Abstract
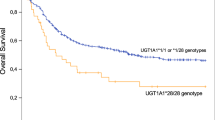
Nilotinib is a novel BCR-ABL inhibitor with significantly improved potency and selectivity over imatinib. In Phase I and Phase II clinical studies of nilotinib in patients with a variety of leukemias, infrequent instances of reversible, benign elevation of bilirubin were observed. Uridine diphosphate glucuronosyltransferase 1A1 (UGT1A1) glucuronidates bilirubin in humans, and a polymorphism in the promoter of the gene that encodes it has been associated with hyperbilirubinemia during treatment with a number of drugs. Pharmacogenetic analysis of that TA-repeat polymorphism found an association between the (TA)7/(TA)7 genotype and risk of hyperbilirubinemia in Phase I patients with imatinib-resistant/intolerant chronic myeloid leukemia (CML) or relapsed/refractory Ph+ acute lymphoblastic leukemia (ALL); this result was replicated in two separate analyses of the chronic phase (CP) and accelerated phase (AP) CML arms of a Phase II study. As nilotinib is not known to be glucuronidated by UGT1A1, the combined impact of inhibition of UGT1A1 activity by nilotinib and genetic polymorphism is the most likely cause of the increased rate of hyperbilirubinemia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bosma PJ, Seppen J, Goldhoorn B, Bakker C, Oude Elferink RP, Chowdhury JR et al. Bilirubin UDP-glucuronosyltransferase 1 is the only relevant bilirubin glucuronidating isoform in man. J Biol Chem 1994; 269: 17960–17964.
Bosma PJ, Chowdhury JR, Bakker C, Gantla S, de Boer A, Oostra BA et al. The genetic basis of the reduced expression of bilirubin UDP-glucuronosyltransferase 1 in Gilbert's syndrome. N Engl J Med 1995; 333: 1171–1175.
Monaghan G, Ryan M, Seddon R, Hume R, Burchell B . Genetic variation in bilirubin UPD-glucuronosyltransferase gene promoter and Gilbert's syndrome. Lancet 1996; 347: 578–581.
Beutler E, Gelbart T, Demina A . Racial variability in the UDP-glucuronosyltransferase 1 (UGT1A1) promoter: a balanced polymorphism for regulation of bilirubin metabolism? Proc Natl Acad Sci USA 1998; 95: 8170–8174.
Premawardhena A, Fisher CA, Liu YT, Verma IC, de Silva S, Arambepola M et al. The global distribution of length polymorphisms of the promoters of the glucuronosyltransferase 1 gene (UGT1A1): hematologic and evolutionary implications. Blood Cells Mol Dis 2003; 31: 98–101.
Iyer L, King CD, Whitington PF, Green MD, Roy SK, Tephly TR et al. Genetic predisposition to the metabolism of irinotecan (CPT-11). Role of uridine diphosphate glucuronosyltransferase isoform 1A1 in the glucuronidation of its active metabolite (SN-38) in human liver microsomes. J Clin Invest 1998; 101: 847–854.
Ando Y, Saka H, Ando M, Sawa T, Muro K, Ueoka H et al. Polymorphisms of UDP-glucuronosyltransferase gene and irinotecan toxicity: a pharmacogenetic analysis. Cancer Res 2000; 60: 6921–6926.
Innocenti F, Undevia SD, Iyer L, Chen PX, Das S, Kocherginsky M et al. Genetic variants in the UDP-glucuronosyltransferase 1A1 gene predict the risk of severe neutropenia of irinotecan. J Clin Oncol 2004; 22: 1382–1388.
Danoff TM, Campbell DA, McCarthy LC, Lewis KF, Repasch MH, Saunders AM et al. A Gilbert's syndrome UGT1A1 variant confers susceptibility to tranilast-induced hyperbilirubinemia. Pharmacogenomics J 2004; 4: 49–53.
Zucker SD, Qin X, Rouster SD, Yu F, Green RM, Keshavan P et al. Mechanism of indinavir-induced hyperbilirubinemia. Proc Natl Acad Sci USA 2001; 98: 12671–12676.
