Abstract
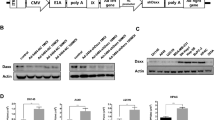
Arming oncolytic adenoviral vectors with anticancer transgenes that can be expressed in a tumor-selective manner may enable the engineering of vectors with increased potency, while retaining their safety profile. Armed oncolytic adenoviral vectors were constructed in which transgene expression has been linked via modified splice acceptor sequences that did not necessitate the deletion of any part of the adenoviral genome. Several oncolytic adenoviral vectors were compared in which the transgene was inserted in place of either the E3 or the L3 region. While all vectors had similar viral growth and cytotoxicity characteristics, the highest level of transgene expression was observed from a vector in which the transgene had been inserted downstream of the L3 23K protease gene, the Ad-23K-GM vector. Notably, no transgene expression occurred with this vector in the absence of DNA replication either in vitro or in vivo. In contrast, viruses in which the transgene was inserted into E3 locations exhibited a low level of transgene expression even in the absence of DNA replication. In summary, by utilizing the L3 region for arming oncolytic viruses, higher levels of tumor-specific transgene expression can be obtained without the need to delete any parts of the viral genome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ko D, Hawkins L, Yu DC . Development of transcriptionally regulated oncolytic adenoviruses. Oncogene 2005; 24: 7763–7774.
Biederer C, Ries S, Brandts CH, McCormick F . Replication-selective viruses for cancer therapy. J Mol Med 2002; 80: 163–175.
Alemany R, Balague C, Curiel DT . Replicative adenoviruses for cancer therapy. Nat Biotechnol 2000; 18: 723–727.
Jakubczak JL, Ryan P, Gorziglia M, Clarke L, Hawkins LK, Hay C et al. An oncolytic adenovirus selective for retinoblastoma tumor suppressor protein pathway-defective tumors: dependence on E1A, the E2F-1 promoter, and viral replication for selectivity and efficacy. Cancer Res 2003; 63: 1490–1499.
Johnson L, Shen A, Boyle L, Kunich J, Pandey K, Lemmon M et al. Selectively replicating adenoviruses targeting deregulated E2F activity are potent, systemic antitumor agents. Cancer Cell 2002; 1: 325–337.
Hermiston T . A demand for next-generation oncolytic adenoviruses. Curr Opin Mol Ther 2006; 8: 322–330.
McCormick F . Cancer-specific viruses and the development of ONYX-015. Cancer Biol Ther 2003; 2: S157–S160.
Parato KA, Senger D, Forsyth PA, Bell JC . Recent progress in the battle between oncolytic viruses and tumours. Nat Rev Cancer 2005; 5: 965–976.
Hermiston TW, Kuhn I . Armed therapeutic viruses: strategies and challenges to arming oncolytic viruses with therapeutic genes. Cancer Gene Ther 2002; 9: 1022–1035.
Haviv YS, Takayama K, Glasgow JN, Blackwell JL, Wang M, Lei X et al. A model system for the design of armed replicating adenoviruses using p53 as a candidate transgene. Mol Cancer Ther 2002; 1: 321–328.
Bristol JA, Zhu M, Ji H, Mina M, Xie Y, Clarke L et al. In vitro and in vivo activities of an oncolytic adenoviral vector designed to express GM-CSF. Mol Ther 2003; 7: 755–764.
Ramesh N, Ge Y, Ennist DL, Zhu M, Mina M, Ganesh S et al. CG0070, a conditionally replicating granulocyte macrophage colony-stimulating factor-armed oncolytic adenovirus for the treatment of bladder cancer. Clin Cancer Res 2006; 12: 305–313.
Zhao L, Gu J, Dong A, Zhang Y, Zhong L, He L et al. Potent antitumor activity of oncolytic adenovirus expressing mda-7/IL-24 for colorectal cancer. Hum Gene Ther 2005; 16: 845–858.
Freytag SO, Rogulski KR, Paielli DL, Gilbert JD, Kim JH . A novel three-pronged approach to kill cancer cells selectively: concomitant viral, double suicide gene, and radiotherapy. Hum Gene Ther 1998; 9: 1323–1333.
Zhan J, Gao Y, Wang W, Shen A, Aspelund A, Young M et al. Tumor-specific intravenous gene delivery using oncolytic adenoviruses. Cancer Gene Ther 2005; 12: 19–25.
Liu TC, Wang Y, Hallden G, Brooks G, Francis J, Lemoine NR et al. Functional interactions of antiapoptotic proteins and tumor necrosis factor in the context of a replication-competent adenovirus. Gene Ther 2005; 12: 1333–1346.
Sova P, Ren XW, Ni S, Bernt KM, Mi J, Kiviat N et al. A tumor-targeted and conditionally replicating oncolytic adenovirus vector expressing TRAIL for treatment of liver metastases. Mol Ther 2004; 9: 496–509.
Hawkins LK, Hermiston T . Gene delivery from the E3 region of replicating human adenovirus: evaluation of the E3B region. Gene Ther 2001; 8: 1142–1148.
Hawkins LK, Hermiston TW . Gene delivery from the E3 region of replicating human adenovirus: evaluation of the ADP region. Gene Ther 2001; 8: 1132–1141.
