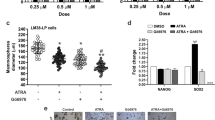
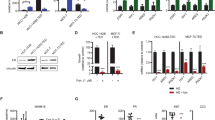
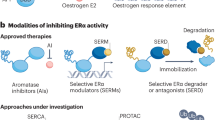
Abstract
Although retinoids are known to be inhibitory to breast cancer cell growth, a key remaining question is whether they would remain effective if administered long-term. We describe here the long-term effects of all-trans retinoic acid on two oestrogen-dependent human breast cancer cell lines MCF7 and ZR-75-1. Although both cell lines were growth inhibited by retinoic acid in the short-term in either the absence or the presence of oestradiol, prolonged culture with 1 μM all-trans retinoic acid resulted in the cells acquiring resistance to the growth inhibitory effects of retinoic acid. Time courses showed that oestrogen deprivation of the cell lines resulted in upregulation of the basal non-oestrogen stimulated growth rate such that cells learned to grow at the same rate without as with oestradiol, but the cells remained growth inhibited by retinoic acid throughout. Addition of 1 μM all-trans retinoic acid to steroid deprivation conditions resulted in reproducible loss of growth response to both retinoic acid and oestradiol, although the time courses were separable in that loss of growth response to retinoic acid preceded that of oestradiol. Loss of growth response to retinoic acid did not involve loss of receptors, ER as measured by steroid binding assay or RARα as measured by Northern blotting. Function of the receptors was retained in terms of the ability of both oestradiol and retinoic acid to upregulate pS2 gene expression, but there was reduced ability to upregulate transiently transfected ERE- and RRE-linked reporter genes. Despite the accepted role of IGFBP3 in retinoic acid-mediated growth inhibition, progression to retinoic acid resistance occurred irrespective of level of IGFBP3, which remained high in the resistant MCF7 cells. Measurement of AP1 activity showed that the two cell lines had markedly different basal AP1 activities, but that progression to resistance was accompanied in both cases by a lost ability of retinoic acid to reduce AP1 activity. These results warn of potential resistance which could arise on long-term treatment with retinoic acid in a clinical situation and echo the problems of progression to endocrine resistance. It seems that whatever the constraints imposed on growth, these cells have a remarkable ability to escape from growth inhibition. However, the ability of retinoic acid to delay progression to oestrogen resistance is encouraging for endocrine therapy, and the concentration-dependence of retinoic acid resistance suggests that progression is not absolute but could be manipulated by dose. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Adamo ML, Shao ZM, Lanau F, Chen JC, Clemmons DR, Roberts CT, LeRoith D and Fontana JA (1992) Insulin-like growth factor-1 (IGF1) and retinoic acid modulation of IGF-binding proteins (IGFBPs): IGFBP-2, -3, and -4 gene expression and protein secretion in a breast cancer cell line. Endocrinol 131: 1858–1866
Anzano MA, Byers SW, Smith JM, Peer CW, Mullen LT, Brown CC, Roberts AB and Sporn MB (1994) Prevention of breast cancer in the rat with 9-cis-retinoic acid as a single agent and in combination with tamoxifen. Cancer Res 54: 4614–4617
Brown AMC, Jeltsch JM, Roberts M and Chambon P (1984) Activation of pS2 gene transcription is a primary response to estrogen in the human breast cancer cell line MCF-7. Proc Natl Acad Sci USA 81: 6344–6348
Budd GT, Adamson PC, Gupta M, Homayoun P, Sandstrom SK, Murphy RF, McLain D, Tuason L, Peereboom D, Bukowski RM and Ganapathi R (1998) Phase I/II trial of all-trans retinoic acid and tamoxifen in patients with advanced breast cancer. Clin Cancer Res 4: 635–642
Butler WB and Fontana JA (1992) Responses to retinoic acid of tamoxifen-sensitive and -resistant sublines of human breast cancer cell line MCF-7. Cancer Res 52: 6164–6167
Chambon P (1996) A decade of molecular biology of retinoic acid receptors. FASEB J 10: 940–954
