Abstract
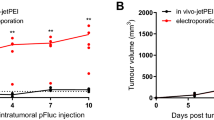
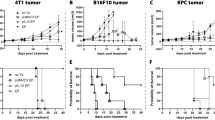
Although electroporation has been shown in recent years to be a powerful method for delivering genes to muscle, no gene therapy via electro-injection has been studied for the treatment of tumors. In an immunocompetent tumor-bearing murine model, we have found that delivery of a low dose of reporter gene DNA (10 μg) to muscle via electroporation under specific pulse conditions (two 25-ms pulses of 375 V/cm) increased the level of gene expression by two logs of magnitude. Moreover, administration of 10 μg of interferon (IFN)-α DNA plasmid using these parameters once a week for 3 weeks increased the survival time and reduced squamous cell carcinoma (SCC) growth at a distant site in the C3H/HeJ-immunocompetent mouse. IFN-α gene therapy delivered to muscle using electroporation demonstrated statistically significant (P < 0.05) therapeutic efficacy for treating SCC located at a distant site, compared with interleukin (IL)-2 or endostatin gene, also delivered by electro-injection. The increased therapeutic efficacy was associated with a high level and extended duration of IFN-α expression in muscle and serum. We also discovered that the high level of IFN-α expression correlated with increased expression levels of the antiangiogenic genes IP-10 and Mig in local tumor tissue, which may have led to the reduction of blood vessels observed at the local tumor site. Delivery of increasing doses (10–100 μg) of IFN-α plasmid DNA by injection alone did not increase antitumor activity, whereas electroporation delivery of increasing doses (10–40 μg) of IFN-α plasmid DNA did increase the survival time. Our data clearly demonstrate the potential utility of electroporation for delivery of gene therapy to muscle for the treatment of residual or disseminated tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Savaawy HME et al. Enhancement of 5-fluorouracil cytotoxicity on human colon cancer cells by retrovirus-mediated interferon-alpha gene transfer Int J Oncol 1999 14: 1143–1151
Pfeffer LM et al. Biological properties of recombinant alpha-interferons: 40th anniversary of the discovery of interferons Cancer Res 1998 58: 2489–2499
Buechner SA . Intralesional interferon alfa-2b in the treatment of basal cell carcinoma: immunohistochemical study on cellular immune reaction leading to tumor regresion J Am Acad Dermatol 1999 24: 731–734
Gutterman JU . Cytokine therapeutics: lessons from interferon-α Proc Natl Acad Sci USA 1994 91: 1198–1205
Ogura H et al. Implantation of genetically manipulated fibroblasts into mice as antitumor α-interferon therapy Cancer Res 1990 50: 5102–5106
Belldegrun A et al. Human renal carcinoma line transfected with interleukin and/or interferon-α gene(s): implication for livecancer vaccines J Natl Cancer Inst 1993 85: 207–216
Ferrantini M et al. Interferon alpha-1 gene transfer into metastatic Friend leukemia cells abrogated tumorigenicity in immunocompetent mice: antitumor therapy by means of interferon-producing cells Cancer Res 1993 53: 4607–4615
Zhang JF et al. Gene therapy with an adeno-associated virus carrying an interferon gene results in tumor growth suppression and egression Cancer Gene Ther 1996 3: 31–38
Hiroishi K, Tuting T, Lotze MT . IFN-alpha-expression tumor cells enhance generation and promoter survival of tumor-specific CTLs J Immunol 2000 164: 567–572
Coleman M et al. Nonviral interferon-alpha gene therapy inhibits growth of established tumors by eliciting a systemic immune response Hum Gene Ther 1998 9: 2223–2230
Hottiger MO et al. Liposome-mediated gene transfer into human basal cell carcinoma Gene Therapy 1999 6: 1929–1935
Ahmed CM et al. In vivo tumor suppression by adenovirus-mediated interferon alpha-2b gene delivery Hum Gene Ther 1999 10: 77–84
Wolff JA et al. Direct gene transfer into mouse muscle in vivo Science 1990 247: 1465–1468
Horton HM et al. A gene therapy for cancer using intramuscular injection of plasmid DNA encoding interferon-α Proc Natl Acad Sci USA 1999 96: 1553–1558
Mathiesen I . Electropermeabilization of skeletal muscle enhances gene transfer in vivo Gene Therapy 1999 6: 508–514
Aihara H, Miyazaki J . Gene transfer into muscle by electroporation in vivo Nat Biotechnol 1998 16: 867–870
Rizzuto G et al. Efficient and regulated erythropoietin production by naked DNA injection and muscle electroporation Proc Natl Acad Sci USA 1999 96: 6417–6422
Belardelli F, Gresser E, Maury C, Maunoury MT . Antitumor effects of interferon in mice injected with interferon-sensitive and interferon-resistant Friend leukemia cells. II. Role of host mechanisms Int J Cancer 1982 30: 821–825
Gresser I . Antitumor effects of interferon Acta Oncol 1989 28: 347–353
