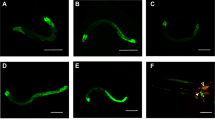
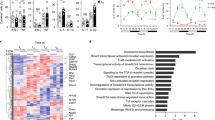
Abstract
Communication between the immune and nervous systems, each of which is able to react rapidly to environmental stimuli, may confer a survival advantage. However, precisely how the nervous system influences the immune response and whether neural modulation of immune function is biologically important are not well understood. Here we report that neuronal exocytosis of neuropeptides from dense core vesicles suppressed the survival of Caenorhabditis elegans and their clearance of infection with the human bacterial pathogen Pseudomonas aeruginosa. This immunomodulatory function was mediated by INS-7, an insulin-like neuropeptide whose induction was associated with Pseudomonas virulence. INS-7 secreted from the nervous system functioned in a non–cell autonomous way to activate the insulin pathway and alter basal and inducible expression of immunity-related genes in intestinal cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Brogden, K.A., Guthmiller, J.M., Salzet, M. & Zasloff, M. The nervous system and innate immunity: the neuropeptide connection. Nat. Immunol. 6, 558–564 (2005).
Sternberg, E.M. Neural regulation of innate immunity: a coordinated nonspecific host response to pathogens. Nat. Rev. Immunol. 6, 318–328 (2006).
Kin, N.W. & Sanders, V.M. It takes nerve to tell T and B cells what to do. J. Leukoc. Biol. 79, 1093–1104 (2006).
Nurrish, S.J. An overview of C. elegans trafficking mutants. Traffic 3, 2–10 (2002).
Gravato-Nobre, M.J. & Hodgkin, J. Caenorhabditis elegans as a model for innate immunity to pathogens. Cell. Microbiol. 7, 741–751 (2005).
Zhang, Y. Neuronal mechanisms of Caenorhabditis elegans and pathogenic bacteria interactions. Curr. Opin. Microbiol. 11, 257–261 (2008).
Liew, F.Y., Xu, D., Brint, E.K. & O'Neill, L.A.J. Negative regulation of Toll-like receptor-mediated immune responses. Nat. Rev. Immunol. 5, 446–458 (2005).
Kim, T. et al. Downregulation of lipopolysaccharide response in Drosophila by negative crosstalk between the AP1 and NF-κB signaling modules. Nat. Immunol. 6, 211–218 (2005).
Pujol, N. et al. A reverse genetic analysis of components of the Toll signaling pathway in Caenorhabditis elegans. Curr. Biol. 11, 809–821 (2001).
Shivers, R.P., Youngman, M.J. & Kim, D.H. Transcriptional responses to pathogens in Caenorhabditis elegans. Curr. Opin. Microbiol. 11, 251–256 (2008).
Shapira, M. et al. A conserved role for a GATA transcription factor in regulating epithelial innate immune responses. Proc. Natl. Acad. Sci. USA 103, 14086–14091 (2006).
Cai, T., Fukushige, T., Notkins, A.L. & Krause, M. Insulinoma-associated protein IA-2, a vesicle transmembrane protein, genetically interacts with UNC-31/CAPS and affects neurosecretion in Caenorhabditis elegans. J. Neurosci. 24, 3115–3124 (2004).
Speese, S. et al. UNC-31 (CAPS) is required for dense-core vesicle but not synaptic vesicle exocytosis in Caenorhabditis elegans. J. Neurosci. 27, 6150–6162 (2007).
Richmond, J. Synaptic function. WormBook doi:10.1895/wormbook.1.69.1 (30 Dec 2005).
Saifee, O., Wei, L. & Nonet, M.L. The Caenorhabditis elegans unc-64 locus encodes a syntaxin that interacts genetically with synaptobrevin. Mol. Biol. Cell 9, 1235–1252 (1998).
Charlie, N.K., Schade, M.A., Thomure, A.M. & Miller, K.G. Presynaptic UNC-31 (CAPS) is required to activate the Gαs pathway of the Caenorhabditis elegans synaptic signaling network. Genetics 172, 943–961 (2006).
Desai, C., Garriga, G., McIntire, S.L. & Horvitz, H.R. A genetic pathway for the development of the Caenorhabditis elegans HSN motor neurons. Nature 336, 638–646 (1988).
Zhang, Y., Lu, H. & Bargmann, C.I. Pathogenic bacteria induce aversive olfactory learning in Caenorhabditis elegans. Nature 438, 179–184 (2005).
