Abstract
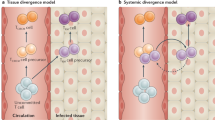
Dendritic cells (DCs) carry antigen from peripheral tissues via lymphatics to lymph nodes. We report here that differentiated DCs can also travel from the periphery into the blood. Circulating DCs migrated to the spleen, liver and lung but not lymph nodes. They also homed to the bone marrow, where they were retained better than in most other tissues. Homing of DCs to the bone marrow depended on constitutively expressed vascular cell adhesion molecule 1 and endothelial selectins in bone marrow microvessels. Two-photon intravital microscopy in bone marrow cavities showed that DCs formed stable antigen-dependent contacts with bone marrow–resident central memory T cells. Moreover, using this previously unknown migratory pathway, antigen-pulsed DCs were able to trigger central memory T cell–mediated recall responses in the bone marrow.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Banchereau, J. et al. Immunobiology of dendritic cells. Annu. Rev. Immunol. 18, 767–811 (2000).
Cavanagh, L.L. & von Andrian, U.H. Travellers in many guises: The origins and destinations of dendritic cells. Immunol. Cell Biol. 80, 448–462 (2002).
Sallusto, F. et al. Rapid and coordinated switch in chemokine receptor expression during dendritic cell maturation. Eur. J. Immunol. 28, 2760–2769 (1998).
Balazs, M., Martin, F., Zhou, T. & Kearney, J. Blood dendritic cells interact with splenic marginal zone B cells to initiate T-independent immune responses. Immunity 17, 341–352 (2002).
Randolph, G.J., Inaba, K., Robbiani, D.F., Steinman, R.M. & Muller, W.A. Differentiation of phagocytic monocytes into lymph node dendritic cells in vivo. Immunity 11, 753–761 (1999).
Legge, K.L. & Braciale, T.J. Accelerated migration of respiratory dendritic cells to the regional lymph nodes is limited to the early phase of pulmonary infection. Immunity 18, 265–277 (2003).
Mullins, D.W. et al. Route of immunization with peptide-pulsed dendritic cells controls the distribution of memory and effector T cells in lymphoid tissues and determines the pattern of regional tumor control. J. Exp. Med. 198, 1023–1034 (2003).
Schuler, G., Schuler-Thurner, B. & Steinman, R.M. The use of dendritic cells in cancer immunotherapy. Curr. Opin. Immunol. 15, 138–147 (2003).
Sallusto, F., Geginat, J. & Lanzavecchia, A. Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu. Rev. Immunol. 22, 745–763 (2004).
Di Rosa, F. & Santoni, A. Bone marrow CD8 T cells are in a different activation state than those in lymphoid periphery. Eur. J. Immunol. 32, 1873–1880 (2002).
Becker, T.C., Coley, S.M., Wherry, E.J. & Ahmed, R. Bone marrow is a preferred site for homeostatic proliferation of memory CD8 T cells. J. Immunol. 174, 1269–1273 (2005).
Mazo, I.B. et al. Bone marrow is a major reservoir and site of recruitment for central memory CD8+ T cells. Immunity 22, 259–270 (2005).
Feuerer, M. et al. Therapy of human tumors in NOD/SCID mice with patient-derived reactivated memory T cells from bone marrow. Nat. Med. 7, 452–458 (2001).
Maraskovsky, E. et al. Dramatic increase in the numbers of functionally mature dendritic cells in Flt3 ligand-treated mice: multiple dendritic cell subpopulations identified. J. Exp. Med. 184, 1953–1962 (1996).
Mora, J.R. et al. Selective imprinting of gut-homing T cells by Peyer's patch dendritic cells. Nature 424, 88–93 (2003).
von Andrian, U.H. & Mackay, C.R. T-cell function and migration. Two sides of the same coin. N. Engl. J. Med. 343, 1020–1034 (2000).
Shortman, K. & Liu, Y.J. Mouse and human dendritic cell subtypes. Nat. Rev. Immunol. 2, 151–161 (2002).
Donnenberg, V.S. et al. Rare-event analysis of circulating human dendritic cell subsets and their presumptive mouse counterparts. Transplantation 72, 1946–1951 (2001).
Wright, D.E., Wagers, A.J., Gulati, A.P., Johnson, F.L. & Weissman, I.L. Physiological migration of hematopoietic stem and progenitor cells. Science 294, 1933–1936 (2001).
