Abstract
Purpose
The main objective of this study is to describe the prevalence, degree and risk of corneal involvement, and visual impact in a pediatric population with blepharokeratoconjunctivitis (BKC).
Methods
Retrospective, observational, case–control study. Clinical records of patients ≤16 years old with BKC seen between 2006 and 2012 were reviewed. The prevalence and relative risk of corneal involvement was evaluated between patients with and without corneal affection through a univariate and multivariate analysis with logistic regression. Visual acuity at presentation and at last follow-up visit was also analyzed.
Results
One hundred and fourteen children with BKC, with a male-to-female ratio of 1 : 1 and a mean age at diagnosis of 9.13 years. The mean follow-up time was 26.4 (±25) months. Corneal involvement was present in 39.5% of patients, varying from superficial punctate keratitis to perforation. Corneal changes were not seen in children under 4 years old. The risk of corneal affection was greater in patients with photophobia, hordeolum, female gender and asymmetric disease (OR of 2.69, 11.6, 2.35 and 2.77, respectively). The mean best-corrected visual acuity at presentation was 0.20 (corneal affected group), compared to 0.11 (unaffected group; P=0.02).
Conclusions
Our study showed an older age at time of diagnosis and a worse visual outcome in patients with BKC and corneal disease compared with previous reports. Early diagnosis and detection of risk factors for corneal involvement, as well as adequate treatment, is mandatory to prevent serious long-term visual repercussions in children with BKC.
Similar content being viewed by others
Introduction
Blepharokeratoconjunctivitis (BKC) is a chronic inflammatory disease of the palpebral margin with secondary conjunctival and corneal involvement that affects the pediatric population. Disease presentation shows a wide clinical spectrum.1 Ambiguous diagnostic criteria and disease definition are reflected in the terminology used in the literature to describe this pathology. Names such as staphylococcal blepharitis, non-tuberculous, or staphylococcal phyctenular disease, childhood rosacea, and blepharokeratitis have all being used to describe this disorder.2
The diagnostic criteria described for chronic BKC include the presence of tearing, photophobia, red eye, blepharitis (external hordeola or meibomian cysts), history of recurrent chalazia, phlyctenular conjunctivitis, keratitis, as well as corneal complications such as ulceration, neovascularization, scarring, and perforation.1, 2, 3, 4, 5 Although the disease is similar in children and adults, the visual outcome is generally worse in children.6 BKC incidence in children has been reported to be 15%,1 with the condition being most common in Asians and more severe in children from the Middle East.4
The pathogenesis of this disorder is unclear, but it has been proposed to be associated with early skin and mucous membrane bacterial colonization, including oral and nasal mucosa, the conjunctiva, and the eyelids with Staphylococcus aureus and coagulase negative.1 The immune-genetic susceptibility of the affected patients; the development of hypersensitivity responses, primarily a type IV (delayed cell mediated) against antigens from the cellular wall (protein-A and teichoic acids, such as ribitol);6, 7 as wells as the toxic and direct action of staphylococcal exotoxins (alpha, beta, and gamma-hemolysins) on the ocular surface, have all been implicated in the development of the disease.6, 7, 8, 9 It is believed that children are more susceptible to corneal damage from this exaggerated and immature immune adaptive response against the bacterial components previously described.10
The management of BKC is challenging, and is currently limited to disease control but not to a definitive cure. At present time, there is not an entirely effective and standardized method of treatment for these patients, and complications such as amblyopia, corneal scarring, thinning, and perforation may occur, along with the development of secondary glaucoma and cataract due to the excessive and chronic use of topical corticosteroids to control inflammation.11
The aim of this study is to report the prevalence, and to analyze the potential risk factors for corneal involvement and visual outcome in Mexican children with BKC.
Patients and methods
A retrospective, observational, case–control study was performed by reviewing the medical records of patients younger than 16 years of age with a clinical diagnosis of chronic BKC who were consecutively seen at the Cornea and Ocular Surface Clinic of our institution from 2006 to 2012. Collection of clinical data was approved by the Ethics and Research Committees of our institution, and conducted according to the tenets of the Declaration of Helsinki.
