Abstract
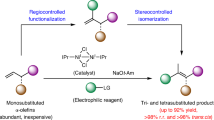
Olefin metathesis has had a large impact on modern organic chemistry, but important shortcomings remain: for example, the lack of efficient processes that can be used to generate acyclic alkenyl halides. Halo-substituted ruthenium carbene complexes decompose rapidly or deliver low activity and/or minimal stereoselectivity, and our understanding of the corresponding high-oxidation-state systems is limited. Here we show that previously unknown halo-substituted molybdenum alkylidene species are exceptionally reactive and are able to participate in high-yielding olefin metathesis reactions that afford acyclic 1,2-disubstituted Z-alkenyl halides. Transformations are promoted by small amounts of a catalyst that is generated in situ and used with unpurified, commercially available and easy-to-handle liquid 1,2-dihaloethene reagents, and proceed to high conversion at ambient temperature within four hours. We obtain many alkenyl chlorides, bromides and fluorides in up to 91 per cent yield and complete Z selectivity. This method can be used to synthesize biologically active compounds readily and to perform site- and stereoselective fluorination of complex organic molecules.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Nunnery, J. K. et al. Biosynthetically intriguing chlorinated lipophilic metabolites from geographically distant tropical marine cyanobacteria. J. Org. Chem. 77, 4198–4208 (2012)
Akiyama, T. et al. Stimulators of adipogenesis from the marine sponge Xestospongia testudinaria . Tetrahedron 69, 6560–6564 (2013)
Johansson Seechurn, C. C. C., Kitching, M. O., Colacot, T. J. & Sniekus, V. Palladium-catalyzed cross-coupling: a historical contextual perspective to the 2010 Nobel Prize. Angew. Chem. Int. Ed. 51, 5062–5085 (2012)
Gillis, E. P., Eastman, K. J., Hill, M. D., Donnelly, D. J. & Meanwell, N. A. Applications of fluorine in medicinal chemistry. J. Med. Chem. 58, 8315–8359 (2015)
Fujiwara, T. & O’Hagan, D. Successful fluorine-containing herbicide agrochemicals. J. Fluor. Chem. 167, 16–29 (2014)
Berger, R., Resnati, G., Metrangolo, P., Weber, E. & Hulliger, J. Organic fluorine compounds: a great opportunity for enhanced materials properties. Chem. Soc. Rev. 40, 3496–3508 (2011)
Kolb, M., Barth, J., Heydt, J.-G. & Jung, M. J. Synthesis and evaluation of mono-, di-, and trifluoroethenyl-GABA derivatives as GABA-T inhibitors. J. Med. Chem. 30, 267–272 (1987)
Silverman, R. B., Bichler, K. A. & Leon, A. J. Unusual mechanistic difference in the activation of γ-aminobutyric acid aminotransferase by (E)- and (Z)-4-amino-6-fluoro-5-hexenoic acid. J. Am. Chem. Soc. 118, 1253–1261 (1996)
Rosen, T. C., Yoshida, S., Kirk, K. L. & Haufe, G. Fluorinated phenylcyclopropylamines as inhibitors of monoamine oxidases. ChemBioChem 5, 1033–1043 (2004)
Morrill, C. & Grubbs, R. H. Synthesis of functionalized vinylboronates via ruthenium-catalyzed cross-metathesis and subsequent conversion to vinyl halides. J. Org. Chem. 68, 6031–6034 (2003)
Pawlu, P., Hreczycho, G., Szudkowska, J., Kubicki, M. & Marciniec, B. New one-pot synthesis of (E)-β-aryl vinyl halides from styrenes. Org. Lett. 11, 3390–3393 (2009)
Bull, J. A., Mousseau, J. J. & Charette, A. B. Convenient one-pot synthesis of (E)-β-aryl vinyl halides from benzyl bromides and dihalomethanes. Org. Lett. 10, 5485–5488 (2008)
Gao, F. & Hoveyda, A. H. α-Selective Ni-catalyzed hydroalumination of aryl- and alkyl-substituted terminal alkynes: practical syntheses of internal vinyl aluminums, halides, or boronates. J. Am. Chem. Soc. 132, 10961–10963 (2010)
Stork, G. & Zhao, K. A stereoselective synthesis of (Z)-1-iodo-1-alkenes. Tetrahedr. Lett . 30, 2173–2174 (1989)
Zhang, X.-P. & Schlosser, M. Highly cis-selective Wittig reactions employing α-heterosubstituted ylids. Tetrahedr. Lett . 34, 1925–1928 (1993)
Crane, E. A., Zabawa, T. P., Farmer, R. L. & Scheidt, K. A. Enantioselective synthesis of (–)-exiguolide by iterative stereoselective dioxinone-directed Prins cyclizations. Angew. Chem. Int. Ed. 50, 9112–9115 (2011)
Landelle, G., Turcotte-Savard, M.-C., Angers, L. & Paquin, J. F. Stereoselective synthesis of both stereoisomers of β-fluorostyrene derivatives from a common intermediate. Org. Lett. 13, 1568–1571 (2011)