Kamath, A.T. et al. The development, maturation, and turnover rate of mouse spleen dendritic cell populations. J. Immunol. 165, 6762–6770 (2000).
Mazo, I.B. et al. Hematopoietic progenitor cell rolling in bone marrow microvessels: Parallel contributions by endothelial selectins and VCAM-1. J. Exp. Med. 188, 465–474 (1998).
Manjunath, N. et al. Effector differentiation is not prerequisite for generation of memory cytotoxic T lymphocytes. J. Clin. Invest. 108, 871–878 (2001).
Weninger, W., Crowley, M.A., Manjunath, N. & von Andrian, U.H. Migratory properties of naive, effector, and memory CD8+ T cells. J. Exp. Med. 194, 953–966 (2001).
Robert, C. et al. Gene therapy to target dendritic cells from blood to lymph nodes. Gene Ther. 10, 1479–1486 (2003).
Sumen, C., Mempel, T.R., Mazo, I.B. & von Andrian, U.H. Intravital microscopy; visualizing immunity in context. Immunity 21, 315–329 (2004).
del Hoyo, G.M. et al. Characterization of a common precursor population for dendritic cells. Nature 415, 1043–1047 (2002).
O'Keeffe, M. et al. Dendritic cell precursor populations of mouse blood: identification of the murine homologues of human blood plasmacytoid pre-DC2 and CD11c+ DC1 precursors. Blood 101, 1453–1459 (2003).
Robert, C. et al. Interaction of dendritic cells with skin endothelium: A new perspective on immunosurveillance. J. Exp. Med. 189, 627–636 (1999).
Fossum, S. Lymph-borne dendritic leucocytes do not recirculate, but enter the lymph node paracortex to become interdigitating cells. Scand. J. Immunol. 27, 97–105 (1988).
Kupiec-Weglinski, J.W., Austyn, J.M. & Morris, P.J. Migration patterns of dendritic cells in the mouse. Traffic from the blood, and T cell-dependent and -independent entry to lymphoid tissues. J. Exp. Med. 167, 632–645 (1988).
Lappin, M.B. et al. Analysis of mouse dendritic cell migration in vivo upon subcutaneous and intravenous injection. Immunology 98, 181–188 (1999).
Frenette, P.S., Subbarao, S., Mazo, I.B., von Andrian, U.H. & Wagner, D.D. Endothelial selectins and vascular cell adhesion molecule-1 promote hematopoietic progenitor homing to bone marrow. Proc. Natl. Acad. Sci. USA 95, 14423–14428 (1998).
Di Rosa, F. & Pabst, R. The bone marrow: a nest for migratory memory T cells. Trends Immunol. 26, 360–366 (2005).
Wherry, E.J. et al. Lineage relationship and protective immunity of memory CD8 T cell subsets. Nat. Immunol. 4, 225–234 (2003).
Zammit, D.J., Cauley, L.S., Pham, Q.M. & Lefrancois, L. Dendritic cells maximize the memory CD8 T cell response to infection. Immunity 22, 561–570 (2005).
Mempel, T.R., Henrickson, S.E. & von Andrian, U.H. T cell priming by dendritic cells in lymph nodes occurs in three distinct phases. Nature 427, 154–159 (2004).
Gesner, B.M. & Gowans, J.L. The output of lymphocytes from the thoracic duct of unanaesthetized mice. Br. J. Exp. Path. 43, 424–430 (1962).
von Andrian, U.H. & Mempel, T.R. Homing and cellular traffic in lymph nodes. Nat. Rev. Immunol. 3, 867–878 (2003).
Randolph, G.J., Beaulieu, S., Lebecque, S., Steinman, R.M. & Muller, W.A. Differentiation of monocytes into dendritic cells in a model of transendothelial trafficking. Science 282, 480–483 (1998).
Enioutina, E.Y., Visic, D. & Daynes, R.A. The induction of systemic and mucosal immune responses to antigen-adjuvant compositions administered into the skin: alterations in the migratory properties of dendritic cells appears to be important for stimulating mucosal immunity. Vaccine 18, 2753–2767 (2000).
Racanelli, V., Behrens, S.E., Aliberti, J. & Rehermann, B. Dendritic cells transfected with cytopathic self-replicating RNA induce crosspriming of CD8+ T cells and antiviral immunity. Immunity 20, 47–58 (2004).