Inclusion criteria for disease diagnosis included tearing, photophobia, red eye, anterior, and/or posterior blepharitis, recurrent chalazia, phlyctenular conjunctivitis, keratitis, and corneal manifestations like ulceration, neovascularization, scarring, and perforation.1, 2, 4, 5 Demographic characteristics and age at the time of diagnosis were registered. The presence of the following signs and symptoms were recorded: red eye, pruritus at the eyelid margins, photophobia, conjunctival discharge, tearing, frequent blinking, foreign body sensation, and ocular pain. Palpebral signs included: secretion (collarettes, crusts and scales, sleeves); eyelid border ulceration; palpebral erythema; chalazia; meibomian gland dysfunction; hordeolum; madarosis; and pigmentary changes. Signs of conjunctival inflammation such as bulbar hyperemia, papillary reaction, follicular reaction, and the formation of phlyctenules were evaluated. The cornea was examined with fluorescein staining for epithelial defects (superficial punctate keratitis, epithelial erosions, margin infiltrates, and ulcers). In addition, corneal vascularization (partial or total), stromal thinning, presence of leukomas, stromal active inflammatory infiltration, ulceration, and perforation were also analyzed. Bilateral or unilateral eye involvement was recorded. Patients with bilateral disease were further classified as symmetrical, if the signs and symptoms were similar in both eyes; and asymmetrical, when only mild blepharitis and conjunctival hyperemia were present in one eye, whereas the contralateral eye showed marked signs of eyelids, conjunctival, and corneal inflammation.
Best-corrected Snellen visual acuity (BCVA) was recorded (converted to logMAR for statistical purposes) for every patient at presentation and at last follow-up available. Patients with a BCVA≤20/50 were classified as ‘poor vision’ and BCVA≤20/200 as ‘legal blindness’, based on the recommendations of the Standardization of Uveitis Nomenclature Working Group.12 Amblyopia, a change in 0.2 logMAR units of BCVA between eyes, was recorded in all patients with and without corneal involvement and classified according to BCVA measurement as mild (20/30 to 20/40), moderate (20/40 to 20/125), or severe (>20/125).13
The total studied population was divided into two groups: those who showed no corneal manifestations (unaffected group), and those with corneal involvement (affected group), in order to compare signs, symptoms, and visual acuities from each group. Patients within the corneal affected group were further subdivided into those with superficial punctate keratitis (SPK) alone, and those with more severe corneal pathology such as, corneal neovascularization (sectorial or total), stromal opacities, corneal thinning, marginal infiltrates, corneal phlyctenules, ulcers, peripheral corneal ulceration, and perforation.
Once enrolled in our service, all children were treated in a similar manner based on disease severity. Regardless of the degree of ocular surface inflammation, all patients were asked to perform lid hygiene with commercially available eyelid cleaning pads for an indefinite period of time. Preferable, unpreserved topical lubricants (hyaluronate or methylcellulose-based) were administered 3 to 4 times a day to each eye for an indefinite period of time to all patients.
Whenever meibomian gland dysfunction, history of chalazia, active chalazion, or hordeolum were present warm compresses were applied to the eyelids for 10 min prior to lid hygiene. Also, dietary supplementation with omega-3 free fatty acids (flaxseed oil) was administered to all these children (2 g/day) for a minimum of 6 weeks, reducing to alternate days for at least 6 months. Erythromycin stearate 5.0 mg or ciprofloxacin-HCl 3.3 mg ointment was applied on the eyelid margins every night for at least 6 weeks to patients with collarette and/or pustular folliculitis. Patients with active inflammation and corneal involvement (moderate to severe disease) also received topical steroids (unpreserved 0.1% dexamethasone phosphate, 0.5% loteprednol etabonate, or 0.1% fluorometholone phosphate) at a frequency range of every hour to every other day for the shortest period of time possible (usually 4–6 weeks total administration time per flare-up). Patients with severe corneal involvement also received twice a day topical unpreserved 0.05% cyclosporine-A emulsion for a minimum of 3 months. In addition, most patients with corneal involvement were given 30–80% of the systemic children recommended dose (50 mg/kg/day) of erythromycin estolate, either as suspension, capsules, or tablets, depending on the age, weight, and ability of the child to swallow. The oral dose was reduced as fast as possible to the lowest dose required for control of palpebral and ocular surface inflammation. In most cases, systemic erythromycin dose was given as low as 125 mg every other day for 6–8 months. Oral tetracyclines were avoided owing to young age-related side effects.14, 15
For statistical analysis, the baseline characteristics were expressed as means with SD or medians and their respective interquartile ranges, depending on whether the continuous variables displayed a normal distribution. A univariate analysis was first conducted by comparing every variable through the non-parametric Mann–Whitney U-test for variables that do not follow a normal distribution, and Student’s t-test was used in cases where variables with a normal distribution were found. The categorical data were evaluated through Fisher’s exact test in contingency tables. Statistical significance was defined as a P-value <0.05. Last, the variables that resulted statistical significant were grouped in a multivariate analysis through binary logistic regression (forward LR method) by taking a positive or negative presence of corneal involvement as a dependent variable. Through this analysis, odds ratio values were obtained with their respective confidence intervals of 95%.
