Abstract
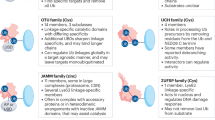
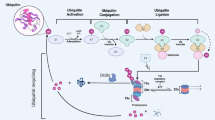
Protein modifications by the covalent linkage of ubiquitin have significant involvement in many cellular processes, including stress response, oncogenesis, viral infection, transcription, protein turnover, organelle biogenesis, DNA repair, cellular differentiation, and cell cycle control. Protein ubiquitination and subsequent degradation by the proteasome require the participation of both ubiquitinating enzymes and deubiquitinating enzymes. Although deubiquitinating enzymes constitute a large family in the ubiquitin system, the study of this class of proteins is still in its infant stage. Recent studies have revealed a variety of molecular and biological functions of deubiquitinating enzymes and their association with human diseases. In this review we will discuss the possible roles that deubiquitinating enzymes may play in cancers.
Similar content being viewed by others
Article PDF
References
Peters JM, Harris JR, Finley D . Ubiquitin and the biology of the cell. New York and London: Plenum, 1998.
Wilkinson KD . Regulation of ubiquitin-dependent processes by deubiquitinating enzymes. FASEB J 1997; 11: 1245–56.
Wilkinson KD . Ubiquitination and deubiquitination: targeting of proteins for degradation by the proteasome. Semin Cell Dev Biol 2000; 11: 141–8.
Chain DG, Casadio A, Schacher S, Hegde AN, Valbrun M, Yamamoto N, et al. Mechanisms for generating the autonomous cAMP-dependent protein kinase required for long-term facilitation in Aplysia. Neuron 1999; 22: 147–56.
Verma R, Aravind L, Oania R, McDonald WH, Yates JR III, Koonin EV, et al. Role of Rpn11 metalloprotease in deubiqui-tination and degradation by the 26S proteasome. Science 2002; 298: 611–5.
Yao T, Cohen RE . A cryptic protease couples deubiquitination and degradation by the proteasome. Nature 2002; 419: 403–7.
Hanna J, Hathaway NA, Tone Y, Crosas B, Elsasser S, Kirkpatrick DS, et al. Deubiquitinating enzyme Ubp6 functions noncatalytically to delay proteasomal degradation. Cell 2006; 127: 99–111.
Liu Y, Fallon L, Lashuel HA, Liu Z, Lansbury PT Jr . The UCH-L1 gene encodes two opposing enzymatic activities that affect alpha-synuclein degradation and Parkinson's disease susceptibility. Cell 2002; 111: 209–18.
Wilkinson KD, Lee KM, Deshpande S, Duerksen-Hughes P, Boss JM, Pohl J . The neuron-specific protein PGP 9.5 is a ubiquitin carboxyl-terminal hydrolase. Science 1989; 246: 670–3.
Dhillon AP, Rode J, Dhillon DP, Moss E, Thompson RJ, Spiro SG, et al. Neural markers in carcinoma of the lung. Br J Cancer 1985; 51: 645–52.
Rode J, Dhillon AP, Doran JF, Jackson P, Thompson RJ . PGP 9.5, a new marker for human neuroendocrine tumours. Histopathology 1985; 9: 147–58.
Hibi K, Westra WH, Borges M, Goodman S, Sidransky D, Jen J . PGP9.5 as a candidate tumor marker for non-small-cell lung cancer. Am J Pathol 1999; 155: 711–5.
Tezel E, Hibi K, Nagasaka T, Nakao A . PGP9.5 as a prognostic factor in pancreatic cancer. Clin Cancer Res 2000; 6: 4764–7.
Yamazaki T, Hibi K, Takase T, Tezel E, Nakayama H, Kasai Y, et al. PGP9.5 as a marker for invasive colorectal cancer. Clin Cancer Res 2002; 8: 192–5.
Otsuki T, Yata K, Takata-Tomokuni A, Hyodoh F, Miura Y, Sakaguchi H, et al. Expression of protein gene product 9.5 (PGP9. 5)/ubiquitin-C-terminal hydrolase 1 (UCHL-1) in human myeloma cells. Br J Haematol 2004; 127: 292–8.
Al-Katib AM, Mohammad RM, Maki A, Smith MR . Induced expression of a ubiquitin COOH-terminal hydrolase in acute lymphoblastic leukemia. Cell Growth Differ 1995; 6: 211–7.
Mohammad RM, Maki A, Pettit GR, Al-Katib AM . Bryostatin 1 induces ubiquitin COOH-terminal hydrolase in acute lympho-blastic leukemia cells. Enzyme Protein 1996; 49: 262–72.