Larsen, C.P., Morris, P.J. & Austyn, J.M. Migration of dendritic leukocytes from cardiac allografts into host spleens. A novel pathway for initiation of rejection. J. Exp. Med. 171, 307–314 (1990).
Saiki, T., Ezaki, T., Ogawa, M. & Matsuno, K. Trafficking of host- and donor-derived dendritic cells in rat cardiac transplantation: allosensitization in the spleen and hepatic nodes. Transplantation 71, 1806–1815 (2001).
Vazquez-Torres, A. et al. Extraintestinal dissemination of Salmonella by CD18-expressing phagocytes. Nature 401, 804–808 (1999).
Pircher, H., Burki, K., Lang, R., Hengartner, H. & Zinkernagel, R.M. Tolerance induction in double specific T-cell receptor transgenic mice varies with antigen. Nature 342, 559–561 (1989).
Koni, P.A. et al. Conditional vascular cell adhesion molecule 1 deletion in mice. Impaired lymphocyte migration to bone marrow. J. Exp. Med. 193, 741–754 (2001).
Boggs, D.R. The total marrow mass of the mouse: a simplified method of measurement. Am. J. Hematol. 16, 277–286 (1984).
Boak, J.L. & Woodruff, M.F.A. Modified technique for collecting mouse thoracic duct lymph. Nature 205, 396–397 (1965).
Lyons, A.B. Analysing cell division in vivo and in vitro using flow cytometric measurement of CFSE dye dilution. J. Immunol. Methods 243, 147–154 (2000).
Mempel, T.R., Scimone, M.L., Mora, J.R. & von Andrian, U.H. In vivo imaging of leukocyte trafficking in blood vessels and tissues. Curr. Opin. Immunol. 16, 406–417 (2004).
Acknowledgements
We thank A. Vazquez-Torres (University of Colorado) for mutant strains of salmonella; G. Cheng and B. Reinhardt for technical support; and L. Scimone for help with intravital microscopy analysis. Supported by National Institutes of Health (AI061663, HL62524, HL54936 and HL56949 to U.H.v.A.); an Amy Potter fellowship, the Charles Hood Foundation and the Multiple Myeloma Foundation (I.B.M.); and Federazione Italiana Ricerca sul Cancro (R.B.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Video 1
Representative 3D time-lapse movie generated by 2-photon intravital microscopy in the skull of an anesthetized mouse. Hoechst 33342-labeled (blue) P14 TCM were injected into recipient mice and allowed to distribute in the body. 18h later, CMTMR-labeled (red) purified spleen DC were injected i.v. without peptide pulsing. Two hours thereafter, the animal was prepared for 2-photon intravital microscopy of skull BM as described in Materials and Methods. The luminal compartment of BM microvessels was delineated by i.v. injection of FITC-dextran (2MDa). 3D image stacks (23 optical sections, 5 µm vertical step size) were acquired every minute during a 30 min scan period. The total imaged volume (w x l x d) was 210 µm x 210 µm x 110 µm. (MOV 2880 kb)
Supplementary Video 2
Representative 3D time-lapse movie generated as described for movie 1, except that DC were pulsed with antigenic gp33 peptide prior to injection. Note that most TCM move at reduced velocity and undergo long-lasting tight contacts with DC. (MOV 2936 kb)
Rights and permissions
About this article
Cite this article
Cavanagh, L., Bonasio, R., Mazo, I. et al. Activation of bone marrow–resident memory T cells by circulating, antigen-bearing dendritic cells. Nat Immunol 6, 1029–1037 (2005). https://doi.org/10.1038/ni1249
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1249
This article is cited by
-
Minimally invasive longitudinal intravital imaging of cellular dynamics in intact long bone
Nature Protocols (2023)
-
Increased activated regulatory T cell subsets and aging Treg-like cells in multiple myeloma and monoclonal gammopathy of undetermined significance: a case control study
Cancer Cell International (2018)
-
In vivo longitudinal visualization of bone marrow engraftment process in mouse calvaria using two-photon microscopy
Scientific Reports (2017)
-
Mature dendritic cell derived from cryopreserved immature dendritic cell shows impaired homing ability and reduced anti-viral therapeutic effects
Scientific Reports (2016)
-
Immune responses in multiple myeloma: role of the natural immune surveillance and potential of immunotherapies
Cellular and Molecular Life Sciences (2016)