJ, Nguyen M, Trono D et al. A third-generation lentivirus vector with a conditional packaging system. J Virol 1998; 72: 8463–8471.
Naldini L, Blomer U, Gallay P, Ory D, Mulligan R, Gage FH et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 1996; 272: 263–267.
Zufferey R, Nagy D, Mandel RJ, Naldini L, Trono D . Multiply attenuated lentiviral vector achieves efficient gene delivery in vivo. Nat Biotechnol 1997; 15: 871–875.
Yu SF, von Ruden T, Kantoff PW, Garber C, Seiberg M, Ruther U et al. Self-inactivating retroviral vectors designed for transfer of whole genes into mammalian cells. Proc Natl Acad Sci USA 1986; 83: 3194–3198.
Zufferey R, Dull T, Mandel RJ, Bukovsky A, Quiroz D, Naldini L et al. Self-inactivating lentivirus vector for safe and efficient in vivo gene delivery. J Virol 1998; 72: 9873–9880.
Wu X, Li Y, Crise B, Burgess SM . Transcription start regions in the human genome are favored targets for MLV integration. Science 2003; 300: 1749–1751.
Lewinski MK, Yamashita M, Emerman M, Ciuffi A, Marshall H, Crawford G et al. Retroviral DNA integration: viral and cellular determinants of target-site selection. PLoS Pathog 2006; 2: e60.
Lagresle-Peyrou C, Benjelloun F, Hue C, Andre-Schmutz I, Bonhomme D, Forveille M et al. Restoration of human B-cell differentiation into NOD-SCID mice engrafted with gene-corrected CD34+ cells isolated from Artemis or RAG1-deficient patients. Mol Ther 2008; 16: 396–403.
Ng YY, Baert MR, Pike-Overzet K, Rodijk M, Brugman MH, Schambach A et al. Correction of B-cell development in Btk-deficient mice using lentiviral vectors with codon-optimized human BTK. Leukemia 2010; 24: 1617–1630.
Mombaerts P, Iacomini J, Johnson RS, Herrup K, Tonegawa S, Papaioannou VE . RAG-1-deficient mice have no mature B and T lymphocytes. Cell 1992; 68: 869–877.
Salmon P, Kindler V, Ducrey O, Chapuis B, Zubler RH, Trono D . High-level transgene expression in human hematopoietic progenitors and differentiated blood lineages after transduction with improved lentiviral vectors. Blood 2000; 96: 3392–3398.
Zhang F, Frost AR, Blundell MP, Bales O, Antoniou MN, Thrasher AJ . A ubiquitous chromatin opening element (UCOE) confers resistance to DNA methylation-mediated silencing of lentiviral vectors. Mol Ther 2010; 18: 1640–1649.
Donello JE, Loeb JE, Hope TJ . Woodchuck hepatitis virus contains a tripartite posttranscriptional regulatory element. J Virol 1998; 72: 5085–5092.
Zufferey R, Donello JE, Trono D, Hope TJ . Woodchuck hepatitis virus posttranscriptional regulatory element enhances expression of transgenes delivered by retroviral vectors. J Virol 1999; 73: 2886–2892.
Pannetier C, Cochet M, Darche S, Casrouge A, Zoller M, Kourilsky P . The sizes of the CDR3 hypervariable regions of the murine T-cell receptor beta chains vary as a function of the recombined germ-line segments. Proc Natl Acad Sci USA 1993; 90: 4319–4323.
van Dongen JJ, Langerak AW, Bruggemann M, Evans PA, Hummel M, Lavender FL et al. Design and standardization of PCR primers and protocols for detection of clonal immunoglobulin and T-cell receptor gene recombinations in suspect lymphoproliferations: report of the BIOMED-2 Concerted Action BMH4-CT98-3936. Leukemia 2003; 17: 2257–2317.
Fugmann SD, Villey IJ, Ptaszek LM, Schatz DG . Identification of two catalytic residues in RAG1 that define a single active site within the RAG1/RAG2 protein complex. Mol Cell 2000; 5: 97–107.
Kane JF . Effects of rare codon clusters on high-level expression of heterologous proteins in Escherichia coli. Curr Opin Biotechnol 1995; 6: 494–500.
Zychlinski D, Schambach A, Modlich U, Maetzig T, Meyer J, Grassman E et al. Physiological promoters reduce the genotoxic risk of integrating gene vectors. Mol Ther 2008; 16: 718–725.
Montini E, Cesana D, Schmidt M, Sanvito F, Bartholomae CC, Ranzani M et al. The genotoxic potential of retroviral vectors is strongly modulated by vector design and integration site selection in a mouse model of HSC gene therapy. J Clin Invest 2009; 119: 964–975.
