Abstract
Tamoxifen contributes to decreased recurrence and mortality of patients with hormone receptor-positive breast cancer. As this drug is metabolized by phase I and phase II enzymes, the interindividual variations of their enzymatic activity are thought to be associated with individual responses to tamoxifen. Among these enzymes, CYP2D6 is considered to be a rate-limiting enzyme in the generation of endoxifen, a principal active metabolite of tamoxifen, and the genetic polymorphisms of CYP2D6 have been extensively investigated in association with the plasma endoxifen concentrations and clinical outcome of tamoxifen therapy. In addition to CYP2D6, other genetic factors including polymorphisms in various drug-metabolizing enzymes and drug transporters have been implicated to their relations to clinical outcome of tamoxifen therapy, but their effects would be small. Although the results of association studies are controversial, accumulation of the evidence has revealed us the important and critical issues in the tamoxifen pharmacogenomics study, namely the quality of genotyping, the coverage of genetic variations, the criteria for sample collection and the source of DNAs, which are considered to be common problematic issues in pharmacogenomics studies. This review points out common critical issues in pharmacogenomics studies through the lessons we have learned from tamoxifen pharmacogenomics, as well as summarizes the results of pharmacogenomics studies for tamoxifen treatment.
Similar content being viewed by others
Introduction
Tamoxifen, a selective estrogen receptor (ER) modulator, has been widely used for the treatment and prevention of recurrence for patients with hormone receptor (ER or progesterone receptor)-positive breast cancers. As >70% of breast cancers are hormone receptor-positive, thousands of breast cancer patients worldwide initiate to take endocrine treatment including tamoxifen each year. In pre- and postmenopausal patients with primary breast cancer, 5 years of adjuvant tamoxifen significantly reduced recurrence rate as well as cancer-specific mortality for 15 years after their primary diagnosis.1 However, approximately one-third of patients treated with adjuvant tamoxifen experience a recurrent disease,1, 2 implicating possible individual differences in responsiveness to tamoxifen.
Tamoxifen is metabolized to more active metabolites or inactive forms by phase I and phrase II enzymes, including cytochrome P450s (CYPs), sulfotransferases (SULTs) and UDP-glucuronosyltransferases (UGTs). The polymorphisms in these drug-metabolizing enzymes are considered to affect individual differences in plasma concentrations of active tamoxifen metabolites and clinical outcome in breast cancer patients treated with tamoxifen. Among these enzymes, CYP2D6 has been most extensively investigated owing to its significant role in production of active metabolites, endoxifen and 4-hydroxytamoxifen.
This review summarizes current reports on the relationships of genetic polymorphisms and other biomarkers to individual differences in clinical outcome of breast cancer patients with tamoxifen treatment. In addition, we investigate reasons or causes of discordant results for the association between CYP2D6 genetic variations and clinical outcome, and would like to highlight various problematic issues in pharmacogenomics studies.
Tamoxifen metabolism
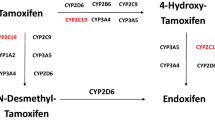
Tamoxifen is extensively metabolized by phase I and phase II enzymes in the human liver (Figure 1).3, 4 Tamoxifen itself has low affinity to the ER as only 1.8% of the affinity of 17β-estradiol.3 The major metabolite N-desmethyltamoxifen is formed by N-demethylation, which is catalyzed mainly by CYP3A4 and CYP3A5, with small contribution by CYP2D6, CYP1A2, CYP2C9 and CYP2C19.5, 6, 7, 8, 9 N-desmethyltamoxifen shows weak affinity to the ER similar to tamoxifen.3, 4 However, 4-hydroxytamoxifen, which is formed by 4-hydroxylation of tamoxifen, has 100-fold higher affinity to the ER and 30- to 100-fold greater potency in suppressing estrogen-dependent breast cancer cell proliferation than tamoxifen.3, 10, 11, 12 This conversion is catalyzed by CYP2D6, CYP2B6, CYP2C9, CYP2C19 and CYP3A4.5, 13, 14, 15 Endoxifen (4-hydroxy-N-desmethyltamoxifen) has a potency equivalent to 4-hydroxytamoxifen,10, 16, 17 and its plasma concentration level exceed that of 4-hydroxytamoxifen by several folds, suggesting endoxifen to be a principal active metabolite.9, 10, 11 Endoxifen formation from N-desmethyltamoxifen is predominantly catalyzed by CYP2D6.18 Several additional metabolites, such as N,N-didesmethyltamoxifen, 4′-hydroxy-N-desmethyltamoxifen and α-hydroxytamoxifen were reported, but no other highly active metabolite has been described so far.4
Tamoxifen and these metabolites are further metabolized by phase II enzymes, such as SULTs and UGTs. SULT1A1 is considered to be the primary SULT responsible for the sulfation of 4-hydroxytamoxifen and endoxifen.19, 20 UGT1A8, UGT1A10, UGT2B7, UGT2B15 and UGT1A4 are involved in the O-glucuronidation of 4-hydroxytamoxifen and endoxifen.21, 22, 23 Tamoxifen and 4-hydroxytamoxifen are glucuronidated by UGT1A4 to the corresponding N+-glucuronides.24, 25 The genetic variations of these drug-metabolizing enzymes are possible to affect tamoxifen metabolism.