Hawkins LK, Johnson L, Bauzon M, Nye JA, Castro D, Kitzes GA et al. Gene delivery from the E3 region of replicating human adenovirus: evaluation of the 6.7K/gp19K region. Gene Ther 2001; 8: 1123–1131.
Jin F, Kretschmer PJ, Hermiston TW . Identification of novel insertion sites in the Ad5 genome that utilize the Ad splicing machinery for therapeutic gene expression. Mol Ther 2005; 12: 1052–1063.
Zhu M, Bristol JA, Xie Y, Mina M, Ji H, Forry-Schaudies S et al. Linked tumor-selective virus replication and transgene expression from E3-containing oncolytic adenoviruses. J Virol 2005; 79: 5455–5465.
Kretschmer PJ, Jin F, Chartier C, Hermiston TW . Development of a transposon-based approach for identifying novel transgene insertion sites within the replicating adenovirus. Mol Ther 2005; 12: 118–127.
Duncan SJ, Gordon FC, Gregory DW, McPhie JL, Postlethwaite R, White R et al. Infection of mouse liver by human adenovirus type 5. J Gen Virol 1978; 40: 45–61.
Engelhardt JF, Ye X, Doranz B, Wilson JM . Ablation of E2A in recombinant adenoviruses improves transgene persistence and decreases inflammatory response in mouse liver. Proc Natl Acad Sci USA 1994; 91: 6196–6200.
Bett AJ, Prevec L, Graham FL . Packaging capacity and stability of human adenovirus type 5 vectors. J Virol 1993; 67: 5911–5921.
Berkner KL, Sharp PA . Generation of adenovirus by transfection of plasmids. Nucleic Acids Res 1983; 11: 6003–6020.
Reddy PS, Ganesh S, Yu DC . Enhanced gene transfer and oncolysis of head and neck cancer and melanoma cells by fiber chimeric oncolytic adenoviruses. Clin Cancer Res 2006; 12: 2869–2878.
Berk AJ . Adenovirus promoters and E1A transactivation. Annu Rev Genet 1986; 20: 45–79.
Suzuki K, Alemany R, Yamamoto M, Curiel DT . The presence of the adenovirus E3 region improves the oncolytic potency of conditionally replicative adenoviruses. Clin Cancer Res 2002; 8: 3348–3359.
Wang Y, Hallden G, Hill R, Anand A, Liu TC, Francis J et al. E3 gene manipulations affect oncolytic adenovirus activity in immunocompetent tumor models. Nat Biotechnol 2003; 21: 1328–1335.
Warrens AN, Jones MD, Lechler RI . Splicing by overlap extension by PCR using asymmetric amplification: an improved technique for the generation of hybrid proteins of immunological interest. Gene 1997; 186: 29–35.
Chartier C, Degryse E, Gantzer M, Dieterle A, Pavirani A, Mehtali M . Efficient generation of recombinant adenovirus vectors by homologous recombination in Escherichia coli. J Virol 1996; 70: 4805–4810.
He TC, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B . A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci USA 1998; 95: 2509–2514.
Gorziglia MI, Lapcevich C, Roy S, Kang Q, Kadan M, Wu V et al. Generation of an adenovirus vector lacking E1, e2a, E3, and all of E4 except open reading frame 3. J Virol 1999; 73: 6048–6055.
Farson D, Tao L, Ko D, Li Q, Brignetti D, Segawa K et al. Development of novel E1-complementary cells for adenoviral production free of replication-competent adenovirus. Mol Ther 2006; 14: 305–311.
Mittereder N, March KL, Trapnell BC . Evaluation of the concentration and bioactivity of adenovirus vectors for gene therapy. J Virol 1996; 70: 7498–7509.
Yu DC, Chen Y, Seng M, Dilley J, Henderson DR . The addition of adenovirus type 5 region E3 enables calydon virus 787 to eliminate distant prostate tumor xenografts. Cancer Res 1999; 59: 4200–4203.
Bauzon M, Castro D, Karr M, Hawkins LK, Hermiston TW . Multigene expression from a replicating adenovirus using native viral promoters. Mol Ther 2003; 7: 526–534.
Wold WS, Tollefson AE, Hermiston TW . E3 transcription unit of adenovirus. Curr Top Microbiol Immunol 1995; 199 (Part 1): 237–274.
Kozak M . At least six nucleotides preceding the AUG initiator codon enhance translation in mammalian cells. J Mol Biol 1987; 196: 947–950.
Shaw AR, Ziff EB . Transcripts from the adenovirus-2 major late promoter yield a single early family of 3′ coterminal mRNAs and five late families. Cell 1980; 22: 905–916.
Acknowledgements
We thank David L Ennist for helpful discussion with regards to E3 insertion sites, and Peter Working for critical reading of the paper. B Batiste, J Ho, T Langer and S Tanciongco are gratefully acknowledged for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Robinson, M., Ge, Y., Ko, D. et al. Comparison of the E3 and L3 regions for arming oncolytic adenoviruses to achieve a high level of tumor-specific transgene expression. Cancer Gene Ther 15, 9–17 (2008). https://doi.org/10.1038/sj.cgt.7701093
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7701093
Keywords
This article is cited by
-
Armed replicating adenoviruses for cancer virotherapy
Cancer Gene Therapy (2009)