Kass, J., Jacob, T.C., Kim, P. & Kaplan, J.M. The EGL-3 proprotein convertase regulates mechanosensory responses of Caenorhabditis elegans. J. Neurosci. 21, 9265–9272 (2001).
Jacob, T.C. & Kaplan, J.M. The EGL-21 carboxypeptidase E facilitates acetylcholine release at Caenorhabditis elegans neuromuscular junctions. J. Neurosci. 23, 2122–2130 (2003).
Tan, M.W., Mahajan-Miklos, S. & Ausubel, F.M. Killing of Caenorhabditis elegans by Pseudomonas aeruginosa used to model mammalian bacterial pathogenesis. Proc. Natl. Acad. Sci. USA 96, 715–720 (1999).
Nonet, M.L., Saifee, O., Zhao, H., Rand, J.B. & Wei, L. Synaptic transmission deficits in Caenorhabditis elegans synaptobrevin mutants. J. Neurosci. 18, 70–80 (1998).
Richmond, J.E., Weimer, R.M. & Jorgensen, E.M. An open form of syntaxin bypasses the requirement for UNC-13 in vesicle priming. Nature 412, 338–341 (2001).
Avery, L., Bargmann, C.I. & Horvitz, H.R. The Caenorhabditis elegans unc-31 gene affects multiple nervous system-controlled functions. Genetics 134, 455–464 (1993).
Banyai, L. & Patthy, L. Amoebapore homologs of Caenorhabditis elegans. Biochim. Biophys. Acta 1429, 259–264 (1998).
Kato, Y. et al. abf-1 and abf-2, ASABF-type antimicrobial peptide genes in Caenorhabditis elegans. Biochem. J. 361, 221–230 (2002).
Nurrish, S., Segalat, L. & Kaplan, J.M. Serotonin inhibition of synaptic transmission: Gα0 decreases the abundance of UNC-13 at release sites. Neuron 24, 231–242 (1999).
Lackner, M.R., Nurrish, S.J. & Kaplan, J.M. Facilitation of synaptic transmission by EGL-30 Gqα and EGL-8 PLCβ: DAG binding to UNC-13 is required to stimulate acetylcholine release. Neuron 24, 335–346 (1999).
Darby, C. & Falkow, S. Mimicry of a G protein mutation by pertussis toxin expression in transgenic Caenorhabditis elegans. Infect. Immun. 69, 6271–6275 (2001).
Garsin, D.A. et al. Long-lived C. elegans daf-2 mutants are resistant to bacterial pathogens. Science 300, 1921 (2003).
Pierce, S.B. et al. Regulation of DAF-2 receptor signaling by human insulin and ins-1, a member of the unusually large and diverse C. elegans insulin gene family. Genes Dev. 15, 672–686 (2001).
Apfeld, J. & Kenyon, C. Regulation of lifespan by sensory perception in Caenorhabditis elegans. Nature 402, 804–809 (1999).
Bargmann, C.I. & Horvitz, H.R. Control of larval development by chemosensory neurons in Caenorhabditis elegans. Science 251, 1243–1246 (1991).
Ailion, M., Inoue, T., Weaver, C.I., Holdcraft, R.W. & Thomas, J.H. Neurosecretory control of aging in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 96, 7394–7397 (1999).
Henderson, S.T. & Johnson, T.E. daf-16 integrates developmental and environmental inputs to mediate aging in the nematode Caenorhabditis elegans. Curr. Biol. 11, 1975–1980 (2001).
Hertweck, M., Gobel, C. & Baumeister, R. C. elegans SGK-1 is the critical component in the Akt/PKB kinase complex to control stress response and life span. Dev. Cell 6, 577–588 (2004).
Lin, K., Hsin, H., Libina, N. & Kenyon, C. Regulation of the Caenorhabditis elegans longevity protein DAF-16 by insulin/IGF-1 and germline signaling. Nat. Genet. 28, 139–145 (2001).
Murphy, C.T. et al. Genes that act downstream of DAF-16 to influence the lifespan of Caenorhabditis elegans. Nature 424, 277–283 (2003).
Hsu, A.L., Murphy, C.T. & Kenyon, C. Regulation of aging and age-related disease by DAF-16 and heat-shock factor. Science 300, 1142–1145 (2003).
Hsin, H. & Kenyon, C. Signals from the reproductive system regulate the lifespan of C. elegans. Nature 399, 362–366 (1999).