Genetic polymorphisms of CYP2D6
CYP2D6 is one of the most important CYP isoforms owing to its central role in the metabolism of a number of clinically important drugs.26 The CYP2D6 gene is located on chromosome 22q13.1, containing two neighboring pseudogenes, CYP2D7 and CYP2D8. This locus is extremely polymorphic with over 80 allelic variants, a subset of which should affect the gene product and result in wide interindividual and ethnic differences in CYP2D6 activity.27 Commonly, four CYP2D6 phenotypes are defined on the basis of their in vivo metabolic capacities: poor metabolizer (PM), intermediate metabolizer (IM), extensive metabolizer (EM) and ultra-rapid metabolizer (UM).28, 29 It has been reported that the PM phenotype, which is caused by carrying two null alleles, is present in 5–10% of Caucasians.30 The CYP2D6*3, CYP2D6*4, CYP2D6*5 and CYP2D6*6 are major null alleles that are related to the PM phenotype and account for nearly 95% of the PMs in Caucasians.31 Among them, CYP2D6*4 shows the highest frequency as 17.5–23.0%.27 CYP2D6*5, which is found at a frequency of ∼5%, lacks an entire CYP2D6 gene. In contrast, <1% of Asians show the PM phenotype,32 and most Asians are categorized as IMs because of the high frequency of a CYP2D6*10 allele.33, 34 The CYP2D6*14, CYP2D6*18, CYP2D6*21, CYP2D6*36 and CYP2D6*44 were null alleles found in Asian populations, although their frequencies are very low.35, 36, 37, 38 The frequencies of UMs, who carry a duplicated/multiplied wild-type CYP2D6 gene(s), are 10–15% in Caucasian, whereas UMs are uncommon in Asians. As described here, because the CYP2D6 gene locus is complex, genotyping of CYP2D6 variants, especially CYP2D6*5, is technically not so easy. Although the accuracy of genotyping partly depends on the quality of DNAs and the platforms of genotyping, wrong genotyping results sometimes cause incorrect interpretation of the research outcome, and result in both false-positive conclusions and false-negative conclusions.
CYP2D6 genotype and clinical outcome of tamoxifen therapy
In recent years, we have seen an explosion of interest in the clinical relevance of CYP2D6 genotype on outcome of breast cancer patients who are treated with tamoxifen. Prospective cohort studies of adjuvant tamoxifen treatment have revealed a wide interindividual variation in the steady state plasma concentrations of active metabolites, endoxifen and 4-hydroxytamoxifen during tamoxifen treatment in patients carrying CYP2D6 genetic variants.8, 9, 11 The patients homozygous for null alleles (categorized as PM) showed nearly one-fourth of endoxifen concentration in plasma, compared with those carrying two normal alleles (categorized as EM).8, 9 The patients carrying two alleles that encode a low-function enzyme, including CYP2D6*10 and CYP2D6*41 (categorized as intermediate metabolizer), had nearly 50% of plasma endoxifen concentration compared with the controls.4, 39, 40, 41 These patients with low endoxifen concentration were suspected to have a poorer clinical outcome.
As shown in Table 1, a number of studies have reported the association between the CYP2D6 genotype and clinical outcome of breast cancer patients receiving the tamoxifen therapy. One of the first studies reported by Goetz et al.42, 43 demonstrated that homozygous carriers of CYP2D6*4 allele had a shorter relapse-free survival (RFS) and disease-free survival than the patients for heterozygous or homozygous for the wild-type allele (hazard ratio (HR), 1.85; P=0.18 for RFS: HR, 1.86; P=0.089 for disease-free survival). Following these reports, Schroth et al.44 published retrospective analysis of 1,325 breast cancer patients with adjuvant tamoxifen monotherapy, and observed that PMs revealed a significantly higher risk of recurrence than EMs with HR of 2.12 for a time to recurrence (P=0.003). These associations were supported by several research groups.45, 46, 47, 48, 49, 50 In Asians, we reported the significant effects of CYP2D6 genotype (especially CYP2D6*10) on RFS in Japanese patients receiving adjuvant tamoxifen monotherapy (HR, 9.52; P=0.000036).40, 51 The worse clinical outcome of tamoxifen therapy in the patients carrying CYP2D6*10 was confirmed in Chinese, Korean, Thai and Malaysian populations.52, 53, 54, 55 However, several discordant results have been also reported.56, 57, 58, 59, 60, 61 More recently, two retrospective analyses of large prospective trails, the ATAC (Alimidex, Tamoxifen, Alone or in Combination) trial and the Breast International Group (BIG) 1-98 trial, were reported.62, 63 In the ATAC analysis, there was no significant association between any of CYP2D6 phenotypic groups and recurrence rates in 588 patients treated with tamoxifen (HR, 1.22; P=0.44; PM relative to EM).62 Similarly, in the BIG 1-98 analysis, no significant difference was found among different CYP2D6 metabolizer groups and cancer-free survival in 973 breast cancer patients (HR, 0.58; P=0.35; EM vs PM).63 As discussed in previous reports, there may be several confounding factors or critical errors in the experimental designs to explain these discrepancies.
One of the most important issues in the pharmacogenomics study is the quality of genotype data. This should be influenced by (i) the accuracy of genotyping methods, (ii) coverage of genotyped alleles and (iii) DNA source. In both of the ATAC and BIG 1-98 studies,62, 63 the authors mentioned the high reproducibility of genotyping methods because of the concordance of genotyping results in duplicate determinations. However, this does not fully guarantee the accuracy of their genotype results. Their genotype results were highly deviated from Hardy–Weinberg equilibrium (χ2 P=10−92 for CYP2D6*4) probably because they used the low-quality genomic DNA extracted from formalin-fixed paraffin-embedded tumor tissues.64, 65, 66, 67 Therefore, they excluded CYP2D6*5 from the analyses, and performed 60-cycle PCR to detect 1846 G>A (CYP2D6*4), which is likely to lead to the misgenotyping results. The importance of wide coverage of CYP2D6 alleles was clearly demonstrated by Schroth et al.68 In the report, the increase of genotyping coverage was shown to increase HR for RFS as well as enhance the statistical power. In our samples, we also detected a lower HR of 5.83 without CYP2D6*5 genotyping data than that of 9.52 (wt/wt vs V/V, N=282; unpublished data). In addition, nearly 30% frequency of loss of heterozygosity at the chromosome 22q, where the CYP2D6 gene is located, in breast cancer cells definitely causes misclassification of patients and leads to misinterpretation of the results if one uses DNAs isolated from tumor tissues (particularly caner-cell rich samples).69
The second critical issue is selection of study participants. To evaluate the effects of CYP2D6 genotype on tamoxifen efficacy, it is scientifically certain that the patients treated only with tamoxifen should be selected. As shown in Table 1, most of studies showing the ‘null’ association included the patients who were treated with a combination of tamoxifen and chemotherapy. We reported significant effects of CYP2D6 genotypes on shorter RFS when we analyzed patients treated with the tamoxifen monotherapy (HR, 9.52; P=0.0032; N=282), but not when we analyzed those with the combination chemotherapy (HR, 0.64; P=0.44; N=167).70, 71 In a combined population (total 449 patents, including 37.2% of those with the combination therapy), HR dropped to 2.45 (95% confidence interval, 1.30–4.54) for wt/wt vs V/V (unpublished data).
