Abstract
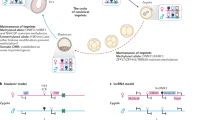
Recent studies have revealed mechanistic parallels between imprinted X-chromosome inactivation and autosomal imprinting. We suggest that neither mechanism was present in ancestral egg-laying mammals, and that both arose when the evolution of the placenta exerted selective pressure to imprint growth-related genes. We also propose that non-coding RNAs and histone modifications were adopted for the imprinting of growth suppressors on the X chromosome and on autosomes. This provides a unified hypothesis for the evolution of X-chromosome inactivation and imprinting.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lyon, M. F. Imprinting and X-chromosome inactivation. Results Probl. Cell Differ. 25, 73–90 (1999).
Heard, E. Recent advances in X-chromosome inactivation. Curr. Opin. Cell Biol. 16, 247–255 (2004).
Brockdorff, N. X-chromosome inactivation: closing in on proteins that bind Xist RNA. Trends Genet. 18, 352–358 (2002).
Lee, J. T. Molecular links between X-inactivation and autosomal imprinting: X-inactivation as a driving force for the evolution of imprinting? Curr. Biol. 13, R242–R254 (2003).
Avner, P. & Heard, E. X-chromosome inactivation: counting, choice and initiation. Nature Rev. Genet. 2, 59–67 (2001).
Andersen, A. A. & Panning, B. Epigenetic gene regulation by noncoding RNAs. Curr. Opin. Cell Biol. 15, 281–289 (2003).
Marin, I., Siegal, M. L. & Baker, B. S. The evolution of dosage-compensation mechanisms. Bioessays 22, 1106–1114 (2000).
Meyer, B. J. Sex in the wormcounting and compensating X-chromosome dose. Trends Genet. 16, 247–253 (2000).
Akhtar, A. Dosage compensation: an intertwined world of RNA and chromatin remodelling. Curr. Opin. Genet. Dev. 13, 161–169 (2003).
Huynh, K. D. & Lee, J. T. Inheritance of a pre-inactivated paternal X chromosome in early mouse embryos. Nature 426, 857–862 (2003).
Okamoto, I., Otte, A. P., Allis, C. D., Reinberg, D. & Heard, E. Epigenetic dynamics of imprinted X inactivation during early mouse development. Science 303, 644–649 (2004).
Mak, W. et al. Reactivation of the paternal X chromosome in early mouse embryos. Science 303, 666–669 (2004).
Lewis, A. et al. Imprinting on distal chromosome 7 in the placenta involves repressive histone methylation independent of DNA methylation. Nature Genet. 36, 1291–1295 (2004).
Umlauf, D. et al. Imprinting along the Kcnq1 domain on mouse chromosome 7 involves repressive histone methylation and recruitment of Polycomb group complexes. Nature Genet. 36, 1296–1300 (2004).
Delaval, K. & Feil, R. Epigenetic regulation of mammalian genomic imprinting. Curr. Opin. Genet. Dev. 14, 188–195 (2004).
Graves, J. A. Mammals that break the rules: genetics of marsupials and monotremes. Annu. Rev. Genet. 30, 233–260 (1996).
Ogawa, Y. & Lee, J. T. Antisense regulation in X inactivation and autosomal imprinting. Cytogenet. Genome Res. 99, 59–65 (2002).
Lyon, M. F. Gene action in the X-chromosome of the mouse (Mus musculus L.). Naturwissenschaften 190, 372–373 (1961).
Lyon, M. F. X-chromosome inactivation. Curr. Biol. 9, R235–R237 (1999).
Cooper, D. W., VandeBerg, J. L., Sharman, G. B. & Poole, W. E. Phosphoglycerate kinase polymorphism in kangaroos provides further evidence for paternal X inactivation. Nature New Biol. 230, 155–157 (1971).
Vandeberg, J. L. Developmental aspects of X chromosome inactivation in eutherian and metatherian mammals. J. Exp. Zool. 228, 271–286 (1983).
Wake, N., Takagi, N. & Sasaki, M. Non-random inactivation of X chromosome in the rat yolk sac. Nature 262, 580–581 (1976).
Xue, F. et al. Aberrant patterns of X chromosome inactivation in bovine clones. Nature Genet. 31, 216–220 (2002).
Takagi, N. & Sasaki, M. Preferential inactivation of the paternally derived X chromosome in the extraembryonic membranes of the mouse. Nature 256, 640–642 (1975).
Frels, W. I. & Chapman, V. M. Expression of the maternally derived X chromosome in the mural trophoblast of the mouse. J. Embryol. Exp. Morphol. 56, 179–190 (1980).