Chen JY, Penco S, Ostrowski J, Balaguer P, Pons M, Starrett JE, Reczek P, Chambon P and Gronemeyer H (1995) RAR-specific agonist/antagonists which dissociate transactivation and AP1 transrepression inhibit anchoarge-independent cell proliferation. EMBO J 14: 1187–1197
Costa A (1993) Breast cancer chemoprevention. Eur J Cancer 29A: 589–592
Daly RJ and Darbre PD (1990) Cellular and molecular events in loss of estrogen sensitivity in ZR-75–1 and T-47-D human breast cancer cells. Cancer Res 50: 5868–5875
Daly RJ, King RJB and Darbre PD (1990) Interaction of growth factors during progression towards steroid independence in T-47-D human breast cancer cells. J Cell Biochem 43: 199–211
Darbre PD and Daly RJ (1989) Effects of oestrogen on human breast cancer cells in culture. Proc Roy Soc Edin 95B: 119–132
Darbre P, Yates J, Curtis S and King RJB (1983) Effect of estradiol on human breast cancer cells in culture. Cancer Res 43: 349–354
Daschner PJ, Ciolino HP, Plouzek CA and Yeh GC (1999) Increased AP-1 activity in drug resistant human breast cancer MCF-7 cells. Breast Cancer Res Treat 53: 229–240
Dawson MI, Chao WR, Pine P, Jong L, Hobbs PD, Rudd CK, Quick TC, Niles RM, Zhang XK, Lombardo A, Ely KR, Shroot B and Fontana JA (1995) Correlation of retinoid binding affinity to retinoic acid receptor α with retinoid inhibition of growth of estrogen receptor-positive MCF-7 mammary carcinoma cells. Cancer Res 55: 4446–4451
Demirpence E, Balaguer P, Trousse F, Nicolas JC, Pons M and Gagne D (1994) Antiestrogenic effects of all-trans retinoic acid and 1,25-dihydroxyvitamin D3 in breast cancer cells occur at the estrogen response element level but through different molecular mechanisms. Cancer Res 54: 1458–1464
DiSepio D, Sutter M, Johnson AT, Chandraratna RA and Nagpal S (1999) Identification of the AP1-antagonism domain of retinoic acid receptors. Mol Cell Biol Res Commun 1: 7–13
Engel LW, Young NA, Trolka TS, Lippman ME, O'Brian SJ and Joyce MJ (1978) Establishment and characterisation of three new continuous cell lines derived from human breast carcinomas. Cancer Res 38: 3352–3364
Fontana JA (1987) Interaction of retinoids and tamoxifen on the inhibition of human mammary carcinoma cell proliferation. Exp Cell Biol 55: 136–144
Fontana JA, Mezu AB, Cooper BN and Miranda D (1990) Retinoid modulation of estradiol-stimulated growth and of protein synthesis and secretion in human breast carcinoma cells. Cancer Res 50: 1997–2002
Fontana JA, Burrows-Mezu A, Clemmons DR and LeRoith D (1991) Retinoic modulation of insulin-like growth binding proteins and inhibition of breast carcinoma proliferation. Endocrinol 128: 1115–1122
Fontana JA, Nervi C, Shao ZM and Jetten AM (1992) Retinoid antagonism of estrogen-responsive transforming growth factor alpha and pS2 gene expression in breast carcinoma cells. Cancer Res 52: 3938–3945
Gottlicher M, Heck S and Herrlich P (1998) Transcriptional cross-talk, the second mode of steroid hormone receptor action. J Mol Med 76: 480–489
Green B, Leake RE (1987) Steroid Hormones: A Practical Approach. IRL Press: Oxford, UK
Grubbs CJ, Moon RC, Sporn MB and Newton DL (1977) Inhibition of mammary cancer by retinyl methyl ester. Cancer Res 37: 599–602
Hossenlopp P, Seurin D, Quinson BS, Hardouin S and Binoux M (1986) Analysis of serum insulin-like growth factor binding proteins using Western blotting: use of the method for titration of the binding proteins and competitive binding studies. Anal Biochem 154: 138–143
Jeng MH, Shupnik MA, Bender TP, Westin EH, Bandyopadhyay D, Kumar R, Masamura S and Santen RJ (1998) Estrogen receptor expression and function in long-term estrogen-deprived human breast cancer cells. Endocrinol 139: 4164–4174
Katzenellenbogen BS, Kendra KL, Norman MJ and Berthois Y (1987) Proliferation, hormonal responsiveness, and estrogen receptor content of MCF-7 human breast cancer cells grown in the short-term and long-term absence of estrogens. Cancer Res 47: 4355–4360
Kazmi SMI, Plante RK, Visconti V and Lau CY (1996) Comparison of N-(4-hydroxyphenyl)retinamide and all-trans retinoic acid in the regulation of retinoid receptor-mediated gene expression in human breast cancer cell lines. Cancer Res 56: 1056–1062
