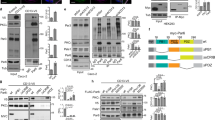
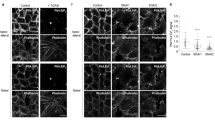
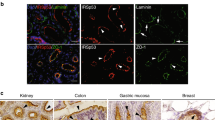
Abstract
Here we combined classical biochemistry with new biophysical approaches to study the organization of glycosylphosphatidylinositol (GPI)-anchored proteins (GPI-APs) with high spatial and temporal resolution at the plasma membrane of polarized epithelial cells. We show that in polarized MDCK cells, after sorting in the Golgi, each GPI-AP reaches the apical surface in homoclusters. Golgi-derived homoclusters are required for their subsequent plasma membrane organization into cholesterol-dependent heteroclusters. By contrast, in nonpolarized MDCK cells, GPI-APs are delivered to the surface as monomers in an unpolarized manner and are not able to form heteroclusters. We further demonstrate that this GPI-AP organization is regulated by the content of cholesterol in the Golgi apparatus and is required to maintain the functional state of the protein at the apical membrane. Thus, in contrast to fibroblasts, in polarized epithelial cells, a selective cholesterol-dependent sorting mechanism in the Golgi regulates both the organization and function of GPI-APs at the apical surface.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Sharma, P. et al. Nanoscale organization of multiple GPI-anchored proteins in living cell membranes. Cell 116, 577–589 (2004).
Goswami, D. et al. Nanoclusters of GPI-anchored proteins are formed by cortical actin-driven activity. Cell 135, 1085–1097 (2008).
Gowrishankar, K. et al. Active remodeling of cortical actin regulates spatiotemporal organization of cell surface molecules. Cell 149, 1353–1367 (2012).
Suzuki, K.G. et al. Transient GPI-anchored protein homodimers are units for raft organization and function. Nat. Chem. Biol. 8, 774–783 (2012).
Brameshuber, M. et al. Imaging of mobile long-lived nanoplatforms in the live cell plasma membrane. J. Biol. Chem. 285, 41765–41771 (2010).
Harder, T. Lipid raft domains and protein networks in T-cell receptor signal transduction. Curr. Opin. Immunol. 16, 353–359 (2004).
Fivaz, M. et al. Differential sorting and fate of endocytosed GPI-anchored proteins. EMBO J. 21, 3989–4000 (2002).
Schuck, S. & Simons, K. Polarized sorting in epithelial cells: raft clustering and the biogenesis of the apical membrane. J. Cell Sci. 117, 5955–5964 (2004).
Simons, K. & Gerl, M.J. Revitalizing membrane rafts: new tools and insights. Nat. Rev. Mol. Cell Biol. 11, 688–699 (2010).
Kusumi, A., Suzuki, K.G., Kasai, R.S., Ritchie, K. & Fujiwara, T.K. Hierarchical mesoscale domain organization of the plasma membrane. Trends Biochem. Sci. 36, 604–615 (2011).
Paladino, S. et al. Protein oligomerization modulates raft partitioning and apical sorting of GPI-anchored proteins. J. Cell Biol. 167, 699–709 (2004).
Paladino, S., Sarnataro, D., Tivodar, S. & Zurzolo, C. Oligomerization is a specific requirement for apical sorting of glycosyl-phosphatidylinositol–anchored proteins but not for non–raft-associated apical proteins. Traffic 8, 251–258 (2007).
Hannan, L.A., Lisanti, M.P., Rodriguez-Boulan, E. & Edidin, M. Correctly sorted molecules of a GPI-anchored protein are clustered and immobile when they arrive at the apical surface of MDCK cells. J. Cell Biol. 120, 353–358 (1993).
Friedrichson, T. & Kurzchalia, T.V. Microdomains of GPI-anchored proteins in living cells revealed by crosslinking. Nature 394, 802–805 (1998).
Varma, R. & Mayor, S. GPI-anchored proteins are organized in submicron domains at the cell surface. Nature 394, 798–801 (1998).
Digman, M.A., Dalal, R., Horwitz, A.F. & Gratton, E. Mapping the number of molecules and brightness in the laser scanning microscope. Biophys. J. 94, 2320–2332 (2008).
Catino, M.A., Paladino, S., Tivodar, S., Pocard, T. & Zurzolo, C. N- and O-glycans are not directly involved in the oligomerization and apical sorting of GPI proteins. Traffic 9, 2141–2150 (2008).
Breuza, L., Garcia, M., Delgrossi, M.H. & Le Bivic, A. Role of the membrane-proximal O-glycosylation site in sorting of the human receptor for neurotrophins to the apical membrane of MDCK cells. Exp. Cell Res. 273, 178–186 (2002).
