Abstract
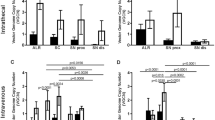
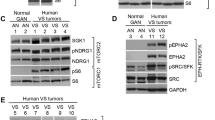
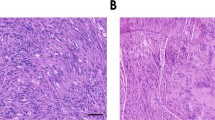
Schwannomas are benign tumors composed of dedifferentiated Schwann cells that form along peripheral nerves causing nerve compression often associated with pain and loss of function. Current surgical therapy involves total or subtotal surgical removal of the tumor, which may cause permanent nerve damage. In the present study, we explore an alternate means of therapy in which schwannomas are injected with a replication-conditional herpes simplex virus (HSV) vector to shrink the tumor through cell lysis during virus propagation. The oncolytic vector used, G47Δ, has deletions in HSV genes, which allow it to replicate selectively in dividing cells, sparing neurons. Two schwannoma cell lines were used to generate subcutaneous tumors in nude mice: HEI193, an immortalized human line previously established from an NF2 patient and NF2S-1, a newly generated spontaneous mouse line. Subcutaneous HEI193 tumors grew about ten times as fast as NF2S-1 tumors, and both regressed substantially following injection of G47Δ. Complete regression of HEI193 tumors was achieved in most animals, whereas all NF2S-1 tumors resumed growth within 2 weeks after vector injection. These studies provide a new schwannoma model for testing therapeutic strategies and demonstrate that oncolytic HSV vectors can be successfully used to shrink growing schwannomas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Short MP, Richardson EPJ, Haines JL, Kwiatkowski DJ . Clinical, neuropathological and genetic aspects of the tuberous sclerosis complex. Brain Pathol 1995; 5: 173–179.
Baser ME, Evans DG, Gutmann DH . Neurofibromatosis 2. Curr Opin Neurol 2003; 16: 27–33.
McClatchey AI, Giovannini M . Membrane organization and tumorigenesis—the NF2 tumor suppressor, Merlin. Genes Dev 2005; 19: 2265–2277.
Jessen KR, Mirsky R . The origin and development of glial cells in peripheral nerves. Nat Rev Neurosci 2005; 6: 671–682.
MacCollin M, Chiocca EA, Evans DG, Friedman JM, Horvitz R, Jaramillo D et al. Diagnostic criteria for schwannomatosis. Neurology 2005; 64: 1838–1845.
Wilkes D, McDermitt DA, Basson CT . Clinical phenotypes and molecular genetic mechanisms of Carney complex. Lancet Oncol 2005; 6: 501–508.
Trofatter JA, MacCollin MM, Rutter JL, Murrell JR, Duyao MP, Parry DM et al. A novel moesin-, ezrin-, radixin-like gene is a candidate for the neurofibromatosis 2 tumor suppressor. Cell 1993; 72: 791–800.
Rouleau GA, Merel P, Lutchman M, Sanson M, Zucman J, Marineau C et al. Alteration in a new gene encoding a putative membrane-organizing protein causes neuro-fibromatosis type 2. Nature 1993; 63: 515–521.
Martuza RL, Eldridge R . Neurofibromatosis 2 (bilateral acoustic neurofibromatosis. N Engl J Med 1988; 318: 684–688.
Giovannini M, Robanus-Maandag E, Niwa-Kawakita M, van der Valk M, Woodruff JM, Goutebroze L et al. Schwann cell hyperplasia and tumors in transgenic mice expressing a naturally occurring mutant NF2 protein. Genes Dev 1999; 13: 978–986.
Messerli SM, Tang Y, Giovannini M, Bronson R, Weissleder R, Breakefield XO . Detection of spontaneous schwannomas by MRI in a transgenic murine model of neurofibromatosis type 2. Neoplasia 2002; 4: 501–509.
Giovannini M, Robanus-Maandag E, van der Valk M, Niwa-Kawakita M, Abramowski V, Goutebroze L et al. Conditional biallelic Nf2 mutation in the mouse promotes manifestations of human neurofibromatosis type 2. Genes Dev 2000; 14: 1617–1630.
Kalamarides M, Niwa-Kawakita M, Leblois H, Abramowski V, Perricaudet M, Janin A et al. Nf2 gene inactivation in arachnoidal cells is rate-limiting for meningioma development in the mouse. Genes Dev 2002; 16: 1060–1065.
Robanus-Maandag E, Giovannini M, van der Valk M, Niwa-Kawakita M, Abramowski V, Antonescu C et al. Synergy of Nf2 and p53 mutations in development of malignant tumours of neural crest origin. Oncogene 2004; 23: 6541–6547.