Results
The clinical records of 114 consecutive patients who met the diagnostic criteria for BKC were reviewed. Of these, 55 (48.2%) were male and 59 (51.8%) females, with a male-to-female ratio of 1 : 1.07. The mean age at the time of diagnosis was 9.3 years (±4.2 years). The mean age at disease onset was 6.2 years (±2.6 years), and disease duration prior to referral or first visit to our clinic was 3.7 years (±5.2 years). The mean follow-up time was 26.4 months (±25.2 months).
The study population was divided according to the presence or absence of corneal involvement. The unaffected group (n=69 patients) was composed of 39 males (56.5%) and 30 females (43.5%), with a mean age at diagnosis of 8.5 years (±4.3 years), whereas the affected group (n=45 patients), included 29 females (64.9%), and 16 males (35.6%), with a mean age at diagnosis of 10.2 years old (±3.9 years). A statistically significant difference was obtained for female and older age at diagnosis (P=0.029 and P=0.04, respectively) between both groups. No corneal inflammatory signs were noted in children younger than 4 years of age, compared with 38.9% of children of 6–12 years old and 53.1% of patients older than 13 years.
The prevalence of corneal involvement in the entire population studied was 39.5% (95% CI=30.53–48.47). A significant difference was found between both groups, with respect to red eye (P=0.007), photophobia (P=0.01), presence of scales (P=0.01), and hordeola (P=0.02; Table 1).
Of the 45 patients with corneal pathology, the most common findings were SPK in 71.1% of patients, followed by corneal leukomas in 40%, and corneal vascularization in 30% (Table 2).
Among all patients with chronic BKC included in the study, the majority presented with bilateral disease (94.7%) and in 58.8% of them, the ocular manifestations were symmetrical. However, it is worth mentioning that in the group of patients with corneal involvement, the disease showed a tendency (57.8%) for asymmetry (P=0.004).
A total of nine (7.8%) patients were excluded from the visual acuity analysis (seven from the unaffected and two from the corneal affected group) because they were unable to read, and therefore visual acuity was not quantitatively estimated. The mean BCVA at presentation of the entire studied population was 0.15 (±0.16). The group without corneal involvement had an initial BCVA of 0.11 (±0.10), compared with 0.20 (±0.21) of patients with corneal disease (Figure 1), with a variance of 0.12 and 0.52, respectively. This difference of BCVA at presentation between both groups was statistically significant (P=0.02). There were a total of seven (6.14%) patients with poor vision (≤20/50) at presentation. Of these, 6/7 (85.7%) patients had corneal involvement, including eyes with a BCVA between 20/150 and finger counting at 7 feet.
A mean clinical follow-up time of 26.4 (±25) months was achieved in 64 of the 105 patients analyzed for visual acuity. There was no follow-up time for longitudinal visual analysis in 51% of patients from the unaffected group and in 25% of the corneal affected group. The final BCVA for these patients did not differ significantly (P=0.26) between unaffected group (0.11±0.10), and the corneal affected group (0.2±0.22).
For visual outcome analysis, we considered relevant to exclude those patients who had SPK as sole corneal manifestation from the corneal affected group. By doing so, we found a statistically significant difference when comparing the initial BCVA (P=0.002) and the final BCVA (P=0.04) between both groups with a marked difference in the variance (Figure 1). Mild refractive amblyopia was detected in one (0.8%) and moderate in three (2.6%) children without corneal involvement, whereas in the corneal affected group, mild refractive, or stimulus deprivation amblyopia was seen in two (1.7%) patients, moderate in four (3.5%) patients, and severe in three (2.63%) patients. When comparing initial with final BCVA in patients with corneal involvement, there was statistical significant improvement of amblyopia (P=0.017) after adequate treatment was applied.
A multivariate analysis was conducted through logistic regression to determine variables considered as possible risk factors for corneal involvement in patients with chronic BKC. Female gender, older age at diagnosis, asymmetric disease, photophobia, and the presence of hordeola had an OR>1 (Table 3).