Keller, P. & Simons, K. Cholesterol is required for surface transport of influenza virus hemagglutinin. J. Cell Biol. 140, 1357–1367 (1998).
Ilangumaran, S. & Hoessli, D.C. Effects of cholesterol depletion by cyclodextrin on the sphingolipid microdomains of the plasma membrane. Biochem. J. 335, 433–440 (1998).
Ridgway, N.D. Interactions between metabolism and intracellular distribution of cholesterol and sphingomyelin. Biochim. Biophys. Acta 1484, 129–141 (2000).
Ohvo-Rekilä, H., Ramstedt, B., Leppimaki, P. & Slotte, J.P. Cholesterol interactions with phospholipids in membranes. Prog. Lipid Res. 41, 66–97 (2002).
Padilla-Parra, S., Auduge, N., Coppey-Moisan, M. & Tramier, M. Quantitative FRET analysis by fast acquisition time domain FLIM at high spatial resolution in living cells. Biophys. J. 95, 2976–2988 (2008).
Padilla-Parra, S. et al. Quantitative comparison of different fluorescent protein couples for fast FRET-FLIM acquisition. Biophys. J. 97, 2368–2376 (2009).
Paladino, S., Pocard, T., Catino, M.A. & Zurzolo, C. GPI-anchored proteins are directly targeted to the apical surface in fully polarized MDCK cells. J. Cell Biol. 172, 1023–1034 (2006).
Paladino, S. et al. Different GPI-attachment signals affect the oligomerisation of GPI-anchored proteins and their apical sorting. J. Cell Sci. 121, 4001–4007 (2008).
Lebreton, S., Paladino, S. & Zurzolo, C. Selective roles for cholesterol and actin in compartmentalization of different proteins in the Golgi and plasma membrane of polarized cells. J. Biol. Chem. 283, 29545–29553 (2008).
Imjeti, N.S. et al. N-glycosylation instead of cholesterol mediates oligomerization and apical sorting of GPI-APs in FRT cells. Mol. Biol. Cell 22, 4621–4634 (2011).
Cunningham, O. et al. Dimerization controls the lipid raft partitioning of uPAR/CD87 and regulates its biological functions. EMBO J. 22, 5994–6003 (2003).
Brown, D.A., Crise, B. & Rose, J.K. Mechanism of membrane anchoring affects polarized expression of two proteins in MDCK cells. Science 245, 1499–1501 (1989).
Lisanti, M.P., Caras, I.W., Davitz, M.A. & Rodriguez-Boulan, E. A glycophospholipid membrane anchor acts as an apical targeting signal in polarized epithelial cells. J. Cell Biol. 109, 2145–2156 (1989).
Sengupta, P. et al. Probing protein heterogeneity in the plasma membrane using PALM and pair correlation analysis. Nat. Methods 8, 969–975 (2011).
Meder, D., Moreno, M.J., Verkade, P., Vaz, W.L. & Simons, K. Phase coexistence and connectivity in the apical membrane of polarized epithelial cells. Proc. Natl. Acad. Sci. USA 103, 329–334 (2006).
Kenworthy, A.K., Petranova, N. & Edidin, M. High-resolution FRET microscopy of cholera toxin B-subunit and GPI-anchored proteins in cell plasma membranes. Mol. Biol. Cell 11, 1645–1655 (2000).
Lippincott-Schwartz, J., Snapp, E. & Kenworthy, A. Studying protein dynamics in living cells. Nat. Rev. Mol. Cell Biol. 2, 444–456 (2001).
Ali, M.H. & Imperiali, B. Protein oligomerization: how and why. Bioorg. Med. Chem. 13, 5013–5020 (2005).
Simons, K. & Toomre, D. Lipid rafts and signal transduction. Nat. Rev. Mol. Cell Biol. 1, 31–39 (2000).
Sabharanjak, S., Sharma, P., Parton, R.G. & Mayor, S. GPI-anchored proteins are delivered to recycling endosomes via a distinct cdc42-regulated, clathrin-independent pinocytic pathway. Dev. Cell 2, 411–423 (2002).
Kirkham, M. & Parton, R.G. Clathrin-independent endocytosis: new insights into caveolae and non-caveolar lipid raft carriers. Biochim. Biophys. Acta 1746, 349–363 (2005).
Johannes, L. & Mayor, S. Induced domain formation in endocytic invagination, lipid sorting, and scission. Cell 142, 507–510 (2010).
Datta, A., Bryant, D.M. & Mostov, K.E. Molecular regulation of lumen morphogenesis. Curr. Biol. 21, R126–R136 (2011).