Kizaki M, Fukuchi Y and Ikeda Y (1999) A novel retinoic acid-resistant acute promyelocytic leukemia model in vitro and in vivo (review). Int J Mol Med 4: 359–364
Koga M and Sutherland RL (1991) Retinoic acid acts synergistically with 1,25-dihydroxyvitamin D3 or antioestrogen to inhibit T47D human breast cancer cell proliferation. J Steroid Biochem Molec Biol 39: 455–460
Lacroix A and Lippman ME (1980) Binding of retinoids to human breast cancer cell lines and their effects on cell growth. J Clin Invest 65: 586–591
Lacroix A, L’Heureux N and Bhat PV (1984) Cytoplasmic retinoic acid-binding protein in retinoic acid-resistant human breast cancer sublines. J Natl Cancer Inst 73: 793–800
Lamph WW, Wamsley P, Sassone-Corsi P and Verma IM (1988) Induction of protooncogene Jun/AP1 by serum and TPA. Nature 334: 629–631
Larssen SS, Madsen MW, Jensen BL and Lykesfeldt AE (1997) Resistance of human breast-cancer cells to the pure steroidal anti-oestrogen ICI 182,780 is not associated with a general loss of estrogen-receptor expression or lack of estrogen responsiveness. Int J Cancer 72: 1129–1136
Li XS, Shao ZM, Sheikh MS, Eiseman JL, Sentz D, Jetten AM, Chen JC, Dawson MI, Aisner S, Rishi AK, Gutierrez P, Schnapper L and Fontana JA (1995) Retinoic acid nuclear receptor β inhibits breast carcinoma anchorage independent growth. J Cell Physiol 165: 449–458
Lotan R (1979) Different susceptibilities of human melanoma and breast cancer cell lines to retinoic acid-induced growth inhibition. Cancer Res 39: 1014–1019
Marth B, Bock G and Daxenbichler G (1985) Effect of 4-hydroxyphenylretinamide and retinoic acid on proliferation and cell cycle of cultured human breast cancer cells. J Natl Cancer Inst 75: 871–875
Masiakowski P, Breathnach R, Bloch J, Gannon F, Krust A and Chambon P (1982) Cloning of cDNA sequences of hormone-regulated genes from the MCF-7 human breast cancer cell line. Nucleic Acids Res 10: 7895–7903
Miller WR (1996). Estrogen and Breast Cancer, Chapman and Hall
Moon RC, Grubbs CJ and Sporn MB (1976) Inhibition of 7,12-dimethyl benz (α) anthracene-induced mammary carcinogenesis by retinyl acetate. Cancer Res 36: 2627–2630
Moon RC, Grubbs CJ, Sporn MB and Goodman DG (1977) Retinyl-acetate inhibits mammary carcinogenesis induced by N-methyl-N-nitrosourea. Nature 267: 620–621
Oh Y (1998) IGF-independent regulation of breast cancer growth by IGF binding proteins. Breast Cancer Res Treat 47: 283–293
Osborne CK, Hobbs K and Trent JM (1987) Biological differences among MCF-7 human breast cancer cell lines from different laboratories. Breast Cancer Res Treat 9: 111–121
Rettura G, Schittek A, Hardy M, Levenson SM, Demetriou A and Seifter E (1975) Antitumour action of vitamin A in mice inoculated with adenocarcinoma cells. J Natl Cancer Inst 54: 1489–1491
Rishi AK, Shao ZM, Baumann RG, Li XS, Sheikh MS, Kimura S, Bashirelahi N and Fontana JA (1995) Estradiol regulation of the human retinoic acid receptor α gene in human breast carcinoma cells is mediated via an imperfect half-palindromic estrogen response element and Sp1 motifs. Cancer Res 55: 4999–5006
Roman SD, Clarke CL, Hall RE, Alexander IE and Sutherland RL (1992) Expression and regulation of retinoic acid receptors in human breast cancer cells. Cancer Res 52: 2236–2242
Roman SD, Ormandy CJ, Manning DL, Blamey RW, Nicholson RI, Sutherland RL and Clarke CL (1993) Estradiol induction of retinoic acid receptors in human breast cancer cells. Cancer Res 53: 5940–5945
Rubin M, Fenig E, Rosenauer A, Menendez-Botet C, Achkar C, Bentel JM, Yahalom J, Mendelsohn J and Miller WH (1994) 9-cis retinoic acid inhibits growth of breast cancer cells and down-regulates estrogen receptor RNA and protein. Cancer Res 54: 6549–6556
Saceda M, Lippman ME, Chambon P, Lindsey RL, Ponglikitmongkol M, Puente M and Martin MB (1988) Regulation of the estrogen receptor in MCF-7 cells by estradiol. Mol Endocrinol 2: 1157–1162