Caballero OL, Resto V, Patturajan M, Meerzaman D, Guo MZ, Engles J, et al. Interaction and colocalization of PGP9.5 with JAB1 and p27(Kip1). Oncogene 2002; 21: 3003–10.
Slingerland J, Pagano M . Regulation of the cdk inhibitor p27 and its deregulation in cancer. J Cell Physiol 2000; 183: 10–17.
Hommura F, Dosaka-Akita H, Mishina T, Nishi M, Kojima T, Hiroumi H, et al. Prognostic significance of p27KIP1 protein and ki-67 growth fraction in non-small cell lung cancers. Clin Cancer Res 2000; 6: 4073–81.
Yao J, Eu KW, Seow-Choen F, Cheah PY . Down-regulation of p27 is a significant predictor of poor overall survival and may facilitate metastasis in colorectal carcinomas. Int J Cancer 2000; 89: 213–6.
Hirano K, Minamoto T . Altered expression of p53 and p27 proteins, alone or combined, as a predictor of metastatic potential in early invasive carcinoma of colon and rectum—a comparative clinicopathologic and molecular analysis. Cancer Detect Prev 2000; 24: 343–55.
Gavioli R, Frisan T, Vertuani S, Bornkamm GW, Masucci MG . c-myc overexpression activates alternative pathways for intracel-lular proteolysis in lymphoma cells. Nat Cell Biol 2001; 3: 283–8.
Rolen U, Kobzeva V, Gasparjan N, Ovaa H, Winberg G, Kisseljov F, et al. Activity profiling of deubiquitinating enzymes in cervical carcinoma biopsies and cell lines. Mol Carcinog 2006; 45: 260–9.
Ciechanover A, DiGiuseppe JA, Bercovich B, Orian A, Richter JD, Schwartz AL, et al. Degradation of nuclear oncoproteins by the ubiquitin system in vitro. Proc Natl Acad Sci USA 1991; 88: 139–43.
Maki CG, Huibregtse JM, Howley PM . In vivo ubiquitination and proteasome-mediated degradation of p53(1). Cancer Res 1996; 56: 2649–54.
Lipkowitz S . The role of the ubiquitination-proteasome pathway in breast cancer: ubiquitin mediated degradation of growth factor receptors in the pathogenesis and treatment of cancer. Breast Cancer Res 2003; 5: 8–15.
Shtiegman K, Yarden Y . The role of ubiquitylation in signaling by growth factors: implications to cancer. Semin Cancer Biol 2003; 13: 29–40.
Amit S, Ben-Neriah Y . NF-kappaB activation in cancer: a challenge for ubiquitination- and proteasome-based therapeutic approach. Semin Cancer Biol 2003; 13: 15–28.
Li M, Chen D, Shiloh A, Luo J, Nikolaev AY, Qin J, et al. Deubiquitination of p53 by HAUSP is an important pathway for p53 stabilization. Nature 2002; 416: 648–53.
Cummins JM, Vogelstein B . HAUSP is required for p53 destabilization. Cell Cycle 2004; 3: 689–92.
Li M, Brooks CL, Kon N, Gu W . A dynamic role of HAUSP in the p53-Mdm2 pathway. Mol Cell 2004; 13: 879–86.
Graner E, Tang D, Rossi S, Baron A, Migita T, Weinstein LJ, et al. The isopeptidase USP2a regulates the stability of fatty acid synthase in prostate cancer. Cancer Cell 2004; 5: 253–61.
Schoenfeld AR, Apgar S, Dolios G, Wang R, Aaronson SA . BRCA2 is ubiquitinated in vivo and interacts with USP11, a deubiquitinating enzyme that exhibits prosurvival function in the cellular response to DNA damage. Mol Cell Biol 2004; 24: 7444–55.
Zhang D, Zaugg K, Mak TW, Elledge SJ . A role for the deubiquitinating enzyme USP28 in control of the DNA-damage response. Cell 2006; 126: 529–42.
Jensen DE, Proctor M, Marquis ST, Gardner HP, Ha SI, Chodosh LA, et al. BAP1: a novel ubiquitin hydrolase which binds to the BRCA1 RING finger and enhances BRCA1-mediated cell growth suppression. Oncogene 1998; 16: 1097–112.
Brummelkamp TR, Nijman SM, Dirac AM, Bernards R . Loss of the cylindromatosis tumour suppressor inhibits apoptosis by activating NF-kappaB. Nature 2003; 424: 797–801.