Nakagawa, M., Saito, A., Soga, A., Yamamoto, N. & Taguchi, T. Chromium mediated stereoselective synthesis of (Z)-1-fluoro-2-alkenyl alkyl and trialkylsilyl ethers from dibromofluoromethylcarbinyl ethers. Tetrahedr. Lett . 46, 5257–5261 (2005)
Ohmura, T., Yamamoto, Y. & Miyaura, N. Rhodium- or iridium-catalyzed trans-hydroboration of terminal alkynes, giving (Z)-1-alkenylboron compounds. J. Am. Chem. Soc. 122, 4990–4991 (2000)
Molander, G. A. & Ellis, N. M. Highly stereoselective synthesis of cis-alkenyl pinacolatoboronates and potassium cis-alkenyltrifluoroborates via a hydroboration/protodeboronation approach. J. Org. Chem. 73, 6841–6844 (2008)
Kiesewetter, E. T. et al. Synthesis of Z-(pinacolato)allylboron and Z-(pinacolato)alkenylboron compounds through stereoselective catalytic cross-metathesis. J. Am. Chem. Soc. 135, 6026–6029 (2013)
Bronner, S. M., Herbert, M. B., Patel, P. R., Marx, V. M. & Grubbs, R. H. Ruthenium-catalyzed Z-selective metathesis catalysts with modified cyclometalated carbene ligands. Chem. Sci. 5, 4091–4098 (2014)
Brown, H. C., Larock, R. C., Gupta, S. K., Rajagopalan, S. & Bhat, N. G. Vinylic organoboranes. 15. Mercuration of 2-alkenyl-1,3,2-benzodioxaboroles and boronic acids. A convenient stereospecific procedure for the conversion of alkynes into (E)-1-halo-1-alkenes via mercuric salts. J. Org. Chem. 54, 6079–6084 (1989)
Petasis, N. A. & Zavialov, I. A. Mild conversion of alkenyl boronic acids to alkenyl halides with halosuccinimides. Tetrahedr. Lett . 37, 567–570 (1996)
Gensch, K. H., Pitman, I. H. & Higuchi, T. Oxidation of thioesters to sulfoxides by iodine. II. Catalytic role of some carboxylic acid anions. J. Am. Chem. Soc. 90, 2096–2104 (1968)
Hamri, S., Rodríguez, J., Basset, J., Guillaumet, G. & Pujol, M. D. A convenient iodination of indoles and derivatives. Tetrahedron 68, 6269–6275 (2012)
Speed, A. W. H., Mann, S. J., O’Brien, R. V., Schrock, R. R. & Hoveyda, A. H. Catalytic Z-selective cross-metathesis in complex molecule synthesis: a convergent stereoselective route to disorazole C1 . J. Am. Chem. Soc. 136, 16136–16139 (2014)
Hanh, T. T. H., Hang, D. T. T., Minh, C. V. & Dat, N. T. Anti-inflammatory effects of fatty acids isolated from Chromolaena odorata . Asian Pac. J. Trop. Med. 4, 760–763 (2011)
López, S., Fernández-Trillo, F., Midón, P., Castedo, L. & Saá, C. First stereoselective syntheses of (–)-siphonodiol and (–)-tetrahydrosiphonodiol, bioactive polyacetylenes from marine sponges. J. Org. Chem. 70, 6346–6352 (2005)
Campbell, M. G. & Ritter, T. Late-stage fluorination: from fundamentals to application. Org. Process Res. Dev. 18, 474–480 (2014)
Imaizumi, Y. et al. Molecular basis of pimarane compounds as novel activators of large-conductance Ca2+-activated channel α-subunit. Mol. Pharmacol. 62, 836–846 (2002)
Macnaughtan, M. L., Johnson, M. J. A. & Kampf, J. W. Olefin metathesis reactions with vinyl halides: formation, observation, interception, and fate of the ruthenium–monohalomethylidene moiety. J. Am. Chem. Soc. 129, 7708–7709 (2007)
Sashuk, V., Samojlowicz, C., Szadkowska, A. & Grela, K. Olefin cross-metathesis with vinyl halides. Chem. Commun. 2468–2470 (2008)
Macnaughtan, M. L., Gary, J. B., Gerlach, D. L., Johnson, M. J. A. & Kamf, J. W. Cross-metathesis of vinyl halides. Scope and limitations of ruthenium-based catalysts. Organometallics 28, 2880–2887 (2009)
Meek, S. J., O’Brien, R. V., Llaveria, J., Schrock, R. R. & Hoveyda, A. H. Catalytic Z-selective olefin metathesis for natural product synthesis. Nature 471, 461–466 (2011)
Vasiliu, M., Li, S., Arduengo, A. J. III & Dixon, D. A. Bond energies in models of the Schrock metathesis catalyst. J. Phys. Chem. C 115, 12106–12120 (2011)
Rosebrugh, L. E., Herbert, M. B., Marx, V. M., Keitz, B. K. & Grubbs, R. H. Highly active ruthenium metathesis catalysts exhibiting unprecedented activity and Z selectivity. J. Am. Chem. Soc. 135, 1276–1279 (2013)
Koh, M. J. et al. High-value alcohols and higher-oxidation-state compounds by catalytic Z-selective cross-metathesis. Nature 517, 181–186 (2015)