McGrath, J. & Solter, D. Completion of mouse embryogenesis requires both the maternal and paternal genomes. Cell 37, 179–183 (1984).
Surani, M. A., Barton, S. C. & Norris, M. L. Development of reconstituted mouse eggs suggests imprinting of the genome during gametogenesis. Nature 308, 548–550 (1984).
Cattanach, B. M. & Kirk, M. Differential activity of maternally and paternally derived chromosome regions in mice. Nature 315, 496–498 (1985).
Constancia, M., Kelsey, G. & Reik, W. Resourceful imprinting. Nature 432, 53–57 (2004).
Wilkins, J. F. & Haig, D. What good is genomic imprinting: the function of parent-specific gene expression. Nature Rev. Genet. 4, 359–368 (2003).
Killian, J. K. et al. M6P/IGF2R imprinting evolution in mammals. Mol. Cell 5, 707–716 (2000).
Killian, J. K. et al. Monotreme IGF2 expression and ancestral origin of genomic imprinting. J. Exp. Zool. 291, 205–212 (2001).
Reik, W. & Walter, J. Genomic imprinting: parental influence on the genome. Nature Rev. Genet. 2, 21–32 (2001).
Ferguson-Smith, A. C. & Surani, M. A. Imprinting and the epigenetic asymmetry between parental genomes. Science 293, 1086–1089 (2001).
Sleutels, F. & Barlow, D. P. The origins of genomic imprinting in mammals. Adv. Genet. 46, 119–163 (2002).
Bartolomei, M. S. & Tilghman, S. M. Genomic imprinting in mammals. Annu. Rev. Genet. 31, 493–525 (1997).
Frank, D. et al. Placental overgrowth in mice lacking the imprinted gene Ipl. Proc. Natl Acad. Sci. USA 99, 7490–7495 (2002).
Takahashi, K., Kobayashi, T. & Kanayama, N. p57Kip2 regulates the proper development of labyrinthine and spongiotrophoblasts. Mol. Hum. Reprod. 6, 1019–1025 (2000).
Takahashi, K. & Nakayama, K. Mice lacking a CDK inhibitor, p57Kip2, exhibit skeletal abnormalities and growth retardation. J. Biochem. (Tokyo) 127, 73–83 (2000).
Wutz, A. et al. Non-imprinted Igf2r expression decreases growth and rescues the Tme mutation in mice. Development 128, 1881–1887 (2001).
Engemann, S. et al. Sequence and functional comparison in the Beckwith–Wiedemann region: implications for a novel imprinting centre and extended imprinting. Hum. Mol. Genet. 9, 2691–2706 (2000).
Stoger, R. et al. Maternal-specific methylation of the imprinted mouse Igf2r locus identifies the expressed locus as carrying the imprinting signal. Cell 73, 61–71 (1993).
de Napoles, M. et al. Polycomb group proteins ring1A/B link ubiquitylation of histone H2A to heritable gene silencing and X inactivation. Dev. Cell 7, 663–676 (2004).
Wang, J. et al. Imprinted X inactivation maintained by a mouse Polycomb group gene. Nature Genet. 28, 371–375 (2001).
Braidotti, G. et al. The Air non-coding RNA — an imprinted cis-silencing transcript. Cold Spring Harb. Symp. Quant. Biol. 69 (in the press).
Mager, J., Montgomery, N. D., de Villena, F. P. & Magnuson, T. Genome imprinting regulated by the mouse Polycomb group protein Eed. Nature Genet. 33, 502–507 (2003).
Sleutels, F., Zwart, R. & Barlow, D. P. The non-coding Air RNA is required for silencing autosomal imprinted genes. Nature 415, 810–813 (2002).
Sado, T. et al. X inactivation in the mouse embryo deficient for Dnmt1: distinct effect of hypomethylation on imprinted and random X inactivation. Dev. Biol. 225, 294–303 (2000).
Li, Y. & Behringer, R. R. Esx1 is an X-chromosome-imprinted regulator of placental development and fetal growth. Nature Genet. 20, 309–311 (1998).
Shi, W. et al. Choroideremia gene product affects trophoblast development and vascularization in mouse extra-embryonic tissues. Dev. Biol. 272, 53–65 (2004).
Chiao, E. et al. Overgrowth of a mouse model of the Simpson–Golabi–Behmel syndrome is independent of IGF signaling. Dev. Biol. 243, 185–206 (2002).
Tada, T., Takagi, N. & Adler, I. D. Parental imprinting on the mouse X chromosome: effects on the early development of X0, XXY and XXX embryos. Genet. Res. 62, 139–148 (1993).