These lines of evidence clearly tell us the importance of complete CYP2D6 genotyping using germline DNAs isolated from very carefully selected samples with tamoxifen monotherapy. All of ‘null’ association studies lacked one or multiple elements of these essential factors, as shown in Table 1. Therefore, large prospective studies satisfying these conditions are needed to make a definite conclusion for the value of CYP2D6 genotyping in tamoxifen therapy.
The patients carrying decreased- or impaired-function CYP2D6 alleles consistently showed lower plasma endoxifen concentrations than those having the homozygous normal genotype.4, 8, 9, 11, 39, 40, 41 Plasma endoxifen levels were suggested to associate with clinical outcome of tamoxifen-treated patients.72 Therefore, several research groups recently conducted CYP2D6 genotype-based dose-adjustment studies.73, 74, 75 Irvin et al.74 demonstrated that endoxifen levels were significantly increased when the dose was increased from 20–40 mg in intermediate metabolizer and PM patients; however, endoxifen levels in PM patients were still significantly lower than the normal individuals. We also investigated the effects of the increase of tamoxifen dose from 20 to 30 mg or 40 mg in the patients heterozygous or homozygous for variant alleles, respectively, and demonstrated that endoxifen concentrations were significantly increased to a similar level of the CYP2D6-normal patients who took 20 mg of tamoxifen (Figure 2).75 In these studies, the incidence of adverse events was not affected by the dose adjustment. Although further verification is required especially for PM patients, these results suggest that increased tamoxifen dose is an effective way to maintain the effective endoxifen concentration for the patients carrying decreased function or null alleles of CYP2D6.
Steady state plasma concentration of endoxifen before and after dose escalation of tamoxifen in breast cancer patients. The horizontal line indicates the median concentration, the box covers the 25th–75th percentiles, and the maximum length of each whisker is 1.5 × the interquartile range, dots outside the whiskers are outliers. Data from Kiyotani et al.75
Polymorphisms in other genes and clinical outcome of tamoxifen therapy
Other CYPs, including CYP2C9, CYP2C19, CYP3A4 and CYP3A5, UGTs and SULTs are also involved in the metabolism of tamoxifen. Among them, CYP3A5*3 is well investigated in association with tamoxifen metabolism or clinical outcome of tamoxifen therapy; however, no significant association was observed.4, 9, 42, 45, 76, 77 For CYP2C19, a significant association with clinical outcome of tamoxifen treatment was found in carriers of CYP2C19*17,45 but not in the carriers of CYP2C19*2 or CYP2C19*3.45, 59 However, the results have also been contradictive and not conclusive.78, 79 Several investigations on genetic variations in the SULT1A1 gene, including single-nucleotide polymorphisms (SNPs) and copy number variations, found no clear association with tamoxifen efficacy56, 58, 79 and tamoxifen metabolism.9, 57 Further analysis would be required by consideration of ‘allele copy number’ of SULT1A1, as demonstrated in the case of CYP2D6.80, 81, 82
There are several reports investigating the involvement of drug transporters in disposition of tamoxifen and its active metabolites, endoxifen and 4-hydroxytamoxifen. ABCB1 (P-glycoprotein, multidrug resistance protein 1) is an ATP-dependent, efflux transporter with broad substrate specificity widely appreciated for its role in mediating cellular resistance to many anticancer agents.83 ABCB1 is reported to be involved in the transport of active tamoxifen metabolites.84, 85 Several ABCB1 polymorphisms have been reported, including 2667 G>A/T and 3435C>T; however, no SNPs were significantly associated with clinical outcome of tamoxifen therapy.40, 54 ABCC2 (multidrug resistance-associated protein 2) has an important role in the biliary excretion of glucuronides or sulfates of drugs, including tamoxifen and its metabolites.17 We found an intronic SNP of ABCC2 (rs3740065), which is in strong linkage disequilibrium (r2=0.89) with −1774 G/delG, to be significantly associated with clinical outcome of patients with tamoxifen therapy through the screening using haplotype-tagging SNPs.40, 86 An in vitro study reporting that ABCC2 was expressed at higher levels in tamoxifen-resistant breast cancer cells suggests the possibility that active metabolites of tamoxifen are transported by ABCC2 from breast cancer cells.87
We also identified a novel locus, containing C10orf11, associated with RFS in the breast cancer patients treated with tamoxifen alone by the genome-wide association study encompassing a total of 462 Japanese patients (HR, 4.53; P=6.28 × 10−8).88 At present, however, no report is available regarding the function of the C10orf11 protein. Large-scale replication study and further functional analysis are required to verify these associations, and to clarify their biological significance and mechanisms that have effects on the clinical outcome of patients receiving tamoxifen therapy.
Other factors affecting clinical outcome of tamoxifen therapy
As well as the genetic polymorphisms modifying the tamoxifen pharmacokinetics, characteristic of cancers, including gene expression profiles or genomic alterations, are also one of important determinants of individual response to tamoxifen. Many molecules have been identified to be involved in the tamoxifen resistance.89, 90 Several microarray analyses revealed the gene signatures to predict the outcome of adjuvant tamoxifen therapy, such as breast cancer intrinsic subtype,91, 92 21-gene signature (used as OncotypeDX)93 and HOXB13/IL17BR expression ratio.94, 95 Goetz et al.96 reported that combination of CYP2D6 genotype and HOXB13/IL17BR was significantly associated with disease-free survival (log-rank P=0.004) and overall survival (log-rank P=0.009). More recently, Ellis et al.97 clarified the elevated frequency of somatic mutations and genome-structure changes in aromatase inhibitor-resistant tumors by whole-genome sequencing. Therefore, prediction of individual response to tamoxifen using cancer characteristics seems to be effective, and may affect the association results of genetic markers.