Murphy, C.T., Lee, S.-J. & Kenyon, C. Tissue entrainment by feedback regulation of insulin gene expression in the endoderm of Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 104, 19046–19050 (2007).
Alper, S., McBride, S.J., Lackford, B., Freedman, J.H. & Schwartz, D.A. Specificity and complexity of the Caenorhabditis elegans innate immune response. Mol. Cell. Biol. 27, 5544–5553 (2007).
Evans, E., Kawli, T. & Tan, M.-W. Pseudomonas aeruginosa suppresses host immunity by activating the DAF-2 insulin-like signaling pathway in C. elegans. PLoS Pathog. 10.1371/journal.ppat.1000175 (2008).
Morita, K., Chow, K.L. & Ueno, N. Regulation of body length and male tail ray pattern formation of Caenorhabditis elegans by a member of TGF-β family. Development 126, 1337–1347 (1999).
Glaser, R. & Kiecolt-Glaser, J.K. Stress-induced immune dysfunction: Implications for health. Nat. Rev. Immunol. 5, 243–251 (2005).
Liberati, N.T. et al. An ordered, nonredundant library of Pseudomonas aeruginosa strain PA14 transposon insertion mutants. Proc. Natl. Acad. Sci. USA 103, 2833–2838 (2006).
Timmons, L., Court, D.L. & Fire, A. Ingestion of bacterially expressed dsRNAs can produce specific and potent genetic interference in Caenorhabditis elegans. Gene 263, 103–112 (2001).
Shibata, Y., Branicky, R., Landaverde, I.O. & Hekimi, S. Redox regulation of germline and vulval development in Caenorhabditis elegans. Science 302, 1779–1782 (2003).
Espelt, M.V., Estevez, A.Y., Yin, X. & Strange, K. Oscillatory Ca2+ signaling in the isolated Caenorhabditis elegans intestine: role of the inositol-1,4,5-trisphosphate receptor and phospholipases C β and γ. J. Gen. Physiol. 126, 379–392 (2005).
Lamitina, S.T., Morrison, R., Moeckel, G.W. & Strange, K. Adaptation of the nematode Caenorhabditis elegans to extreme osmotic stress. Am. J. Physiol. Cell Physiol. 286, C785–C791 (2004).
Acknowledgements
We thank W. Chen and E. Evans for strain construction (ins-7(lf) 'rescue' under Pins-7 and Punc-119); A. Fire (Stanford University) for advice, vectors and use of the microinjection apparatus; S. Cohen for microscope use; K. Miller (Oklahoma Medical Research Foundation), K. Strange (Vanderbilt University), C. Darby (University of California, San Francisco), C. Kenyon (University of California, San Francisco), and K. Shen (Stanford University) for sharing strains and vectors; and all members of the Tan laboratory, A. Brunet and V. Nanjundiah for critical review and discussion of this work. Some nematode strains were provided by the Caenorhabditis Genetics Center (funded by the US National Institutes of Health National Center for Research Resources); the ins-7 mutant was provided by the Japanese National BioResource Project (funded by the Ministry of Education, Culture, Science, Sports and Technology, Japan). Supported by the US National Institutes of Health (GM66269 to M.-W.T.).
Author information
Authors and Affiliations
Contributions
T.K. did the experiments, and T.K. and M.-W.T. designed the experiments and wrote the paper.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–15 and Tables 1–8 (PDF 1717 kb)
Rights and permissions
About this article
Cite this article
Kawli, T., Tan, MW. Neuroendocrine signals modulate the innate immunity of Caenorhabditis elegans through insulin signaling. Nat Immunol 9, 1415–1424 (2008). https://doi.org/10.1038/ni.1672
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1672
This article is cited by
-
A novel N6-Deoxyadenine methyltransferase METL-9 modulates C. elegans immunity via dichotomous mechanisms
Cell Research (2023)
-
Identification and characterization of differentially expressed genes in Caenorhabditis elegans in response to pathogenic and nonpathogenic Stenotrophomonas maltophilia
BMC Microbiology (2020)
-
MALT-1 mediates IL-17 neural signaling to regulate C. elegans behavior, immunity and longevity
Nature Communications (2020)
-
Isocitrate dehydrogenase-mediated metabolic disorders disrupt active immunization against fungal pathogens in eusocial termites
Journal of Pest Science (2020)
-
NPR-9 regulates the innate immune response in Caenorhabditis elegans by antagonizing the activity of AIB interneurons
Cellular & Molecular Immunology (2018)