Abstract
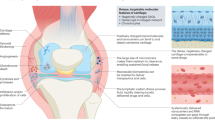
Rheumatoid arthritis (RA) is a chronic inflammatory disease characterized by systemic inflammation and joint destruction. Novel therapies have emerged during the past decade, marking a new era in the treatment of RA. Meanwhile, in vivo and in vitro gene-transfer studies have provided valuable insights into mechanisms of disease pathogenesis. Advanced gene-delivery techniques and animal models promise further progress in RA research and the development of novel therapeutic strategies for this disease. In this article we provide an overview of the wide spectrum of potential targets that have been identified so far, discuss currently available gene-transfer methods, and outline the barriers that need to be overcome for these approaches to be successfully applied in daily practice.
Key Points
-
Cytokines, activating factors of synovial cells, matrix-degrading enzymes and regulators of cell survival and apoptosis have been characterized as potential therapeutic targets in rheumatoid arthritis (RA)
-
Gene-transfer experiments in animal models have been useful for the identification of disease-relevant molecules
-
The severe combined immunodeficient mouse co-implantation model is currently the most informative model for the investigation of cytokine-independent processes in RA
-
The first clinical gene-transfer studies in RA patients have been conducted and the results are available
-
A variety of risks and technical hurdles need to be overcome before gene-transfer approaches can become established as therapies for RA
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Pierer M et al. (2003) The SCID mouse model: novel therapeutic targets—lessons from gene transfer. Springer Semin Immunopathol 25: 65–78
van de Loo FA et al. (2004) Gene therapy in animal models of rheumatoid arthritis: are we ready for the patients? Arthritis Res Ther 6: 183–196
Tarner IH et al. (2003) Treatment of autoimmune disease by adoptive cellular gene therapy. Ann NY Acad Sci 998: 512–519
Annenkov A and Chernajovsky Y (2000) Engineering mouse T lymphocytes specific to type II collagen by transduction with a chimeric receptor consisting of a single chain Fv and TCR zeta. Gene Ther 7: 714–722
Hacein-Bey-Abina S et al. (2003) LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1. Science 302: 415–419
Distler JH et al. (2005) Nucleofection: a new, highly efficient transfection method for primary human keratinocytes. Exp Dermatol 14: 315–320
Adriaansen J et al. (2006) A novel approach for gene therapy: engraftment of fibroblasts containing the artificial chromosome expression system at the site of inflammation. J Gene Med 8: 63–71
Basu J and Willard HF (2005) Artificial and engineered chromosomes: non-integrating vectors for gene therapy. Trends Mol Med 11: 251–258
Goossens PH et al. (2001) The influence of synovial fluid on adenovirus-mediated gene transfer to the synovial tissue. Arthritis Rheum 44: 48–52
Apparailly F et al. (2005) Adeno-associated virus pseudotype 5 vector improves gene transfer in arthritic joints. Hum Gene Ther 16: 426–434
Gould DJ et al. (2004) Inhibition of established collagen-induced arthritis with a tumour necrosis factor-α inhibitor expressed from a self-contained doxycycline regulated plasmid. Arthritis Res Ther 6: R103–R113
Ohashi S et al. (2002) Successful genetic transduction in vivo into synovium by means of electroporation. Biochem Biophys Res Commun 293: 1530–1535
Evans CH (2005) Gene therapy: what have we accomplished and where do we go from here? J Rheumatol Suppl 72: 17–20
Pan RY et al. (1999) Disease-inducible transgene expression from a recombinant adeno-associated virus vector in a rat arthritis model. J Virol 73: 3410–3417
Gouze E et al. (2003) Lentiviral-mediated gene delivery to synovium: potent intra-articular expression with amplification by inflammation. Mol Ther 7: 460–466
Corbel SY and Rossi FM (2002) Latest developments and in vivo use of the Tet system: ex vivo and in vivo delivery of tetracycline-regulated genes. Curr Opin Biotechnol 13: 448–452
Wieser C et al. (2005) Regulated and constitutive expression of anti-inflammatory cytokines by nontransforming herpesvirus saimiri vectors. Gene Ther 12: 395–406
Varley AW et al. (1997) A two-component expression system that responds to inflammatory stimuli in vivo. Nat Biotechnol 15: 1002–1006