Prior to first visit to our clinic, only 8 (7.0%) patients had been treated with lid hygiene, 77 (67.5%) with topical antibiotics, 29 (25.4%) with anti-allergic drugs, 13 (11.4%) with decongestants, and most of them (82.4%) received tear substitutes. Only 6/69 patients without corneal manifestations had been previously treated with topical steroids, whereas 26/45 patients with corneal disease received different formulations of topical steroids. A total of 52 (45.6%) patients had also been treated with systemic antibiotics of different kinds. No patient received omega-3 supplementation prior to referral.
Once admitted to our clinic, all patients were treated with lid hygiene and tear substitutes regardless of disease severity. In all patients with MGD (n=14; 12.2%), history of chalazia (n=57; 50.0%), or hordeolum (n=6; 5.2%) warm compresses were indicated before lid hygiene and massage were applied to the eyelid margins. Flax seed oil containing 1000 mg of Omega-3 was also indicated twice a day for the first 6 weeks, then every other day to these patients for a mean time of 9.4 months (range, 4–23 months) Erythromycin stearate 5.0 mg (n=54; 47.3%), or ciprofloxacin-HCL 3.3 mg (n=46; 40.3%) ointment was administered after lid hygiene to the anterior eyelid margin in patients who presented with collarette, (n=100; 87.7%), history of chalazia (n=57; 50.0%), or hordeolum (n=6; 5.2%). All patients with active inflammation and corneal involvement (moderate to severe disease) were also treated with topical steroids (n=39; 86.6%) for a mean time of 3.5 weeks (range, 2–10 weeks) for flare-ups as stated before. Of these, preservative-free 0.1% dexamethasone phosphate was administered to 9 (20.0%) patients, 0.5% loteprednol etabonate to 19 (42.2%) patients, and 0.1% flurometholone fosfate to 17 (37.7%) patients. Preservative-free 0.02% cyclosporine-A was administered only to 5 (11.1%) patients with active and severe corneal involvement for a mean time of 6.5 months (range, 3.5–14 months). Finally, systemic erythromycin estolate was administered orally to 40 (88.8%) patients with severe corneal involvement. The minimum maintenance erythromycin dose administered was 125 mg every other day, and the mean time of therapeutic episodes was 6.4 months (range, 2.5–8 months).
Discussion
BKC is a chronic inflammatory disease of the palpebral margins with a wide variety of clinical manifestations on the ocular surface and a potential for severe corneal damage, compromising the visual outcome.2, 10 We report herein a large cohort of pediatric Hispanic patients with chronic BKC in whom we analyzed the prevalence and potential risk factors for corneal involvement and poor visual outcome.
Finding an older mean age at the time of diagnosis (9.3 years) in the present study reflects a delayed diagnosis, a fact supported by the acknowledgement of signs and symptoms of the disease initiating at a mean age of 6.2 years, as reported by patients or their relatives on their first visit to our clinic. Moreover, misdiagnosis was also evidenced by previous treatments unrelated to the definitive disorder given by other physicians prior to referral. In this respect, only few patients (7%) were on lid hygiene, 67.5% were on topical antibiotics, 25.4% were treated with anti-allergic drugs, 11.4% with decongestants, and none of them were on omega-3 supplementation. A delayed diagnosis was also suggested when we compared this figure with studies performed in countries where the age at diagnosis was younger such as, the United States (6.5 years), England (5.4 to 6.9 years), and India (6.7 years).1, 2, 4, 10 In the present report, an older age of diagnosis showed a tendency for a higher risk of corneal involvement (OR=1.13, 95% CI=1.01–1.27). This tendency was reinforced by the fact that 53.5% of patients older than 13 years of age had corneal involvement, compared with only 25% of patients younger than 6 years of age. In accordance to this, Hamada et al,16 described a BKC subgroup conformed by Caucasian adolescents who presented to their clinic at a median age of 15.2 years and disease duration of 4.3 years with severe inflammation characterized by significant corneal damage. These patients experienced a delayed referral for specialized attention, and 3/10 required aggressive systemic immunosuppression in order to control inflammation and avoid serious ocular morbidity, particularly corneal perforation.16
Previous reports have found a greater female to male ratio in children with BKC, although this has not been shown to be significant.2 In this study, there was no significant difference according to gender; however, it is important to note that the female group had a tendency toward a greater risk for corneal disease (OR=2.35, 95% CI=1.08–5.1).