Singh RK et al. Interferons α and β downregulate the expression of basic fibroblast growth factor in human carcinomas Proc Natl Acad Sci USA 1995 92: 4562–4566
Tuting T et al. Interferon-α gene therapy for cancer: retroviral transduction of fibroblasts and particle-mediated transfection of tumor cells are both effective strategies for gene delivery in murine tumor models Gene Therapy 1997 4: 1053–1060
Slaton JW et al. Interferon-alpha-mediated down-regulation of angiogenesis-related genes and therapy of bladder cancer are dependent on optimization of biological dose and schedule Clin Cancer Res 1999 5: 2726–2734
Tannenbaum C et al. The CXC chemokines IP-10 and Mig are necessary for IL-12 mediated regression of the mouse RENCA tumor J Immunol 1998 161: 927–932
Sgadari C et al. Interferon-inducible protein-10 identified as a mediator of tumor necrosis in vivo Proc Natl Acad Sci USA 1996 93: 13791–13796
Angiolillo A et al. Human interferon-inducible protein 10 is a potent inhibitor of angiogenesis J Exp Med 1995 182: 155–162
Coughlin C et al. Tumor cell responses to IFN-gamma affect tumorigenicity and response to IL-12 therapy and antiangiogenesis Immunity 1998 9: 25–34
Asselin-Paturel C et al. Transfer of the murine interleukin-12 gene in vivo by a Semliki Forest virus vector induces B16 tumor regression through inhibition of tumor blood vessel formation monitored by Doppler ultrasonography Gene Therapy 1999 6: 606–615
Horton MR et al. Hyaluronan fragments synergize with interferon-gamma to induce the C-X-C chemokines mig and interferon-inducible protein-10 in mouse macrophages J Biol Chem 1998 273: 35088–35094
MacLaughlin FC et al. Plasmid gene delivery: advantages and limitations. In: Gregoriadis G, McCormack B (eds). Targeting of Drugs: Strategies for Gene Constructs and Delivery IOS 2000
Li S et al. Increased level and duration of expression in muscle by co-expression of a transactivator using plasmid systems Gene Therapy 1999 6: 2055–2011
Barnhart KM et al. Enhancer and promoter chimeras in plasmids designed for intramuscular injection: a comparative in vivo and in vitro study Hum Gene Ther 1998 9: 2545–2553
Mumper RJ et al. Protective interactive noncondensing (PINC) polymers for enhanced plasmid distribution and expression in rat skeletal muscle J Control Rel 1998 52: 10876–10880
Heller R et al. In vivo gene electroinjection and expression in rat liver FEBS Lett 1996 389: 225–228
Nishi T et al. High-efficiency in vivo gene transfer using intrarterial plasmid DNA injection following in vivo electroporation Cancer Res 1996 56: 1050–1055
Mir LM et al. High-efficiency gene transfer into skeletal muscle mediated by electric pulses Proc Natl Acad Sci USA 1999 96: 4262–4267
Blezinger P et al. Systemic inhibition of tumor growth and tumor metastases by intramuscular administration of the endostatin gene Nat Biotechnol 1999 17: 343–348
Blezinger P et al. Intratracheal administration of interleukin 12 plasmid–cationic lipid complexes inhibits murine lung metastases Hum Gene Ther 1999 10: 723–731
Grandis J, Chang MJ, Yu WD, Johnson C . Antitumor activity of interleukin-1-alpha and cisplatin in a murine model system Arch Otolaryngol Head Neck Surg 1999 121: 197–200
O'Malley B, Cope K, Johnson C, Schwartz M . A new immunocompeternt murine model for oral cancer Arch Otolaryngol Head Neck Surg 1997 123: 20–24
Snyderman C, Abbas M, Wagner R, D'Amico F . Inhibition of growth of a murine squamous cell carcinoma by a cyclooxygenase inhibitior increases leukotriene B4 production Arch Otolaryngol Head Neck Surg 1995 121: 1017–1020
Li S, Wilkinson MF . A translation-like and nuclear-associated mechanism dictate the nonsense codon-mediated TCR mature transcript decay J Exp Med 1997 185: 985–992
Acknowledgements
We would like to thank Valentis, Inc., and Gentronics, Inc., for allowing us to use their gene constructs and electroporator, respectively, for this study. This work was partially supported by the Sam Walton Fund and a University of Arkansas for Medical Sciences pilot grant.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Li, S., Zhang, X., Xia, X. et al. Intramuscular electroporation delivery of IFN-α gene therapy for inhibition of tumor growth located at a distant site. Gene Ther 8, 400–407 (2001). https://doi.org/10.1038/sj.gt.3301418
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301418
Keywords
This article is cited by
-
CD8+T cell–specific induction of NKG2D receptor by doxorubicin plus interleukin-12 and its contribution to CD8+T cell accumulation in tumors
Molecular Cancer (2014)
-
Plasmid injection and application of electric pulses alter endogenous mRNA and protein expression in B16.F10 mouse melanomas
Cancer Gene Therapy (2010)
-
Administering Plasmid DNA Encoding Tumor Vessel–anchored IFN-α for Localizing Gene Product Within or Into Tumors
Molecular Therapy (2008)