Silverio, D. L. et al. Simple organic molecules as catalysts for enantioselective synthesis of amines and alcohols. Nature 494, 216–221 (2013)
Hoye, R. C., Andersen, G. L., Brown, S. G. & Schultz, E. E. Total synthesis of clathculins A and B. J. Org. Chem. 75, 7400–7403 (2010)
Jang, H., Zhugralin, A. R., Lee, Y. & Hoveyda, A. H. Highly selective methods for synthesis of internal (α-) vinylboronates through efficient NHC–Cu-catalyzed hydroboration of terminal alkynes. Utility in synthesis and mechanistic basis for selectivity. J. Am. Chem. Soc. 133, 7859–7871 (2011)
Hoveyda, A. H. Evolution of catalytic enantioselective olefin metathesis: from ancillary transformation to purveyor of stereochemical identity. J. Org. Chem. 79, 4763–4792 (2014)
Fustero, S., Simón-Fuentes, A., Barrio, P. & Haufe, G. Olefin metathesis reactions with fluorinated substrates, catalysts, and solvents. Chem. Rev. 115, 871–930 (2015)
Zhang, H., Yu, E. C., Torker, S., Schrock, R. R. & Hoveyda, A. H. Preparation of macrocyclic Z-enoates and (E,Z)- or (Z,E)-dienoates through catalytic stereoselective ring-closing metathesis. J. Am. Chem. Soc. 136, 16493–16496 (2014)
Gillingham, D. G. & Hoveyda, A. H. Chiral N-heterocyclic carbenes in natural product synthesis: application of Ru-catalyzed asymmetric ring-opening/cross-metathesis and Cu-catalyzed allylic alkylation to total synthesis of baconipyrone C. Angew. Chem. Int. Ed. 46, 3860–3864 (2007)
Addington, D. E. et al. Impact of second-generation anti-psychotics and perphenazine on depressive symptoms in a randomized trial for chronic schizophrenia. J. Clin. Psychiatry 72, 75–80 (2011)
English, A. R., Girard, D., Jasys, V. J., Martingano, R. J. & Kellogg, M. S. Orally effective acid prodrugs of the β-lactamase inhibitor sulbactam. J. Med. Chem. 33, 344–347 (1990)
Furuya, T. & Ritter, T. Fluorination of boronic acids mediated by silver(I) triflate. Org. Lett. 11, 2860–2863 (2009)
Haginaka, J., Yasuda, H., Uno, T. & Nakagawa, T. Alkaline degradation and determination by high-performance liquid chromatography. Chem. Pharm. Bull. 32, 2752–2758 (1984)
Townsend, E. M. et al. High oxidation state molybdenum imido heteroatom-substituted alkyliene complexes. Organometallics 32, 4612–4617 (2013)
Acknowledgements
This research was supported by the United States National Institutes of Health, Institute of General Medical Sciences (GM-59426 and in part GM-57212).
Author information
Authors and Affiliations
Contributions
M.J.K., T.T.N. and H.Z. were involved in the discovery, design and development of the new Z-selective cross-metathesis strategies and their applications. A.H.H., M.J.K., T.T.N. and H.Z. conceived the research programme. A.H.H. designed and directed the investigations. A.H.H. and R.R.S. conceived the studies that led to the development of Mo MAP complexes. A.H.H. wrote the manuscript with revisions provided by M.J.K., T.T.N. and H.Z.
Corresponding author
Ethics declarations
Competing interests
The catalysts and technologies developed are licensed by a company that was founded by A.H.H. and R.R.S.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data (see Contents for more details). (PDF 10349 kb)
Rights and permissions
About this article
Cite this article
Koh, M., Nguyen, T., Zhang, H. et al. Direct synthesis of Z-alkenyl halides through catalytic cross-metathesis. Nature 531, 459–465 (2016). https://doi.org/10.1038/nature17396
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature17396
This article is cited by
-
Iron-catalysed reductive cross-coupling of glycosyl radicals for the stereoselective synthesis of C-glycosides
Nature Synthesis (2022)
-
Stereodefined alkenes with a fluoro-chloro terminus as a uniquely enabling compound class
Nature Chemistry (2022)
-
Cobalt-catalysed enantioselective C(sp3)–C(sp3) coupling
Nature Catalysis (2021)
-
Air-stable 18-electron adducts of Schrock catalysts with tuned stability constants for spontaneous release of the active species
Communications Chemistry (2021)
-
Palladium Nanoparticle-Bentonite Hybrid Using Leaves of Syzygium aqueum Plant from India: Design and Assessment in the Catalysis of –C–C– Coupling Reaction
Chemistry Africa (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.