With respect to eye involvement, Hammersmith et al,2 reported bilateral disease in 97% of their cases with asymmetric clinical manifestations in only 21% of them. In the present study, the disease was also bilateral in most patients (94.3%), however, there was a greater prevalence (41.2%) for asymmetry. Interestingly, asymmetric presentation showed a twofold higher risk for developing corneal involvement (OR=2.77, 95% CI=1.12–6.84).
The degree of corneal involvement in BKC reported in the literature varies significantly from 5 to 100% of cases.1, 3, 6, 11, 17, 18 This great variation may be due to differences in demographic, socioeconomic, and cultural characteristics of populations studied. Intentional selection bias of patients included for analysis is also a great contributor for such a wide range of corneal manifestations in BKC, and so is the lack of well-established diagnostic criteria that clearly defines the disease.1, 4, 5, 11, 17, 18 In the present study, where all patients with BKC consecutively seen in our clinic during a 6-year period were included for analysis, and diagnostic criteria established by Gupta1 and Doan5 were applied to all patients, the prevalence of corneal involvement was 39.5%. This may represent a more accurate figure although it is also biased by the fact that our center is a tertiary care facility, and patients with a milder disease may had not attended our service.
Viswalingam et al4 found that the most common corneal findings in BKC were sub-epithelial punctate keratitis and marginal infiltrates (63.6% each); epithelial punctate keratitis, (59.1%) and sectorial neovascularization (34.1%). In the present study, the most common corneal manifestations were superficial punctate keratitis, leukomas (including peripheral and central nummular leukomas), and corneal neovascularization, similar to previous reports.5, 11
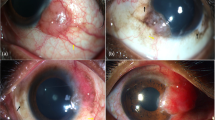
Jones et al10 found corneal involvement in at least one eye of 27 children, the mean initial BCVA was 0.28, significantly improving to 0.02 in 70% of the affected eyes after adequate treatment (P=0.012). In their study, the multivariate analysis found no association between final visual acuity and corneal involvement, gender, ethnic group, or age at onset of symptoms. However, a decrease visual acuity was found when treatment was initiated >2 years after the onset of symptoms.10 In the present report, patients with poor visual acuity showed dense central and peripheral opacities, superficial, and stromal neovascularization, stromal thinning, and even corneal perforation usually associated with a prolonged clinical course of the disease (Figure 2).
(a) Left eye of a 12-year-old female with chronic BKC showing a dense inferotemporal leukoma that partially obstructs the visual axis; (b) left eye of a 9-year-old male showing a central stromal infiltrate with extensive neovascularization on all quadrants; (c) right eye of a 7-year-old female, seen 1 year after a corneal tectonic graft was performed owing to peripheral cornel perforation.
When comparing the BCVA at presentation of patients from the unaffected group with those from the corneal affected group, a significant difference was found between both groups (P=0.02). Moreover, when comparing the initial with the last visit BCVA between both groups, a statistical significant difference (initial: P=0.002, and final: P=0.04) was seen after subtracting those patients with superficial punctate keratitis from the corneal affected group, showing that this corneal sign alone does not have an impact in visual outcome. On the other hand, a poor visual outcome was seen in those patients who presented with opaque corneas, corneal neovascularization, inflammatory infiltrates, ulceration, and perforation. These patients also experienced the largest prevalence (20.0%) of amblyopia. Moderate anisometropic amblyopia related to significant regular and also irregular astigmatism was present in four (3.5%) patients, and severe refractive or stimulus deprivation amblyopia was found in three (2.63%) patients with significant corneal damage. In addition, these patients required a more aggressive therapeutic approach with frequent topical steroids and cyclosporine-A, as well as oral erythromycin for prolonged periods of time.15 In this respect, we decided to implement a therapeutic protocol based on disease severity and level of corneal involvement.10, 11, 14, 15, 16, 18 Lid hygiene and lubricant eye drops were the standard of care for all patients.10, 14 Warm compresses prior to lid massage and omega-3 supplementation were added to patients with MGD, history of chalazia and hordeolum.14, 19, 20, 21, 22, 23 Omega-3 supplementation may have an adjunctive effect on the management of MGD and inflammatory dry eye.22, 23, 24, 25, 26, 27, 28, 29, 30, 31 It has been shown that omega-3 modulate prostaglandin metabolism toward anti-inflammatory prostaglandin (PGE3) synthesis due to competitive inhibition