Trompouki E, Hatzivassiliou E, Tsichritzis T, Farmer H, Ashworth A, Mosialos G . CYLD is a deubiquitinating enzyme that negatively regulates NF-kappaB activation by TNFR family members. Nature 2003; 424: 793–6.
Kovalenko A, Chable-Bessia C, Cantarella G, Israel A, Wallach D, Courtois G . The tumour suppressor CYLD negatively regulates NF-kappaB signalling by deubiquitination. Nature 2003; 424: 801–5.
Liu LQ, Ilaria R Jr, Kingsley PD, Iwama A, van Etten RA, Palis J, et al. A novel ubiquitin-specific protease, UBP43, cloned from leukemia fusion protein AML1-ETO-expressing mice, functions in hematopoietic cell differentiation. Mol Cell Biol 1999; 19: 3029–38.
Migone TS, Humbert M, Rascle A, Sanden D, D'Andrea A, Johnston JA . The deubiquitinating enzyme DUB-2 prolongs cytokine-induced signal transducers and activators of transcription activation and suppresses apoptosis following cytokine withdrawal. Blood 2001; 98: 1935–41.
Janssen JW, Schleithoff L, Bartram CR, Schulz AS . An onco-genic fusion product of the phosphatidylinositol 3-kinase p85beta subunit and HUMORF8, a putative deubiquitinating enzyme. Oncogene 1998; 16: 1767–72.
Geddy PM, Gray S, Reid WA . Mast cell density and PGP 9.5-immunostained nerves in angioleiomyoma: their relationship to painful symptoms. Histopathology 1993; 22: 387–90.
Zhang HW, Iida Y, Andoh T, Nojima H, Murata J, Saiki I, et al. Mechanical hypersensitivity and alterations in cutaneous nerve fibers in a mouse model of skin cancer pain. J Pharmacol Sci 2003; 167–70.
Sasaki H, Yukiue H, Moriyama S, Kobayashi Y, Nakashima Y, Kaji M, et al. Expression of the protein gene product 9.5, PGP9.5, is correlated with T-status in non-small cell lung cancer. Jpn J Clin Oncol 2001; 31: 532–5.
Brichory F, Beer D, Le Naour F, Giordano T, Hanash S . Proteomics-based identification of protein gene product 9.5 as a tumor antigen that induces a humoral immune response in lung cancer. Cancer Res 2001; 61: 7908–12.
Vassilev LT, Vu BT, Graves B, Carvajal D, Podlaski F, Filipovic Z, et al. In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science 2004; 303: 844–8.
Adams J . Proteasome inhibitors as new anticancer drugs. Curr Opin Oncol 2002; 628–34.
Lenz HJ . Clinical update: proteasome inhibitors in solid tumors. Cancer Treat Rev 2003; 29 ( Suppl 1): 41–8.
Richardson P . Clinical update: proteasome inhibitors in hemato-logic malignancies. Cancer Treat Rev 2003; 29 ( Suppl 1): 33–9.
Orlowski RZ, Dees EC . The role of the ubiquitination-proteasome pathway in breast cancer: applying drugs that affect the ubiquitin-proteasome pathway to the therapy of breast cancer. Breast Cancer Res 2003; 5: 1–7.
Mack PC, Davies AM, Lara PN, Gumerlock PH, Gandara DR . Integration of the proteasome inhibitor PS-341 (Velcade) into the therapeutic approach to lung cancer. Lung Cancer 2003; 41 ( Suppl 1): S89–96.
Liu Y, Lashuel HA, Choi S, Xing X, Case A, Ni J, et al. Discovery of inhibitors that elucidate the role of UCH-L1 activity in the H1299 lung cancer cell line. Chem Biol 2003; 10: 837–46.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by grants from the US Health Service NIH/NCI (No CA109371) and (No CA66077).
Rights and permissions
About this article
Cite this article
Yang, Jm. Emerging roles of deubiquitinating enzymes in human cancer. Acta Pharmacol Sin 28, 1325–1330 (2007). https://doi.org/10.1111/j.1745-7254.2007.00687.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00687.x
Keywords
This article is cited by
-
USP39 interacts with SIRT7 to promote cervical squamous cell carcinoma by modulating autophagy and oxidative stress via FOXM1
Journal of Translational Medicine (2023)
-
HAUSP regulates c-MYC expression via de-ubiquitination of TRRAP
Cellular Oncology (2015)
-
A putative OTU domain-containing protein 1 deubiquitinating enzyme is differentially expressed in thyroid cancer and identifies less-aggressive tumours
British Journal of Cancer (2014)