Foster H, Sharp PS, Athanasopoulos T, Trollet C, Graham IR, Foster K et al. Codon and mRNA sequence optimization of microdystrophin transgenes improves expression and physiological outcome in dystrophic mdx mice following AAV2/8 gene transfer. Mol Ther 2008; 16: 1825–1832.
Moreno-Carranza B, Gentsch M, Stein S, Schambach A, Santilli G, Rudolf E et al. Transgene optimization significantly improves SIN vector titers, gp91phox expression and reconstitution of superoxide production in X-CGD cells. Gene Ther 2009; 16: 111–118.
Wayne J, Suh H, Misulovin Z, Sokol KA, Inaba K, Nussenzweig MC . A regulatory role for recombinase activating genes, RAG-1 and RAG-2, in T cell development. Immunity 1994; 1: 95–107.
Shinkai Y, Rathbun G, Lam KP, Oltz EM, Stewart V, Mendelsohn M et al. RAG-2-deficient mice lack mature lymphocytes owing to inability to initiate V(D)J rearrangement. Cell 1992; 68: 855–867.
Hacein-Bey-Abina S, Hauer J, Lim A, Picard C, Wang GP, Berry CC et al. Efficacy of gene therapy for X-linked severe combined immunodeficiency. N Engl J Med 2010; 363: 355–364.
Sandrin V, Boson B, Salmon P, Gay W, Negre D, Le Grand R et al. Lentiviral vectors pseudotyped with a modified RD114 envelope glycoprotein show increased stability in sera and augmented transduction of primary lymphocytes and CD34+ cells derived from human and nonhuman primates. Blood 2002; 100: 823–832.
Anliker B, Abel T, Kneissl S, Hlavaty J, Caputi A, Brynza J et al. Specific gene transfer to neurons, endothelial cells and hematopoietic progenitors with lentiviral vectors. Nat Methods 2010; 7: 929–935.
Acknowledgements
We thank Edwin de Haas for cell sorting and all other members of the Staal laboratory for their technical contributions. We thank Alexandre G Troullioud Lucas for his contribution to testing the coRAG1 vectors. We are grateful for the biotechnical assistance of Yvette Caljouw in the mouse facility. We are grateful to L Naldini (San Raffaele Telethon Institute for Gene Therapy Milano, Italy) for providing the helper plasmids pMDLg/pRRE, pRSV-Rev and pMD2.VSVG, and the original transfer vector. This work was in part supported by grants of the European Union: INHERINET (QLK3-CT-2001-0427) and CONSERT (LSHB-CT-2004-005242), in part by grants of the Netherlands Organization for Health Research and Development (Translational Gene Therapy Research Programme of ZonMw, 43100016 and Innovational Research Incentives Scheme Veni grant, 91610083), Kika (Children Cancer Free, 09/036) and REBIRTH Excellence Cluster (Exc62/1).
Author contributions
KPO, FJTS, JJMvD and CB designed research; KPO, MR, YYN and MRMB performed experiments; AS, FZ, AJT and CB contributed vital new reagents; KPO, MR and FJTS analyzed data; KPO and FJTS interpreted data; YYN, AS, RCH, ACL, RGMB, GJAD, AJT, CB, MCC, JJMvD, AL and RH contributed in essential discussions. CL, SHBA and MCC contributed unpublished experiments that helped the direction of the work; KPO and FJTS wrote manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Pike-Overzet, K., Rodijk, M., Ng, YY. et al. Correction of murine Rag1 deficiency by self-inactivating lentiviral vector-mediated gene transfer. Leukemia 25, 1471–1483 (2011). https://doi.org/10.1038/leu.2011.106
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.106
Keywords
This article is cited by
-
The “Editors” Take to RAG: Promise of CRISPR/Cas9/rAAV6-Based Gene Therapy for RAG2 Deficiency
Journal of Clinical Immunology (2021)
-
Gene Editing Rescues In vitro T Cell Development of RAG2-Deficient Induced Pluripotent Stem Cells in an Artificial Thymic Organoid System
Journal of Clinical Immunology (2021)
-
RAG Deficiency: Two Genes, Many Diseases
Journal of Clinical Immunology (2018)
-
Hematopoietic stem cell gene therapy for the cure of blood diseases: primary immunodeficiencies
Rendiconti Lincei. Scienze Fisiche e Naturali (2018)
-
Gene therapy for primary immune deficiencies: a Canadian perspective
Allergy, Asthma & Clinical Immunology (2017)