of the arachidonic acid pathway at the cyclo- and lipo-oxygenase level.22, 32, 33 Therefore, increasing systemic levels of omega-3 free fatty acids (EFA) result in decreased production of prostaglandin-E2 (PGE2) metabolites, thromboxane-A2, and leukotriene-B4, all of which have a proinflammatory profile.22, 32, 33, 34 Furthermore, eicosapentaenoic acid (EPA), blocks the gene expression of proinflammatory cytokines, interleukin-1, tumor necrosis factor (TNF), aggrecanases, and cyclooxygenase.22, 32, 33, 34, 35 On the other hand, EFA containing omega-3, are used by meibomian glands to synthesize meibum. Dietary intake of EPA, and docosahexaenoic acid in particular, has been shown to affect the polar lipid profiles by clearing and thinning meibomian gland secretions.22, 23, 24 Despite the evidence-based recommendations provided in the American Academy of Ophthalmology Preferred Practice Guidelines for blepharitis36 and the International Workshop on MGD subcommittee’s report on treatment for MGD,14 there is little in the literature regarding the long-term use of flaxseed oil in children, and the studies published so far have shown limited evidence to demonstrate its effectiveness in the management of blepharitis.28
Whenever collarette secretion and/or pustular folliculitis was present, patients were also recommended to apply topical antibiotics (erythromycin or ciprofloxacin ointment) to control and reduce bacterial colonization on the eyelid margins.10, 28, 36 Topical steroids have been shown to be successful in the management of the acute phase or flare-ups of MGD.3, 4, 10 Although corticosteroids provide a rapid control of inflammation, they could not be used for prolonged periods of time owing to their known ocular side effects, cataract, and glaucoma.3, 4, 5, 36 On the other hand, topical CsA may be used for longer periods of time to control inflammation (at least 3 months to reach therapeutic effect), but with the inconvenience of high cost and low-tolerance profile.37, 38 CsA was used in 5 (11.1%) of our patients with active and severe corneal involvement for a mean time of 6.5 months, with three patients showing adequate control of inflammation. The other two patients stopped the medication owing to intolerance. Systemic erythromycin has been the antibiotic of choice in most published series of BKC in children, and was given to almost all our patients (88.8%) with corneal involvement.2, 3, 5, 10, 15, 17, 18, 39 Erythromycin can modulate the production of proinflammatory cytokines and neutrophil adhesion, among other anti-inflammatory effects.10, 39 Although gastrointestinal upset is a frequent side effect reported with this antibiotic, this is a rare problem at a lower dose, as shown in the present study where a dose as low as 125 mg every other day was sustained for 6.4 months in most patients treated.10, 15, 39
In spite of all efforts made to retain most patients under therapy, the lack of follow-up time in half of patients of the unaffected group and 25% of the corneal affected group limited our capacity to determine definitive conclusions about visual outcome. Nevertheless, we found a significant difference when comparing initial and final BCVA between unaffected and corneal affected groups (Figure 1). We observed that patients with severe ocular surface disease had better compliance and longer follow-up time. These patients also require more aggressive and prolonged therapy in order to control inflammation and maintain the eyes away from further complications (Figure 2).
Conclusions
BKC is a chronic inflammatory disease of the ocular surface that frequently affects the pediatric population. The prevalence of corneal involvement varies depending on the population studied, and probably increases with delayed diagnosis and a longer clinical course without appropriate treatment. In contrast to similar studies from developed countries, we found that BKC was diagnosed at an older age (mean of 9.3 years). A past ocular history of symptoms and signs of BKC initiating at a mean age of 6.2 years, as reported by patients or their relatives on their first visit, along with previous therapeutic modalities unrelated to the definitive disease, suggested a prolonged clinical course and delay diagnosis.
The risk factors related to corneal involvement in patients with BKC included: female gender, asymmetric disease, diagnosis at an older age, and the presence of photophobia and hordeola. Visual acuity at presentation and at the end of follow-up time was also significantly decreased in patients with corneal involvement compared with those without it. Finally, longer follow-up time and better therapeutic compliance was evident in patients with severe corneal damage. These patients also require a more aggressive therapeutic approach in order to overcome ocular complications.

References
Gupta N, Dhawan A, Beri S, D’souza P . Clinical spectrum of pediatric blepharokeratoconjunctivitis. J AAPOS 2010; 14: 527–529.