Miagkov AV et al. (2002) Endogenous regulation of a therapeutic transgene restores homeostasis in arthritic joints. J Clin Invest 109: 1223–1229
Bakker AC et al. (2002) C3-Tat/HIV-regulated intraarticular human interleukin-1 receptor antagonist gene therapy results in efficient inhibition of collagen-induced arthritis superior to cytomegalovirus-regulated expression of the same transgene. Arthritis Rheum 46: 1661–1670
Pugliese A et al. (2005) A review of HIV-1 Tat protein biological effects. Cell Biochem Funct 23: 223–227
van de Loo FA et al. (2004) An inflammation-inducible adenoviral expression system for local treatment of the arthritic joint. Gene Ther 11: 581–590
Kim SH et al. (2002) Ex vivo gene delivery of IL-1Ra and soluble TNF receptor confers a distal synergistic therapeutic effect in antigen-induced arthritis. Mol Ther 6: 591–600
Müller-Ladner U et al. (1997) Human IL-1Ra gene transfer into human synovial fibroblasts is chondroprotective. J Immunol 158: 3492–3498
Ghivizzani SC et al. (1998) Direct adenovirus-mediated gene transfer of interleukin 1 and tumor necrosis factor α soluble receptors to rabbit knees with experimental arthritis has local and distal anti-arthritic effects. Proc Natl Acad Sci USA 95: 4613–4618
Evans CH et al. (2005) Gene transfer to human joints: progress toward a gene therapy of arthritis. Proc Natl Acad Sci USA 102: 8698–8703
Bresnihan B (2002) Anakinra as a new therapeutic option in rheumatoid arthritis: clinical results and perspectives. Clin Exp Rheumatol 20: S32–S34
Zhang HG et al. (2000) Adeno-associated virus production of soluble tumor necrosis factor receptor neutralizes tumor necrosis factor alpha and reduces arthritis. Hum Gene Ther 11: 2431–2442
Müller-Ladner U et al. (1999) Gene transfer of cytokine inhibitors into human synovial fibroblasts in the SCID mouse model. Arthritis Rheum 42: 490–497
Quattrocchi E et al. (1999) Paradoxical effects of adenovirus-mediated blockade of TNF activity in murine collagen-induced arthritis. J Immunol 163: 1000–1009
Bloquel C et al. (2004) Gene therapy of collagen-induced arthritis by electrotransfer of human tumor necrosis factor-α soluble receptor I variants. Hum Gene Ther 15: 189–201
Mease P et al. A phase 1 dose-escalation study of intra-articular administration of a recombinant adeno-associated vector containing the TNFR:Fc fusion gene, in inflammatory arthritis. 13th Annual Congress of the European Society of Gene Therapy 2005, Oct 29–Nov 1
Lubberts E et al. (2000) Intra-articular IL-10 gene transfer regulates the expression of collagen-induced arthritis (CIA) in the knee and ipsilateral paw. Clin Exp Immunol 120: 375–383
Neidhart M et al. (2004) Deficient expression of interleukin-10 receptor α-chain in rheumatoid arthritis synovium--limitation of inflammatory animal models. Arthritis Rheum 50: S112
Cottard V et al. (2000) Adeno-associated virus-mediated delivery of IL-4 prevents collagen-induced arthritis. Gene Ther 7: 1930–1939
Woods JM et al. (2001) IL-4 adenoviral gene therapy reduces inflammation, proinflammatory cytokines, vascularization, and bony destruction in rat adjuvant-induced arthritis. J Immunol 166: 1214–1222
Nabbe KC et al. (2005) Local IL-13 gene transfer prior to immune-complex arthritis inhibits chondrocyte death and matrix-metalloproteinase-mediated cartilage matrix degradation despite enhanced joint inflammation. Arthritis Res Ther 7: R392–R401
Koenders MI et al. (2005) Induction of cartilage damage by overexpression of T cell interleukin-17A in experimental arthritis in mice deficient in interleukin-1. Arthritis Rheum 52: 975–983
Joosten LA et al. (2004) Interleukin-18 promotes joint inflammation and induces interleukin-1-driven cartilage destruction. Am J Pathol 165: 959–967
Song XY et al. (1998) Plasmid DNA encoding transforming growth factor-β1 suppresses chronic disease in a streptococcal cell wall-induced arthritis model. J Clin Invest 101: 2615–2621
Tak PP et al. (2001) Inhibitor of nuclear factor κB kinase β is a key regulator of synovial inflammation. Arthritis Rheum 44: 1897–1907
Miagkov AV et al. (1998) NF-κB activation provides the potential link between inflammation and hyperplasia in the arthritic joint. Proc Natl Acad Sci USA 95: 13859–13864
Yamamoto A et al. (2003) Suppression of arthritic bone destruction by adenovirus-mediated dominant-negative Ras gene transfer to synoviocytes and osteoclasts. Arthritis Rheum 48: 2682–2692