Conclusion
Although a large number of investigations on tamoxifen pharmacogenomics have been performed, the association results between CYP2D6 genotype and clinical outcome are still controversial. However, accumulation of the evidence clarifies some of the causes of these controversial results, particularly some scientific issues in the false-negative results, and implies the importance of the quality of genotyping as well as sample selections in the tamoxifen pharmacogenomics study. The important issues learned from the tamoxifen and breast cancer studies are commonly applicable in pharmacogenomics studies. As we are aiming to establish the personalized medicine system in which we select a right patient and provide an appropriate dose of a right drug, the pharmacogenomics study also requires the accurate genotyping using a sufficient number of appropriate patients in order to obtain truly positive results and avoid false-positive and false-negative results. Finally, genotype-guided dose-adjustment based on the CYP2D6 genotypes will be a good example for the personalized medicine. To reduce the medical care cost without losing the quality of medical care, it is very important to use the drugs, which are available at lower cost, on the basis of individual genetic information. As several novel associated SNPs/loci have been identified, integration of genotypes of CYP2D6 and other genes as well as tumor characteristics should be the future approach to predict clinical efficacy of tamoxifen and provide better quality of lives to breast cancer patients.
References
Early Breast Cancer Trialists' Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 365, 1687–1717 (2005).
Early Breast Cancer Trialists' Collaborative Group (EBCTCG). Tamoxifen for early breast cancer: an overview of the randomised trials. Lancet 351, 1451–1467 (1998).
Wakeling, A. E. & Slater, S. R. Estrogen-receptor binding and biologic activity of tamoxifen and its metabolites. Cancer Treat. Rep. 64, 741–744 (1980).
Mürdter, T. E., Schroth, W., Bacchus-Gerybadze, L., Winter, S., Heinkele, G., Simon, W. et al. Activity levels of tamoxifen metabolites at the estrogen receptor and the impact of genetic polymorphisms of phase I and II enzymes on their concentration levels in plasma. Clin. Pharmacol. Ther. 89, 708–717 (2011).
Boocock, D. J., Brown, K., Gibbs, A. H., Sanchez, E., Turteltaub, K. W. & White, I. N. Identification of human CYP forms involved in the activation of tamoxifen and irreversible binding to DNA. Carcinogenesis 23, 1897–1901 (2002).
Crewe, H. K., Notley, L. M., Wunsch, R. M., Lennard, M. S. & Gillam, E. M. Metabolism of tamoxifen by recombinant human cytochrome P450 enzymes: formation of the 4-hydroxy, 4′-hydroxy and N-desmethyl metabolites and isomerization of trans-4-hydroxytamoxifen. Drug Metab. Dispos. 30, 869–874 (2002).
Coller, J. K., Krebsfaenger, N., Klein, K., Wolbold, R., Nussler, A., Neuhaus, P. et al. Large interindividual variability in the in vitro formation of tamoxifen metabolites related to the development of genotoxicity. Br. J. Clin. Pharmacol. 57, 105–111 (2004).
Borges, S., Desta, Z., Li, L., Skaar, T. C., Ward, B. A., Nguyen, A. et al. Quantitative effect of CYP2D6 genotype and inhibitors on tamoxifen metabolism: implication for optimization of breast cancer treatment. Clin. Pharmacol. Ther. 80, 61–74 (2006).
Jin, Y., Desta, Z., Stearns, V., Ward, B., Ho, H., Lee, K. H. et al. CYP2D6 genotype, antidepressant use, and tamoxifen metabolism during adjuvant breast cancer treatment. J. Natl. Cancer Inst. 97, 30–39 (2005).
Johnson, M. D., Zuo, H., Lee, K. H., Trebley, J. P., Rae, J. M., Weatherman, R. V. et al. Pharmacological characterization of 4-hydroxy-N-desmethyl tamoxifen, a novel active metabolite of tamoxifen. Breast Cancer Res. Treat. 85, 151–159 (2004).
Stearns, V., Johnson, M. D., Rae, J. M., Morocho, A., Novielli, A., Bhargava, P. et al. Active tamoxifen metabolite plasma concentrations after coadministration of tamoxifen and the selective serotonin reuptake inhibitor paroxetine. J. Natl. Cancer Inst. 95, 1758–1764 (2003).
Clarke, R., Liu, M. C., Bouker, K. B., Gu, Z., Lee, R. Y., Zhu, Y. et al. Antiestrogen resistance in breast cancer and the role of estrogen receptor signaling. Oncogene 22, 7316–7339 (2003).
Crewe, H. K., Ellis, S. W., Lennard, M. S. & Tucker, G. T. Variable contribution of cytochromes P450 2D6, 2C9 and 3A4 to the 4-hydroxylation of tamoxifen by human liver microsomes. Biochem. Pharmacol. 53, 171–178 (1997).
Dehal, S. S. & Kupfer, D. CYP2D6 catalyzes tamoxifen 4-hydroxylation in human liver. Cancer Res. 57, 3402–3406 (1997).
Coller, J. K., Krebsfaenger, N., Klein, K., Endrizzi, K., Wolbold, R., Lang, T. et al. The influence of CYP2B6, CYP2C9 and CYP2D6 genotypes on the formation of the potent antioestrogen Z-4-hydroxy-tamoxifen in human liver. Br. J. Clin. Pharmacol. 54, 157–167 (2002).
Borgna, J. L. & Rochefort, H. Hydroxylated metabolites of tamoxifen are formed in vivo and bound to estrogen receptor in target tissues. J. Biol. Chem. 256, 859–868 (1981).
Lien, E. A., Solheim, E., Lea, O. A., Lundgren, S., Kvinnsland, S. & Ueland, P. M. Distribution of 4-hydroxy-N-desmethyltamoxifen and other tamoxifen metabolites in human biological fluids during tamoxifen treatment. Cancer Res. 49, 2175–2183 (1989).