Kantarjian H, Giles F, Wunderle L, Bhalla K, O'Brien S, Wassmann B et al. Nilotinib in imatinib-resistant CML and Philadelphia chromosome-positive ALL. N Engl J Med 2006; 354: 2542–2551.
Kantarjian HM, Gattermann N, Hochhaus A, Larson R, Rafferty T, Weitzman A et al. A Phase II study of nilotinib, a novel tyrosine kinase inhibitor administered to imatinib-resistant and -intolerant patients with chronic myelogenous leukemia (CML) in accelerated phase (AP). Blood (ASH Annual Meeting Abstracts) 2006; 108: 2169.
le Coutre P, Bhalla K, Giles F, Baccarani M, Ossenkoppele GJ, Hochhaus A et al. A Phase II study of nilotinib, a novel tyrosine kinase inhibitor administered to imatinib-resistant and -intolerant patients with chronic myelogenous leukemia (CML) in chronic phase (CP). Blood (ASH Annual Meeting Abstracts) 2006; 108: 165.
Cancer Therapy Evaluation Program, National Cancer Institute. Common Terminology Criteria for Adverse Events v3.0. 2006. Available from http://ctep.cancer.gov/reporting/ctc_v30.html.
Maitland ML, Vasisht K, Ratain MJ . TPMT, UGT1A1 and DPYD: genotyping to ensure safer cancer therapy? Trends Pharmacol Sci 2006; 27: 432–437.
Monaghan G, McLellan A, McGeehan A, Li Volti S, Mollica F, Salemi I et al. Gilbert's syndrome is a contributory factor in prolonged unconjugated hyperbilirubinemia of the newborn. J Pediatr 1999; 134: 441–446.
Ferraris A, D'Amato G, Nobili V, Torres B, Marcellini M, Dallapiccola B . Combined test for UGT1A1 -3279T-->G and A(TA)nTAA polymorphisms best predicts Gilbert's syndrome in Italian pediatric patients. Genet Test 2006; 10: 121–125.
Rouits E, Boisdron-Celle M, Dumont A, Guerin O, Morel A, Gamelin E . Relevance of different UGT1A1 polymorphisms in irinotecan-induced toxicity: a molecular and clinical study of 75 patients. Clin Cancer Res 2004; 10: 5151–5159.
Mercke Odeberg J, Andrade J, Holmberg K, Hoglund P, Malmqvist U, Odeberg J . UGT1A polymorphisms in a Swedish cohort and a human diversity panel, and the relation to bilirubin plasma levels in males and females. Eur J Clin Pharmacol 2006; 62: 829–837.
Acknowledgements
In addition to those investigators listed as authors, Kapil Bhala participated as an investigator in this trial. We thank Wade Brown, Elisabeth Leroy and Michele Gysen for their management of sample collection and genotyping. This study was supported by research funding from Novartis Pharmaceuticals. Work was performed at Novartis Institutes for Biomedical Research and Novartis Pharmaceuticals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singer, J., Shou, Y., Giles, F. et al. UGT1A1 promoter polymorphism increases risk of nilotinib-induced hyperbilirubinemia. Leukemia 21, 2311–2315 (2007). https://doi.org/10.1038/sj.leu.2404827
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2404827
Keywords
This article is cited by
-
Relationship between achievement of major molecular response or deep molecular response and nilotinib plasma concentration in patients with chronic myeloid leukemia receiving first-line nilotinib therapy
Cancer Chemotherapy and Pharmacology (2022)
-
Genetic polymorphisms associated with adverse reactions of molecular-targeted therapies in renal cell carcinoma
Medical Oncology (2018)
-
In vitro UGT1A1 inhibition by tyrosine kinase inhibitors and association with drug-induced hyperbilirubinemia
Cancer Chemotherapy and Pharmacology (2018)
-
Role of Preemptive Genotyping in Preventing Serious Adverse Drug Events in South Korean Patients
Drug Safety (2017)
-
Pharmacogenetics and Pharmacogenomics of Targeted Therapeutics in Chronic Myeloid Leukemia
Molecular Diagnosis & Therapy (2017)