Hammersmith KM, Cohen EJ, Blake TD, Laibson PR, Rapuano CJ . Blepharokeratoconjunctivitis in children. Arch Ophthalmol 2005; 123: 1667–1670.
Farpour B, McClellan KA . Diagnosis and management of chronic blepharokeratoconjunctivitis in children. J Pediatr Ophthalmol Strabismus 2001; 38: 207–212.
Viswalingam M, Rauz S, Morlet N, Dart JK . Blepharokeratoconjunctivitis in children: diagnosis and treatment. Br J Ophthalmol 2005; 89: 400–403.
Doan S, Gabison EE, Nghiem-Buffet S, Abitbol O, Gatinel D, Hoang-Xuan T . Long-term visual outcome of childhood blepharokeratoconjunctivitis. Am J Ophthalmol 2007; 143: 528–529.
Mondino BJ, Adamu SA, Pitchenkian-Halabi H . Antibody studies in a rabbit model of corneal phlyctenulosis and catarrhal infiltrates related to Staphylococcus aureus. Invest Ophthalmol Vis Sci 1991; 32: 1854–1863.
Ficker L, Seal D, Wright P . Staphylococcal infection and the limbus: study of the cell-mediated immune response. Eye 1989; 3: 190–193.
Kumar A, Zhang J, Yu FS . Innate immune response of corneal epithelial cells to Staphylococcus aureus infection: Role of peptidoglycan in stimulating proinflammatory cytokine secretion. Invest Ophthalmol Vis Sci 2004; 45: 3513–3522.
Smolin G, Okumoto M . Staphylococcal blepharitis. Arch Ophthalmol 1977; 95: 812–816.
Jones SM, Weinstein JM, Cumberland P, Klein N, Nischal KK . Visual outcome and corneal changes in children with chronic blepharokeratoconjunctivitis. Ophthalmology 2007; 114: 2271–2280.
Teo L, Mehta JS, Htoon HM, Tan DTH . Severity of pediatric blepharokeratoconjunctivitis in Asian eyes. Am J Ophthalmol 2012; 153: 564–570.
Jabs DA, Nussenblatt RB, Rosenbaum JT . Standardization of Uveitis Nomenclature (SUN) Working Group. Standardization of uveitis nomenclature for reporting clinical data. Results of the First International Workshop. Am J Ophthalmol 2005; 140: 509–516.
West S, Williams C . Amblyopia. BMJ Clinical Evidence 2011; 2011: 0709.
Geerling G, Tauber J, Baudouin C, Goto E, Matsumoto Y, O’Brien T et al. The International Workshop on Meibomian Gland Dysfunction: Report of the Subcommittee on Management and Treatment of Meibomian Gland Dysfunction. Invest Ophthalmol Vis Sci 2011; 52: 2050–2064.
Cetinkaya A, Akova YA . Pediatric ocular acne rosacea: Long-term treatment with systemic antibiotics. Am J Ophthalmol 2006; 142: 816–821.
Hamada S, Kahn I, Denniston AK, Rauz S . Childhood blepharokeratoconjunctivitis: characterizing a severe phenotype in white adolescents. Br J Ophthalmol 2012; 96: 949–955.
Nazir SA, Murphy S, Siatkowski RM, Chodosh J, Siatkowski RL . Ocular rosacea in childhood. Am J Ophthalmol 2004; 137: 138–144.
Erzurum SA, Feder RS, Greenwald MJ . Acne rosacea with keratitis in childhood. Arch Ophthalmol 1993; 111: 228–230.
Nagymihalyi A, Dikstein S, Tiffany JM . The influence of eyelid temperature on the delivery of meibomian oil. Exp Eye Res 2004; 78: 367–370.
Olson MC, Korb DR, Greiner JV . Increase in tear film lipid layer thickness following treatment with warm compresses in patients with meibomian gland dysfunction. Eye Contact Lens 2003; 29: 96–99.
Goto E, Endo K, Suzuki A, Fujikura Y, Tsubota K . Improvement of tear stability following warm compression in patients with meibomian gland dysfunction. Adv Exp Med Biol 2002; 506: 1149–1152.
Macsai MS . The role of Omega-3 dietary supplementation in blepharitis and meibomian gland dysfunction. (An AOS thesis). Trans Am Ophthalmol Soc 2008; 106: 336–356.