Desta, Z., Ward, B. A., Soukhova, N. V. & Flockhart, D. A. Comprehensive evaluation of tamoxifen sequential biotransformation by the human cytochrome P450 system in vitro: prominent roles for CYP3A and CYP2D6. J. Pharmacol. Exp. Ther. 310, 1062–1075 (2004).
Falany, J. L., Pilloff, D. E., Leyh, T. S. & Falany, C. N. Sulfation of raloxifene and 4-hydroxytamoxifen by human cytosolic sulfotransferases. Drug Metab. Dispos. 34, 361–368 (2006).
Gjerde, J., Hauglid, M., Breilid, H., Lundgren, S., Varhaug, J. E., Kisanga, E. R. et al. Effects of CYP2D6 and SULT1A1 genotypes including SULT1A1 gene copy number on tamoxifen metabolism. Ann. Oncol. 19, 56–61 (2008).
Nishiyama, T., Ogura, K., Nakano, H., Ohnuma, T., Kaku, T., Hiratsuka, A. et al. Reverse geometrical selectivity in glucuronidation and sulfation of cis- and trans-4-hydroxytamoxifens by human liver UDP-glucuronosyltransferases and sulfotransferases. Biochem. Pharmacol. 63, 1817–1830 (2002).
Ogura, K., Ishikawa, Y., Kaku, T., Nishiyama, T., Ohnuma, T., Muro, K. et al. Quaternary ammonium-linked glucuronidation of trans-4-hydroxytamoxifen, an active metabolite of tamoxifen, by human liver microsomes and UDP-glucuronosyltransferase 1A4. Biochem. Pharmacol. 71, 1358–1369 (2006).
Sun, D., Sharma, A. K., Dellinger, R. W., Blevins-Primeau, A. S., Balliet, R. M., Chen, G. et al. Glucuronidation of active tamoxifen metabolites by the human UDP glucuronosyltransferases. Drug Metab. Dispos. 35, 2006–2014 (2007).
Kaku, T., Ogura, K., Nishiyama, T., Ohnuma, T., Muro, K. & Hiratsuka, A. Quaternary ammonium-linked glucuronidation of tamoxifen by human liver microsomes and UDP-glucuronosyltransferase 1A4. Biochem. Pharmacol. 67, 2093–2102 (2004).
Sun, D., Chen, G., Dellinger, R. W., Duncan, K., Fang, J. L. & Lazarus, P. Characterization of tamoxifen and 4-hydroxytamoxifen glucuronidation by human UGT1A4 variants. Breast Cancer Res. 8, R50 (2006).
Ingelman-Sundberg, M. Genetic polymorphisms of cytochrome P450 2D6 (CYP2D6): clinical consequences, evolutionary aspects and functional diversity. Pharmacogenomics J. 5, 6–13 (2005).
Bradford, L. D. CYP2D6 allele frequency in European Caucasians, Asians, Africans and their descendants. Pharmacogenomics 3, 229–243 (2002).
Sachse, C., Brockmoller, J., Bauer, S. & Roots, I. Cytochrome P450 2D6 variants in a Caucasian population: allele frequencies and phenotypic consequences. Am. J. Hum. Genet. 60, 284–295 (1997).
Griese, E. U., Zanger, U. M., Brudermanns, U., Gaedigk, A., Mikus, G., Morike, K. et al. Assessment of the predictive power of genotypes for the in-vivo catalytic function of CYP2D6 in a German population. Pharmacogenetics 8, 15–26 (1998).
Broly, F., Gaedigk, A., Heim, M., Eichelbaum, M., Morike, K. & Meyer, U. A. Debrisoquine/sparteine hydroxylation genotype and phenotype: analysis of common mutations and alleles of CYP2D6 in a European population. DNA Cell Biol. 10, 545–558 (1991).
Sachse, C., Brockmoller, J., Hildebrand, M., Muller, K. & Roots, I. Correctness of prediction of the CYP2D6 phenotype confirmed by genotyping 47 intermediate and poor metabolizers of debrisoquine. Pharmacogenetics 8, 181–185 (1998).
Nakamura, K., Goto, F., Ray, W. A., McAllister, C. B., Jacqz, E., Wilkinson, G. R. et al. Interethnic differences in genetic polymorphism of debrisoquin and mephenytoin hydroxylation between Japanese and Caucasian populations. Clin. Pharmacol. Ther. 38, 402–408 (1985).
Yokota, H., Tamura, S., Furuya, H., Kimura, S., Watanabe, M., Kanazawa, I. et al. Evidence for a new variant CYP2D6 allele CYP2D6J in a Japanese population associated with lower in vivo rates of sparteine metabolism. Pharmacogenetics 3, 256–263 (1993).
Tateishi, T., Chida, M., Ariyoshi, N., Mizorogi, Y., Kamataki, T. & Kobayashi, S. Analysis of the CYP2D6 gene in relation to dextromethorphan O-demethylation capacity in a Japanese population. Clin. Pharmacol. Ther. 65, 570–575 (1999).
Wang, S. L., Huang, J. D., Lai, M. D., Liu, B. H. & Lai, M. L. Molecular basis of genetic variation in debrisoquin hydroxylation in Chinese subjects: polymorphism in RFLP and DNA sequence of CYP2D6. Clin. Pharmacol. Ther. 53, 410–418 (1993).
Yokoi, T., Kosaka, Y., Chida, M., Chiba, K., Nakamura, H., Ishizaki, T. et al. A new CYP2D6 allele with a nine base insertion in exon 9 in a Japanese population associated with poor metabolizer phenotype. Pharmacogenetics 6, 395–401 (1996).
Chida, M., Yokoi, T., Nemoto, N., Inaba, M., Kinoshita, M. & Kamataki, T. A new variant CYP2D6 allele (CYP2D6*21) with a single base insertion in exon 5 in a Japanese population associated with a poor metabolizer phenotype. Pharmacogenetics 9, 287–293 (1999).
Yamazaki, H., Kiyotani, K., Tsubuko, S., Matsunaga, M., Fujieda, M., Saito, T. et al. Two novel haplotype of CYP2D6 gene in a Japanese population. Drug Metab. Pharmacokinet. 18, 269–271 (2003).