Pap T et al. (2004) Cooperation of Ras- and c-Myc-dependent pathways in regulating the growth and invasiveness of synovial fibroblasts in rheumatoid arthritis. Arthritis Rheum 50: 2794–2802
Wakisaka S et al. (1998) Possible correction of abnormal rheumatoid arthritis synovial cell function by jun D transfection in vitro. Arthritis Rheum 41: 470–481
Krause A et al. (2002) Rheumatoid arthritis synoviocyte survival is dependent on Stat3. J Immunol 169: 6610–6616
Shouda T et al. (2001) Induction of the cytokine signal regulator SOCS3/CIS3 as a therapeutic strategy for treating inflammatory arthritis. J Clin Invest 108: 1781–1788
Sherr CJ and Roberts JM (1999) CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev 13: 1501–1512
Yao Q et al. (2001) Gene transfer of p53 to arthritic joints stimulates synovial apoptosis and inhibits inflammation. Mol Ther 3: 901–910
Nasu K et al. (2000) Adenoviral transfer of cyclin-dependent kinase inhibitor genes suppresses collagen-induced arthritis in mice. J Immunol 165: 7246–7252
Yao Q et al. (2003) Intra-articular adenoviral-mediated gene transfer of trail induces apoptosis of arthritic rabbit synovium. Gene Ther 10: 1055–1060
Yao Q et al. (2000) Adenoviral mediated delivery of FAS ligand to arthritic joints causes extensive apoptosis in the synovial lining. J Gene Med 2: 210–219
Kobayashi T et al. (2000) Novel gene therapy for rheumatoid arthritis by FADD gene transfer: induction of apoptosis of rheumatoid synoviocytes but not chondrocytes. Gene Ther 7: 527–533
Sant SM et al. (1998) Molecular lysis of synovial lining cells by in vivo herpes simplex virus-thymidine kinase gene transfer. Hum Gene Ther 9: 2735–2743
Goossens PH et al. (1999) Feasibility of adenovirus-mediated nonsurgical synovectomy in collagen-induced arthritis-affected rhesus monkeys. Hum Gene Ther 10: 1139–1149
Distler JH et al. (2003) Angiogenic and angiostatic factors in the molecular control of angiogenesis. Q J Nucl Med 47: 149–161
Afuwape AO et al. (2003) Adenoviral delivery of soluble VEGF receptor 1 (sFlt-1) abrogates disease activity in murine collagen-induced arthritis. Gene Ther 10: 1950–1960
Chen Y et al. (2005) Gene therapy targeting the Tie2 function ameliorates collagen-induced arthritis and protects against bone destruction. Arthritis Rheum 52: 1585–1594
Yin G et al. (2002) Endostatin gene transfer inhibits joint angiogenesis and pannus formation in inflammatory arthritis. Mol Ther 5: 547–554
Kim JM et al. (2002) Angiostatin gene transfer as an effective treatment strategy in murine collagen-induced arthritis. Arthritis Rheum 46: 793–801
Jou IM et al. (2005) Thrombospondin 1 as an effective gene therapeutic strategy in collagen-induced arthritis. Arthritis Rheum 52: 339–344
Rutkauskaite E et al. (2004) Ribozymes that inhibit the production of matrix metalloproteinase 1 reduce the invasiveness of rheumatoid arthritis synovial fibroblasts. Arthritis Rheum 50: 1448–1456
van der Laan WH et al. (2003) Cartilage degradation and invasion by rheumatoid synovial fibroblasts is inhibited by gene transfer of TIMP-1 and TIMP-3. Gene Ther 10: 234–242
Apparailly F et al. (2002) Adenovirus-mediated gene transfer of urokinase plasminogen inhibitor inhibits angiogenesis in experimental arthritis. Gene Ther 9: 192–200
van der Laan WH et al. (2000) Cartilage degradation and invasion by rheumatoid synovial fibroblasts is inhibited by gene transfer of a cell surface-targeted plasmin inhibitor. Arthritis Rheum 43: 1710–1718
Schedel J et al. (2004) Targeting cathepsin L (CL) by specific ribozymes decreases CL protein synthesis and cartilage destruction in rheumatoid arthritis. Gene Ther 11: 1040–1047
Saidenberg-Kermanac'h N et al. (2003) Efficacy of interleukin-10 gene electrotransfer into skeletal muscle in mice with collagen-induced arthritis. J Gene Med 5: 164–171
Acknowledgements
The work is partially funded by the Articulum Fellowship Program provided by Pfizer as well as by the Swiss National Science Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Moritz, F., Distler, O., Ospelt, C. et al. Technology Insight: gene transfer and the design of novel treatments for rheumatoid arthritis. Nat Rev Rheumatol 2, 153–162 (2006). https://doi.org/10.1038/ncprheum0117
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncprheum0117
This article is cited by
-
Development of a new humanized mouse model to study acute inflammatory arthritis
Journal of Translational Medicine (2012)