Pinna A, Piccinini P, Carta F . Effect of oral linoleic and gamma-linolenic acid on meibomian gland dysfunction. Cornea 2007; 26: 260–264.
Sullivan BD, Cermak JM, Sullivan RM, Papas AS, Evans JE, Dana MR et al. Correlations between nutrient intake and the polar lipid profiles of meibomian gland secretions in women with Sjogren’s syndrome. Adv Exp Med Biol 2002; 506: 441–447.
Oleñik A, Jiménez-Alfaro I, Alejandre-Alba A, Mahillo-Fernández I . A randomized, double-masked study to evaluate the effect of omega-3 fatty acids supplementation in meibomian gland dysfunction. Clin Interv Aging 2013; 8: 1133–1138.
Wojtowicz JC, Butovich I, Uchiyama E, Aronowicz J, Agee S, McCulley JP . Pilot, prospective, randomized, double-masked, placebo-controlled clinical trial of an omega-3 supplement for dry eye. Cornea 2011; 30: 308–314.
Creuzot C, Passemard M, Viau S, Joffre C, Pouliquen P, Elena PP et al. Improvement of dry eye symptoms with polyunsaturated fatty acids. J Fr Ophtalmol 2006; 29: 868–873.
Lindsley K, Matsumura S, Hatef E, Akpek EK . Interventions for chronic blepharitis. Cochrane Database Syst Rev 2012; 5: CD005556.
Miljanovic B, Trivedi KA, Dana MR, Gilbard JP, Buring JE, Schaumberg DA . Relation between dietary n-3 and n-6 fatty acids and clinically diagnosed dry eye syndrome in women. Am J Clin Nutr 2005; 82: 887–893.
Ambrosio RJ Jr, Stelzner SK, Boerner CF . Nutrition and dry eye: the role of lipids. Rev Refract Surg 2002; 1: 29–32.
Rand AL, Asbell PA . Nutritional supplements for dry eye syndrome. Curr Opin Ophthalmol 2011; 22: 279–282.
Calder PC . n-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am J Clin Nutr 2006; 83 (6 Suppl): 1505S–1519S.
Erdinest N, Shmueli O, Grossman Y, Ovadia H, Solomon A . Anti-inflammatory effects of alpha linolenic acid on human corneal epithelial cells. Invest Ophthalmol Vis Sci 2012; 53: 4396–4406.
Lewis RA, Lee TH, Austen KF . Effects of Omega-3 Fatty Acids on the Generation of Products of the 5-lipoxygenase Pathway. Academic Press: Orlando, FL, USA, 1986.
Endres S, Ghorbani R, Kelley VE, Georgilis K, Lonnemann G, van der Meer JWM et al. The effect of dietary supplementation with n-3 polyunsaturated fatty acids on the synthesis of interleukin-1 and tumor necrosis factor by mononuclear cells. N Engl J Med 1989; 320: 265–271.
American Academy of Ophthalmology Cornea/External Disease Panel. Preferred Practice Pattern Guidelines. Blepharitis. American Academy of Ophthalmology: San Francisco, CA, USA, 2013 Available at https://www.aao.org/ppp (accessed on 18 September 2015).
Auw-Hädrich C, Reinhard T . [Treatment of chronic blepharokeratoconjunctivitis with local calcineurin inhibitors]. Ophthalmologe 2009; 106: 635–638.
Rubin M, Rao SN . Efficacy of topical cyclosporin 0.05% in the treatment of posterior blepharitis. J Ocul Pharmacol Ther 2006; 22: 47–53.
Meisler DM, Raizman MB, Traboulsi EI . Oral erythromycin treatment for childhood blepharokeratitis. J AAPOS 2000; 4: 379–380.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Poster presented at the 2014 ANNUAL AAPOS MEETING, Palm Springs, CA, USA.
Rights and permissions
About this article
Cite this article
Rodríguez-García, A., González-Godínez, S. & López-Rubio, S. Blepharokeratoconjunctivitis in childhood: corneal involvement and visual outcome. Eye 30, 438–446 (2016). https://doi.org/10.1038/eye.2015.249
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2015.249
This article is cited by
-
The visual impact of higher-order aberrations in patients with pediatric blepharokeratoconjunctivitis
International Ophthalmology (2024)
-
Evaluation of the ocular surface characteristics and Demodex infestation in paediatric and adult blepharokeratoconjunctivitis
BMC Ophthalmology (2019)