Lim, H. S., Lee, J. H., Lee, S. K., Lee, S. E., Jang, I. J. & Ro, J. Clinical implications of CYP2D6 genotypes predictive of tamoxifen pharmacokinetics in metastatic breast cancer. J. Clin. Oncol. 25, 3837–3845 (2007).
Kiyotani, K., Mushiroda, T., Imamura, C. K., Hosono, N., Tsunoda, T., Kubo, M. et al. Significant effect of polymorphisms in CYP2D6 and ABCC2 on clinical outcomes of adjuvant tamoxifen therapy for breast cancer patients. J. Clin. Oncol. 28, 1287–1293 (2010).
Lim, J. S., Chen, X. A., Singh, O., Yap, Y. S., Ng, R. C., Wong, N. S. et al. Impact of CYP2D6, CYP3A5, CYP2C9 and CYP2C19 polymorphisms on tamoxifen pharmacokinetics in Asian breast cancer patients. Br. J. Clin. Pharmacol. 71, 737–750 (2011).
Goetz, M. P., Rae, J. M., Suman, V. J., Safgren, S. L., Ames, M. M., Visscher, D. W. et al. Pharmacogenetics of tamoxifen biotransformation is associated with clinical outcomes of efficacy and hot flashes. J. Clin. Oncol. 23, 9312–9318 (2005).
Goetz, M. P., Knox, S. K., Suman, V. J., Rae, J. M., Safgren, S. L., Ames, M. M. et al. The impact of cytochrome P450 2D6 metabolism in women receiving adjuvant tamoxifen. Breast Cancer Res. Treat. 101, 113–121 (2007).
Schroth, W., Goetz, M. P., Hamann, U., Fasching, P. A., Schmidt, M., Winter, S. et al. Association between CYP2D6 polymorphisms and outcomes among women with early stage breast cancer treated with tamoxifen. JAMA 302, 1429–1436 (2009).
Schroth, W., Antoniadou, L., Fritz, P., Schwab, M., Muerdter, T., Zanger, U. M. et al. Breast cancer treatment outcome with adjuvant tamoxifen relative to patient CYP2D6 and CYP2C19 genotypes. J. Clin. Oncol. 25, 5187–5193 (2007).
Newman, W. G., Hadfield, K. D., Latif, A., Roberts, S. A., Shenton, A., McHague, C. et al. Impaired tamoxifen metabolism reduces survival in familial breast cancer patients. Clin. Cancer Res. 14, 5913–5918 (2008).
Bijl, M. J., van Schaik, R. H., Lammers, L. A., Hofman, A., Vulto, A. G., van Gelder, T. et al. The CYP2D6*4 polymorphism affects breast cancer survival in tamoxifen users. Breast Cancer Res. Treat. 118, 125–130 (2009).
Ramón, Y., Altés, A., Paré, L., del Rio, E., Alonso, C., Barnadas, A. et al. Impact of CYP2D6 polymorphisms in tamoxifen adjuvant breast cancer treatment. Breast Cancer Res. Treat. 119, 33–38 (2010).
Damodaran, S. E., Pradhan, S. C., Umamaheswaran, G., Kadambari, D., Reddy, K. S. & Adithan, C. Genetic polymorphisms of CYP2D6 increase the risk for recurrence of breast cancer in patients receiving tamoxifen as an adjuvant therapy. Cancer Chemother. Pharmacol. 70, 75–81 (2012).
Goetz, M. P., Suman, V. J., Hoskin, T. L., Gnant, M., Filipits, M., Safgren, S. L. et al. CYP2D6 metabolism and patient outcome in the Austrian Breast and Colorectal Cancer Study Group trial (ABCSG) 8. Clin. Cancer Res. 19, 500–507 (2013).
Kiyotani, K., Mushiroda, T., Sasa, M., Bando, Y., Sumitomo, I., Hosono, N. et al. Impact of CYP2D6*10 on recurrence-free survival in breast cancer patients receiving adjuvant tamoxifen therapy. Cancer Sci. 99, 995–999 (2008).
Xu, Y., Sun, Y., Yao, L., Shi, L., Wu, Y., Ouyang, T. et al. Association between CYP2D6*10 genotype and survival of breast cancer patients receiving tamoxifen treatment. Ann. Oncol. 19, 1423–1429 (2008).
Park, H. S., Choi, J. Y., Lee, M. J., Park, S., Yeo, C. W., Lee, S. S. et al. Association between genetic polymorphisms of CYP2D6 and outcomes in breast cancer patients with tamoxifen treatment. J. Korean Med. Sci. 26, 1007–1013 (2011).
Teh, L. K., Mohamed, N. I., Salleh, M. Z., Rohaizak, M., Shahrun, N. S., Saladina, J. J. et al. The risk of recurrence in breast cancer patients treated with tamoxifen: polymorphisms of CYP2D6 and ABCB1. AAPS J. 14, 52–59 (2012).
Sukasem, C., Sirachainan, E., Chamnanphon, M., Pechatanan, K., Sirisinha, T., Ativitavas, T. et al. Impact of CYP2D6 polymorphisms on tamoxifen responses of women with breast cancer: a microarray-based study in Thailand. Asian Pac. J. Cancer Prev. 13, 4549–4553 (2012).
Nowell, S. A., Ahn, J., Rae, J. M., Scheys, J. O., Trovato, A., Sweeney, C. et al. Association of genetic variation in tamoxifen-metabolizing enzymes with overall survival and recurrence of disease in breast cancer patients. Breast Cancer Res. Treat. 91, 249–258 (2005).
Wegman, P., Vainikka, L., Stal, O., Nordenskjold, B., Skoog, L., Rutqvist, L. E. et al. Genotype of metabolic enzymes and the benefit of tamoxifen in postmenopausal breast cancer patients. Breast Cancer Res. 7, R284–R290 (2005).
Wegman, P., Elingarami, S., Carstensen, J., Stal, O., Nordenskjold, B. & Wingren, S. Genetic variants of CYP3A5, CYP2D6, SULT1A1, UGT2B15 and tamoxifen response in postmenopausal patients with breast cancer. Breast Cancer Res. 9, R7 (2007).