Reik, W. et al. Chromosome loops, insulators and histone methylation: new insights into regulation of imprinting in clusters. Cold Spring Harb. Symp. Quant. Biol. 69 (in the press).
Wilkins, J. F. & Haig, D. Genomic imprinting of two antagonistic loci. Proc. R. Soc. Lond. B 268, 1861–1867 (2001).
McQueen, H. A., McBride, D., Miele, G., Bird, A. P. & Clinton, M. Dosage compensation in birds. Curr. Biol. 11, 253–257 (2001).
Grutzner, F. & Graves, J. A. A platypus' eye view of the mammalian genome. Curr. Opin. Genet. Dev. 14, 642–649 (2004).
Grutzner, F., Deakin, J., Rens, W., El-Mogharbel, N. & Marshall Graves, J. A. The monotreme genome: a patchwork of reptile, mammal and unique features? Comp. Biochem. Physiol. A 136, 867–881 (2003).
Rens, W. et al. Resolution and evolution of the duck-billed platypus karyotype with an X1Y1X2Y2X3Y3X4Y4X5Y5 male sex chromosome constitution. Proc. Natl Acad. Sci. USA 101, 16257–16261 (2004).
Grutzner, F. et al. In the platypus a meiotic chain of ten sex chromosomes shares genes with the bird Z and mammal X chromosomes. Nature 432, 913–917 (2004).
Ohlsson, R., Paldi, A. & Graves, J. A. Did genomic imprinting and X chromosome inactivation arise from stochastic expression? Trends Genet. 17, 136–141 (2001).
Plagge, A. et al. The imprinted signaling protein XLαs is required for postnatal adaptation to feeding. Nature Genet. 36, 818–826 (2004).
Kaslow, D. C. & Migeon, B. R. DNA methylation stabilizes X chromosome inactivation in eutherians but not in marsupials: evidence for multistep maintenance of mammalian X dosage compensation. Proc. Natl Acad. Sci. USA 84, 6210–6214 (1987).
Suzuki, S. et al. Genomic imprinting of IGF2, p57KIP2 and PEG1/MEST in a marsupial, the tammar wallaby. Mech. Dev. 122, 213–222 (2005).
Zeng, S. M. & Yankowitz, J. X-inactivation patterns in human embryonic and extra-embryonic tissues. Placenta 24, 270–275 (2003).
Grati, F. R. et al. Biparental expression of ESX1L gene in placentas from normal and intrauterine growth-restricted pregnancies. Eur. J. Hum. Genet. 12, 272–278 (2004).
Ray, P. F., Winston, R. M. & Handyside, A. H. XIST expression from the maternal X chromosome in human male preimplantation embryos at the blastocyst stage. Hum. Mol. Genet. 6, 1323–1327 (1997).
Daniels, R., Zuccotti, M., Kinis, T., Serhal, P. & Monk, M. XIST expression in human oocytes and preimplantation embryos. Am. J. Hum. Genet. 61, 33–39 (1997).
Fitzpatrick, G. V., Soloway, P. D. & Higgins, M. J. Regional loss of imprinting and growth deficiency in mice with a targeted deleltion of KvDMR1. Nature Genet. 32, 426–431 (2002).
Acknowledgements
The authors thank G. Kelsey, M. Constancia, M. Hemberger, W. Dean, G. Smits and D. Haig for advice and discussion. This work was supported by grants from the UK Biotechnology and Biological Sciences Research Council, the UK Medical Research Council and the European Union.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
Entrez Gene
FURTHER INFORMATION
Epigenome Network of Excellence web site
Harwell Mouse Imprinting web site
Rights and permissions
About this article
Cite this article
Reik, W., Lewis, A. Co-evolution of X-chromosome inactivation and imprinting in mammals. Nat Rev Genet 6, 403–410 (2005). https://doi.org/10.1038/nrg1602
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrg1602
This article is cited by
-
X-chromosome inactivation: the gift that keeps on giving
Nature Structural & Molecular Biology (2023)
-
Maternal SMCHD1 controls both imprinted Xist expression and imprinted X chromosome inactivation
Epigenetics & Chromatin (2022)
-
Structural basis for MTA1c-mediated DNA N6-adenine methylation
Nature Communications (2022)
-
Maternal Underweight and Obesity Are Associated with Placental Pathologies in Human Pregnancy
Reproductive Sciences (2022)
-
The Role of Epigenetic Changes in Ovarian Cancer: A Review
Indian Journal of Gynecologic Oncology (2021)