Salacinski PRP, McLean C, Sykes JEC, Clement-Jones VV and Lowry PJ (1981) Iodination of proteins, glycoproteins, and peptides using a solid phase oxidising agent, 1,3,4,6-tetrachloro-3,6-diphenyl glycouril (Iodogen). Anal Biochem 117: 136–146
Sambrook J, Fritsch EF and Maniatis T (1989). Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory Press: New York, USA
Shang Y, Baumrucker CR and Green MH (1999) Signal relay by retinoic acid receptors α and β in the retinoic acid-induced expression of insulin-like growth factor-binding protein-3 in breast cancer cells. J Biol Chem 274: 18005–18010
Sheikh MS, Shao ZM, Chen JC, Hussain A, Jetten AM and Fontana JA (1993) Estrogen receptor-negative breast cancer cells transfected with the estrogen receptor exhibit increased RAR alpha gene expression and sensitivity to growth inhibition by retinoic acid. J Cell Biochem 53: 394–404
Sheikh MS, Shao ZM, Li XS, Dawson M, Jetten AM, Wu S, Conley BA, Garcia M, Rochefort H and Fontana JA (1994) Retinoid-resistant estrogen receptor-negative human breast carcinoma cells transfected with retinoic acid receptor-alpha acquire sensitivity to growth inhibition by retinoids. J Biol Chem 269: 21440–21447
Slack JL (1999) Biology and treatment of acute progranulocytic leukemia. Curr Opin Hematol 6: 236–240
Sleigh MJ (1986) A nonchromatographic assay for expression of the chloramphenicol acetyltransferase gene in eucaryotic cells. Anal Biochem 156: 251–256
Soprano DR, Chen LX, Wu S, Donigan AM, Borghaei RC and Soprano KJ (1996) Overexpression of both RAR and RXR restores AP-1 repression in ovarian adenocarcinoma cells resistant to retinoic acid-dependent growth inhibition. Oncogene 12: 577–584
Stephen RL (1998) The role of retinoic acid in the growth regulation of human breast cancer cells. Ph D Thesis, The University of Reading, UK
Takatsuka J, Takahashi N and De Luca LM (1996) Retinoic acid metabolism and inhibition of cell proliferation: an unexpected liaison. Cancer Res 56: 675–678
Ueda H, Ono M, Hagino Y and Kuwano M (1985) Isolation of retinoic acid-resistant clones from human breast cancer cell line MCF-7 with altered activity of cellular retinoic acid-binding protein. Cancer Res 45: 3332–3338
Umesono K, Murakami KK, Thompson CC and Evans RM (1991) Direct repeats as selective response elements for the thyroid hormone, retinoic acid, and vitamin D3 receptors. Cell 65: 1255–1266
van der Burg B, van der Leede BM, Kwakkenbos-Isbrucker L, Salverda S, de Laat SW and van der Saag PT (1993) Retinoic acid resistance of estradiol-independent breast cancer cells coincides with diminished retinoic acid receptor function. Mol Cell Endocrinol 91: 149–157
van der Burg B, Slager-Davidov R, van der Leede BM, de Laat SW and van der Saag PT (1995) Differential regulation of AP1 activity by retinoic acid in hormone-dependent and -independent breast cancer cells. Mol Cell Endocrinol 112: 143–152
Wakeling AE (1993) Are breast tumours reistant to tamoxifen also resistant to pure antioestrogens?. J Steroid Biochem Molec Biol 47: 107–114
Welshons WV and Jordan VC (1987) Adaptation of estrogen-dependent MCF-7 cells to low estrogen (phenol red-free) culture. Eur J Cancer Clin Oncol 23: 1935–1939
Wetherall NT and Taylor CM (1986) The effects of retinoid treatment and antiestrogens on the growth of T47D human breast cancer cells. Eur J Cancer Clin Oncol 22: 53–59
Wigler M, Pellicer A, Silverstein S, Axel R, Urlaub G and Chasin L (1979) DNA mediated transfer of the adenine phosphoribosyltransferase locus into mammalian cells. Proc Natl Acad Sci USA 76: 1373–1376
Yang LM, Kim HT, Munoz-Medellin D, Reddy P and Brown PH (1997) Induction of retinoid resistance in breast cancer cells by overexpression of c-jun. Cancer Res 57: 4652–4661
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Stephen, R., Darbre, P. Loss of growth inhibitory effects of retinoic acid in human breast cancer cells following long-term exposure to retinoic acid. Br J Cancer 83, 1183–1191 (2000). https://doi.org/10.1054/bjoc.2000.1388
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1388