Okishiro, M., Taguchi, T., Jin Kim, S., Shimazu, K., Tamaki, Y. & Noguchi, S. Genetic polymorphisms of CYP2D6*10 and CYP2C19*2, *3 are not associated with prognosis, endometrial thickness, or bone mineral density in Japanese breast cancer patients treated with adjuvant tamoxifen. Cancer 115, 952–961 (2009).
Abraham, J. E., Maranian, M. J., Driver, K. E., Platte, R., Kalmyrzaev, B., Baynes, C. et al. CYP2D6 gene variants: association with breast cancer specific survival in a cohort of breast cancer patients from the United Kingdom treated with adjuvant tamoxifen. Breast Cancer Res. 12, R64 (2010).
Park, I. H., Ro, J., Park, S., Lim, H. S., Lee, K. S., Kang, H. S. et al. Lack of any association between functionally significant CYP2D6 polymorphisms and clinical outcomes in early breast cancer patients receiving adjuvant tamoxifen treatment. Breast Cancer Res. Treat. 131, 455–461 (2012).
Rae, J. M., Drury, S., Hayes, D. F., Stearns, V., Thibert, J. N., Haynes, B. P. et al. CYP2D6 and UGT2B7 genotype and risk of recurrence in tamoxifen-treated breast cancer patients. J. Natl Cancer Inst. 104, 452–460 (2012).
Regan, M. M., Leyland-Jones, B., Bouzyk, M., Pagani, O., Tang, W., Kammler, R. et al. CYP2D6 genotype and tamoxifen response in postmenopausal women with endocrine-responsive breast cancer: the breast international group 1-98 trial. J. Natl Cancer Inst. 104, 441–451 (2012).
Pharoah, P. D., Abraham, J. & Caldas, C. Re: CYP2D6 genotype and tamoxifen response in postmenopausal women with endocrine-responsive breast cancer: the Breast International Group 1-98 trial and Re: CYP2D6 and UGT2B7 genotype and risk of recurrence in tamoxifen-treated breast cancer patients. J. Natl Cancer Inst. 104, 1263–1264 (2012).
Nakamura, Y., Ratain, M. J., Cox, N. J., McLeod, H. L., Kroetz, D. L. & Flockhart, D. A. Re: CYP2D6 genotype and tamoxifen response in postmenopausal women with endocrine-responsive breast cancer: the Breast International Group 1-98 trial. J. Natl Cancer Inst. 104, 1264 (2012).
Stanton, V. Re: CYP2D6 genotype and tamoxifen response in postmenopausal women with endocrine-responsive breast cancer: the Breast International Group 1-98 trial. J. Natl Cancer Inst 104, 1265–1266 (2012).
Brauch, H., Schroth, W., Goetz, M. P., Mürdter, T. E., Winter, S., Ingle, J. N. et al. Tamoxifen use in postmenopausal breast cancer: CYP2D6 matters. J. Clin. Oncol. 31, 176–180 (2013).
Schroth, W., Hamann, U., Fasching, P. A., Dauser, S., Winter, S., Eichelbaum, M. et al. CYP2D6 polymorphisms as predictors of outcome in breast cancer patients treated with tamoxifen: expanded polymorphism coverage improves risk stratification. Clin. Cancer Res. 16, 4468–4477 (2010).
Hirano, A., Emi, M., Tsuneizumi, M., Utada, Y., Yoshimoto, M., Kasumi, F. et al. Allelic losses of loci at 3p25.1, 8p22, 13q12, 17p13.3, and 22q13 correlate with postoperative recurrence in breast cancer. Clin. Cancer Res. 7, 876–882 (2001).
Kiyotani, K., Mushiroda, T., Hosono, N., Tsunoda, T., Kubo, M., Aki, F. et al. Lessons for pharmacogenomics studies: association study between CYP2D6 genotype and tamoxifen response. Pharmacogenet. Genomics 20, 565–568 (2010).
Kiyotani, K., Mushiroda, T., Nakamura, Y. & Zembutsu, H. Pharmacogenomics of tamoxifen: roles of drug metabolizing enzymes and transporters. Drug Metab. Pharmacokinet. 27, 122–131 (2012).
Madlensky, L., Natarajan, L., Tchu, S., Pu, M., Mortimer, J., Flatt, S. W. et al. Tamoxifen metabolite concentrations, CYP2D6 genotype, and breast cancer outcomes. Clin. Pharmacol. Ther. 89, 718–725 (2011).
Barginear, M. F., Jaremko, M., Peter, I., Yu, C., Kasai, Y., Kemeny, M. et al. Increasing tamoxifen dose in breast cancer patients based on CYP2D6 genotypes and endoxifen levels: effect on active metabolite isomers and the antiestrogenic activity score. Clin. Pharmacol. Ther. 90, 605–611 (2011).
Irvin, W. J. Jr., Walko, C. M., Weck, K. E., Ibrahim, J. G., Chiu, W. K., Dees, E. C. et al. Genotype-guided tamoxifen dosing increases active metabolite exposure in women with reduced CYP2D6 metabolism: a multicenter study. J. Clin. Oncol. 29, 3232–3239 (2011).
Kiyotani, K., Mushiroda, T., Imamura, C. K., Tanigawara, Y., Hosono, N., Kubo, M. et al. Dose-adjustment study of tamoxifen based on CYP2D6 genotypes in Japanese breast cancer patients. Breast Cancer Res. Treat. 131, 137–145 (2012).
Tucker, A. N., Tkaczuk, K. A., Lewis, L. M., Tomic, D., Lim, C. K. & Flaws, J. A. Polymorphisms in cytochrome P4503A5 (CYP3A5) may be associated with race and tumor characteristics, but not metabolism and side effects of tamoxifen in breast cancer patients. Cancer Lett. 217, 61–72 (2005).
Gjerde, J., Geisler, J., Lundgren, S., Ekse, D., Varhaug, J. E., Mellgren, G. et al. Associations between tamoxifen, estrogens, and FSH serum levels during steady state tamoxifen treatment of postmenopausal women with breast cancer. BMC Cancer 10, 313 (2010).
