Abstract
The endoplasmic reticulum (ER) serves as the major intracellular Ca2+ store and has a role in the synthesis and folding of proteins. BAX (BCL2-associated X protein) inhibitor-1 (BI-1) is a Ca2+ leak channel also implicated in the response against protein misfolding, thereby connecting the Ca2+ store and protein-folding functions of the ER. We found that BI-1-deficient mice suffer from leukopenia and erythrocytosis, have an increased number of splenic marginal zone B cells and higher abundance and nuclear translocation of NF-κB (nuclear factor-κ light-chain enhancer of activated B cells) proteins, correlating with increased cytosolic and ER Ca2+ levels. When put into culture, purified knockout T cells and even more so B cells die spontaneously. This is preceded by increased activity of the mitochondrial initiator caspase-9 and correlated with a significant surge in mitochondrial Ca2+ levels, suggesting an exhausted mitochondrial Ca2+ buffer capacity as the underlying cause for cell death in vitro. In vivo, T-cell-dependent experimental autoimmune encephalomyelitis and B-cell-dependent antibody production are attenuated, corroborating the ex vivo results. These results suggest that BI-1 has a major role in the functioning of the adaptive immune system by regulating intracellular Ca2+ homeostasis in lymphocytes.
Similar content being viewed by others
Main
The endoplasmic reticulum (ER) serves as the major intracellular calcium (Ca2+) store, the release of which controls a vast array of cellular functions from short-term responses such as contraction and secretion to long-term regulation of cell growth and proliferation.1 Dysregulated release of ER Ca2+, in contrast, initiates programmed cell death by several mechanisms including mitochondrial Ca2+ overload, depolarization, ATP loss and cytochrome c release.2 Besides this, the ER also has a key role in the synthesis, folding and sorting of proteins destined for the secretory pathway. The deleterious consequences of an increase in unfolded proteins is called ER stress and can be antagonized by the unfolded protein response (UPR), a mechanism that coordinates a simultaneous increase in the ER folding capacity and a decrease in folding load. In the case of insufficient adaptation to ER stress, cells undergo apoptosis.3
BAX (BCL2-associated X protein) inhibitor-1 (BI-1) is an evolutionarily conserved protein that bridges both the Ca2+ homeostasis and UPR functions of the ER.4 BI-1 was first identified in a screen for human proteins capable of inhibiting BAX-mediated cell death in yeast.5 In mammalian cells, BI-1's antiapoptotic function is most pronounced in paradigms of ER stress6 and involves changes in the amount of Ca2+ that can be released from intracellular stores.6, 7 BI-1 is a highly hydrophobic protein that forms a Ca2+ pore responsible for its Ca2+ leak properties8 and is the founding member of a family of six proteins with similar properties.9 The increase in the ER Ca2+ leak mediated by BI-1 is blocked at a more acidic pH10 – a function recently corroborated by a structural analysis of a bacterial homolog of BI-1.11
Despite its evolutionarily conserved role in important functions such as ER stress and Ca2+ regulation, bi-1−/− mice were reported to have no phenotypic abnormalities but increased infarct volumes in a stroke model, and increased sensitivity to tunicamycin-induced kidney toxicity.6 Moreover, livers from BI-1-deficient mice regenerate faster than those from wild-type (WT) mice and this correlates with increased nuclear translocation of nuclear factor of activated T cells (NFATs)12, a Ca2+-dependent process. BI-1 knockout (KO) mice also express more of the spliced form of X-box-binding protein-1 (sXBP-1) in their liver and kidney,13 which is generated by the endoribonuclease activity of inositol requiring enzyme 1 (IRE1), and is considered an indicator of increased UPR activity. This was later reproduced and attributed to an inhibitory function of BI-1 on IRE1α mediated via a direct interaction of the two proteins.14
In our study, we found that bi-1−/− mice are more obese and suffer from leukopenia. T and B cells from these mice show significant changes in cellular Ca2+ homeostasis and dynamics, and are more prone to spontaneous death in culture but, surprisingly, demonstrate no signs of ongoing ER stress within the homeostatic system of the living animal. These changes lead to an attenuated functioning of the adaptive immune system in vivo. Our results suggest that a major role of BI-1 in vivo involves its effects on the intracellular Ca2+ homeostasis in lymphocytes in line with its function as an ER Ca2+ leak channel.