Tanos, B. & Rodriguez-Boulan, E. The epithelial polarity program: machineries involved and their hijacking by cancer. Oncogene 27, 6939–6957 (2008).
Zacharias, D.A., Violin, J.D., Newton, A.C. & Tsien, R.Y. Partitioning of lipid-modified monomeric GFPs into membrane microdomains of live cells. Science 296, 913–916 (2002).
Hellriegel, C., Caiolfa, V.R., Corti, V., Sidenius, N. & Zamai, M. Number and brightness image analysis reveals ATF-induced dimerization kinetics of uPAR in the cell membrane. FASEB J. 25, 2883–2897 (2011).
Bridgman, P.C. & Nakajima, Y. Membrane lipid heterogeneity associated with acetylcholine receptor particle aggregates in Xenopus embryonic muscle cells. Proc. Natl. Acad. Sci. USA 78, 1278–1282 (1981).
Dalal, R.B., Digman, M.A., Horwitz, A.F., Vetri, V. & Gratton, E. Determination of particle number and brightness using a laser scanning confocal microscope operating in the analog mode. Microsc. Res. Tech. 71, 69–81 (2008).
Malengo, G. et al. Fluorescence correlation spectroscopy and photon counting histogram on membrane proteins: functional dynamics of the glycosylphosphatidylinositol-anchored urokinase plasminogen activator receptor. J. Biomed. Opt. 13, 031215 (2008).
Seber, G.A.F. Multivariate Observations (J. Wiley & Sons, New York, USA, 1984).
Hillesheim, L.N., Chen, Y. & Muller, J.D. Dual-color photon counting histogram analysis of mRFP1 and EGFP in living cells. Biophys. J. 91, 4273–4284 (2006).
Tivodar, S. et al. Analysis of detergent-resistant membranes associated with apical and basolateral GPI-anchored proteins in polarized epithelial cells. FEBS Lett. 580, 5705–5712 (2006).
Mezghrani, A. et al. Manipulation of oxidative protein folding and PDI redox state in mammalian cells. EMBO J. 20, 6288–6296 (2001).
Manders, E.M.M., Verbeek, F.J. & Aten, J.A. Measurement of co-localization of objects in dualcolor confocal images. J. Microsc. 169, 375–382 (1993).
Acknowledgements
We thank P. Casanova (Institut Pasteur) for his technical support, N. Auduge (Institut Jacques Monod, ImagoSeine) for his help in FLIM analysis, P. Riccio (University of Naples Federico II) for her help in statistical analysis and the imaging facility at Pasteur Institute (PFID) and at CEINGE Institute (DIM). We thank A. Le Bivic (Institut de Biologie du Développement de Marseille-Luminy (IBDML), Marseille, France), S. Mayor (National Centre for Biological Sciences (NCBS), Bangalore, India) and N. Sidenius (Fondazione Italiana Ricerca sul Cancro Institute of Molecular Oncology (IFOM), Milan, Italy) for cDNA encoding p75-GFP, CHO cells expressing GFP-FR, and cDNAs encoding mGFP-GPI and mGFP-mGFP GPI (uPAR), respectively. This work was supported by the Agence Nationale de la Recherche (ANR) (05-BLAN 296-01 and ANR-09-BLAN-0122) and the European Union FP7 (Priority, grant 222887) to C.Z.; an Associazione Italiana per la Ricerca sul Cancro (AIRC) grant (MFAG 2007-2009) to S.P.; ANR PFTV2007 and Fondation pour la Recherche Médicale (FRM) 'Grands Equipements' to M.C.-M.; and grants NIH-P41 P41-RRO3155 and NIH-P50-GM076516 from the US National Institutes of Health to E.G.
Author information
Authors and Affiliations
Contributions
C.Z. conceived the project. C.Z., S.P. and S.L. designed experiments. S.P. planned and performed N&B and biochemical experiments, and S.L. planned and performed FLIM experiments. S.T. planned and performed biochemical experiments. E.G. developed the N&B technique, E.G. and G.O. helped with N&B data analysis, and M.T. and M.C.-M. developed the FLIM technique and helped with data analysis. F.F. performed the mathematical analysis of N&B data. S.P., S.L. and C.Z. wrote the manuscript. All authors discussed the results and manuscript text.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results and Supplementary Figures 1–9. (PDF 10083 kb)
Rights and permissions
About this article
Cite this article
Paladino, S., Lebreton, S., Tivodar, S. et al. Golgi sorting regulates organization and activity of GPI proteins at apical membranes. Nat Chem Biol 10, 350–357 (2014). https://doi.org/10.1038/nchembio.1495
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1495
This article is cited by
-
GM130 regulates pulmonary surfactant protein secretion in alveolar type II cells
Science China Life Sciences (2022)