Ruiter, R., Bijl, M. J., van Schaik, R. H., Berns, E. M., Hofman, A., Coebergh, J. W. et al. CYP2C19*2 polymorphism is associated with increased survival in breast cancer patients using tamoxifen. Pharmacogenomics 11, 1367–1375 (2010).
Moyer, A. M., Suman, V. J., Weinshilboum, R. M., Avula, R., Black, J. L., Safgren, S. L. et al. SULT1A1, CYP2C19 and disease-free survival in early breast cancer patients receiving tamoxifen. Pharmacogenomics 12, 1535–1543 (2011).
Hosono, N., Kato, M., Kiyotani, K., Mushiroda, T., Takata, S., Sato, H. et al. CYP2D6 genotyping for functional-gene dosage analysis by allele copy number detection. Clin. Chem. 55, 1546–1554 (2009).
Hosono, N., Kubo, M., Tsuchiya, Y., Sato, H., Kitamoto, T., Saito, S. et al. Multiplex PCR-based real-time invader assay (mPCR-RETINA): a novel SNP-based method for detecting allelic asymmetries within copy number variation regions. Hum. Mutat. 29, 182–189 (2008).
Kiyotani, K., Shimizu, M., Kumai, T., Kamataki, T., Kobayashi, S. & Yamazaki, H. Limited effects of frequent CYP2D6*36-*10 tandem duplication allele on in vivo dextromethorphan metabolism in a Japanese population. Eur. J. Clin. Pharmacol. 66, 1065–1068 (2010).
Goda, K., Bacso, Z. & Szabo, G. Multidrug resistance through the spectacle of P-glycoprotein. Curr. Cancer Drug Targets 9, 281–297 (2009).
Teft, W. A., Mansell, S. E. & Kim, R. B. Endoxifen, the active metabolite of tamoxifen, is a substrate of the efflux transporter P-glycoprotein (multidrug resistance 1). Drug Metab. Dispos. 39, 558–562 (2011).
Iusuf, D., Teunissen, S. F., Wagenaar, E., Rosing, H., Beijnen, J. H. & Schinkel, A. H. P-glycoprotein (ABCB1) transports the primary active tamoxifen metabolites endoxifen and 4-hydroxytamoxifen and restricts their brain penetration. J. Pharmacol. Exp. Ther. 337, 710–717 (2011).
Kiyotani, K., Mushiroda, T., Nakamura, Y. & Zembutsu, H. ABCC2 and clinical outcome of tamoxifen therapy: reply to T. Lang et al. J. Clin. Oncol. 28, e449 (2010).
Choi, H. K., Yang, J. W., Roh, S. H., Han, C. Y. & Kang, K. W. Induction of multidrug resistance associated protein 2 in tamoxifen-resistant breast cancer cells. Endocr. Relat. Cancer 14, 293–303 (2007).
Kiyotani, K., Mushiroda, T., Tsunoda, T., Morizono, T., Hosono, N., Kubo, M. et al. A genome-wide association study identifies locus at 10q22 associated with clinical outcomes of adjuvant tamoxifen therapy for breast cancer patients in Japanese. Hum. Mol. Genet. 21, 1665–1672 (2012).
Musgrove, E. A. & Sutherland, R. L. Biological determinants of endocrine resistance in breast cancer. Nat. Rev. Cancer 9, 631–643 (2009).
Giuliano, M., Schifp, R., Osborne, C. K. & Trivedi, M. V. Biological mechanisms and clinical implications of endocrine resistance in breast cancer. Breast 20 (Suppl 3), S42–S49 (2011).
Sørlie, T., Perou, C. M., Tibshirani, R., Aas, T., Geisler, S., Johnsen, H. et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl Acad. Sci. USA 98, 10869–10874 (2001).
Sørlie, T., Tibshirani, R., Parker, J., Hastie, T., Marron, J. S., Nobel, A. et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc. Natl Acad. Sci. USA 100, 8418–8423 (2003).
Paik, S., Shak, S., Tang, G., Kim, C., Baker, J., Cronin, M. et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N. Engl. J. Med. 351, 2817–2826 (2004).
Ma, X. J., Wang, Z., Ryan, P. D., Isakoff, S. J., Barmettler, A., Fuller, A. et al. A two-gene expression ratio predicts clinical outcome in breast cancer patients treated with tamoxifen. Cancer Cell 5, 607–616 (2004).
Jansen, M. P., Sieuwerts, A. M., Look, M. P., Ritstier, K., Meijer-van Gelder, M. E., van Staveren, I. L. et al. HOXB13-to-IL17BR expression ratio is related with tumor aggressiveness and response to tamoxifen of recurrent breast cancer: a retrospective study. J. Clin. Oncol. 25, 662–668 (2007).
Goetz, M. P., Suman, V. J., Couch, F. J., Ames, M. M., Rae, J. M., Erlander, M. G. et al. Cytochrome P450 2D6 and homeobox 13/interleukin-17B receptor: combining inherited and tumor gene markers for prediction of tamoxifen resistance. Clin. Cancer Res. 14, 5864–5868 (2008).
Ellis, M. J., Ding, L., Shen, D., Luo, J., Suman, V. J., Wallis, J. W. et al. Whole-genome analysis informs breast cancer response to aromatase inhibition. Nature 486, 353–360 (2012).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kiyotani, K., Mushiroda, T., Zembutsu, H. et al. Important and critical scientific aspects in pharmacogenomics analysis: lessons from controversial results of tamoxifen and CYP2D6 studies. J Hum Genet 58, 327–333 (2013). https://doi.org/10.1038/jhg.2013.39
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhg.2013.39
Keywords
This article is cited by
-
A pooled analysis of CYP2D6 genotype in breast cancer prevention trials of low-dose tamoxifen
Breast Cancer Research and Treatment (2016)
-
Pharmacogenomic diversity of tamoxifen metabolites and estrogen receptor genes in Hispanics and non-Hispanic whites with breast cancer
Breast Cancer Research and Treatment (2014)