Messerli SM, Prabhakar S, Tang Y, Mahmood U, Weissleder R, Bronson R et al. Treatment of schwannomas with an oncolytic recombinant herpes simplex virus in murine models of neurofibromatosis type 2. Hum Gene Ther 2006; 17: 20–30.
Aghi M, Chiocca EA . Genetically engineered herpes simplex viral vectors in the treatment of brain tumors: a review. Cancer Invest 2003; 21: 278–292.
Markert JM . Biologic warfare for a good cause: HSV-1 anti-tumor therapy. Clin Neurosci 2004; 51: 73–80.
Mineta T, Rabkin SD, Yazaki T, Hunter WD, Martuza RL . Attenuated multi-mutated herpes simplex virus-1 for the treatment of malignant gliomas. Nat Med 1995; 1: 938–943.
Todo T, Martuza RL, Rabkin SD, Johnson PA . Oncolytic herpes simplex virus vector with enhanced MHC class I presentation and tumor cell killing. Proc Natl Acad Sci USA 2001; 98: 6396–6401.
Hung G, Li X, Faudoa R, Xeu Z, Kluwe L, Rhim JS et al. Establishment and characterization of a schwannoma cell line from a patient with neurofibromatosis 2. Int J Oncol 2002; 20: 475–482.
Galve-Roperh I, Sanchez C, Cortes ML, del Pulgar TG, Izquierdo M, Guzman M . Anti-tumoral action of cannabinoids: involvement of sustained ceramide accumulation and extracellular signal-regulated kinase activation. Nat Med 2000; 6: 313–319.
Kleihues P, Cavenee WK . Pathology and Genetics of Tumours of the Nervous System. IARC Press: Lyon, 2000.
Gutmann DH, Giovannini M . Mouse models of neurofibromatosis 1 and 2. Neoplasia 2002; 4: 279–290.
McClatchey AI, Cichowski K . Mouse models of neurofibromatosis. Biochem Biophys Acta 2001; 1471: M73–M80.
Mashour GA, Moulding HD, Chahlavi A, Khan GA, Rabkin SD, Martuza RL et al. Therapeutic efficacy of G207 in a novel peripheral nerve sheath tumor model. Exp Neurol 2001; 169: 64–71.
Chiocca EA . Oncolytic viruses. Nat Rev Cancer 2002; 2: 938–950.
Markert JM, Medlock MD, Rabkin SD, Gillespie GY, Todo T, Hunter WD et al. Conditionally replicating herpes simplex virus mutant, G207 for the treatment of malignant glioma: results of a phase I trial. Gene Ther 2000; 7: 867–874.
Hill A, Jugovic P, York I, Russ G, Bennink J, Yewdell J et al. Herpes simplex virus turns off the TAP to evade host immunity. Nature 1995; 375: 411–415.
Liu R, Varghese S, Rabkin SD . Oncolytic herpes simplex virus vector therapy of breast cancer in C3(1)/SV40 T-antigen transgenic mice. Cancer Res 2005; 65: 1532–1540.
Acknowledgements
We thank Dr Mia MacCollin for her invaluable help in obtaining human schwannoma tissue, Dr Hung at the House Ear Institute for providing the human schwannoma cell line and Ms Suzanne McDavitt for skilled editorial assistance. This work was funded by the Department of the Army, US Army Medical Research and Material Command Award # DAMD17-00-1-0537 and W81XWH-04-1-0237. The Terrill Family and the Texas Neurofibromatosis Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prabhakar, S., Messerli, S., Stemmer-Rachamimov, A. et al. Treatment of Implantable NF2 Schwannoma Tumor Models with Oncolytic Herpes Simplex Virus G47Δ. Cancer Gene Ther 14, 460–467 (2007). https://doi.org/10.1038/sj.cgt.7701037
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7701037
Keywords
This article is cited by
-
A phase I/II study of triple-mutated oncolytic herpes virus G47∆ in patients with progressive glioblastoma
Nature Communications (2022)
-
Schwannoma gene therapy by adeno-associated virus delivery of the pore-forming protein Gasdermin-D
Cancer Gene Therapy (2019)
-
Oncolytic herpes simplex virus and immunotherapy
BMC Immunology (2018)
-
Pediatric cancer gone viral. Part II: potential clinical application of oncolytic herpes simplex virus-1 in children
Molecular Therapy - Oncolytics (2015)
-
Antitumor Efficacy of 34.5ENVE: A Transcriptionally Retargeted and “Vstat120”-expressing Oncolytic Virus
Molecular Therapy (2012)