Results
BI-1 mice are obese and suffer from leukopenia and erythrocytosis
Previously described BI-1 KO mice6 were backcrossed to WT C57/BL6 mice and identified by genotyping because of the lack of functional antibodies.15 A thorough phenotypic analysis of WT and KO littermates revealed that BI-1 KO mice at the age of 3 months are significantly more obese, even on a normal chow diet, and suffer from leukopenia (shown to be mainly lymphopenia and neutropenia by manual inspection) and erythrocytosis (Table 1). The effect of BI-1 deficiency therefore appears to be most pronounced with respect to homeostasis of the hematolymphoid system and energy balance.
BI-1 deficiency causes significant alterations of the B-cell compartment in the spleen
We focused on the immune system and studied the secondary lymphoid organs. The spleen, thymus, Peyers’ patches and lymph nodes showed no gross abnormalities and were of similar size. The bone marrow, thymus, the peritoneal cavity and the spleen contained similar counts of total cells (Supplementary Figure 1a), and the subcellular composition of cells in the bone marrow (pro/pre, immature and mature recirculating B cells) and the thymus (CD4 and CD8 T cells) determined by flow cytometry were comparable (Supplementary Figures 1b–d). We also observed no significant difference in the percentage as well as total cell counts of splenic B and T cells of BI-1 KO mice compared with age-matched controls (Figure 1a).
BI-1 deficiency causes significant alterations of the B-cell compartment. Flow cytometric analysis of splenic cells from 8- to 12-week old WT and BI-1 KO mice. T and B cells were stained with (a) B220 and CD90.2 antibodies, and (b) MZ and FO B cells with IgM and CD21 antibodies pregated on live B220+ B cells. Bar graphs show the mean±S.E.M., n=3. Data were analyzed by two-tailed t-tests, *P<0.05. (c) Representative immunofluorescence analysis of MZ B cells stained with α-CD1d and metallophillic macrophages stained with α-MOMA-1 in spleen tissue sections
Immature B cells leave the bone marrow and migrate to the spleen where they complete their maturation process. In the spleen, these cells become follicular (FO) B cells, which recirculate in the periphery, or marginal zone (MZ) B cells, which are largely sessile. An analysis of these cell types, immature (CD21−IgM−), FO (CD21intIgMhigh) and MZ (CD21highIgMneg-low) B cells revealed a significant elevation of MZ B cells in KO spleens (KO 20.5±5.2% versus WT 10.9±2.7%) at the expense of FO B cells (Figure 1b). This was also evident in splenic sections stained with an anti-CD1d antibody, which recognizes MZ B cells. Interestingly, in the KO mice, MZ B cells were also found within the follicles, whereas in controls, strong CD1d staining was only detected in the MZ as expected (Figure 1c). Taken together, these results demonstrate a significant increase in the generation, differentiation or maintenance of mature MZ cells at the expense of FO B cells in BI-1 KO mice compared with WT controls.
Increased constitutive nuclear translocation of the transcription factor NF-κB in BI-1 KO splenocytes
Such a shift towards MZ B cells has been previously linked to NF-κB (nuclear factor-κ light-chain enhancer of activated B cells) signaling. NF-κB1 (p105/p50) and NF-κB2 (p100/p52) need to be processed to become active and translocate to the nucleus where they associate with RelA (p65), c-Rel and RelB in different hetero- and homodimers to form the transcriptionally active complex.16 Mice deficient in RelB show reduced numbers of MZ B cells,17 whereas mice lacking a negative regulator of NF-κB have more MZ B cells18 similar to mice that lack the inhibitory p100 subunit of NF-κB2.19
We therefore quantified the abundance of all NF-κB proteins in the cytosol and the nucleus by immunoblotting of subcellular fractions obtained from freshly isolated splenocytes. This revealed an increased cytosolic abundance of c-Rel and RelA and an increased nuclear abundance of RelB in BI-1 KO cells (Figure 2a), whereas the proteins that lack transcriptional activation domains, NF-κB1/2, remained unchanged (Supplementary Figure 2a). Because of the feeble nuclear abundance of RelA (p65) in these unstimulated cells, we used immunocytochemistry with another antibody and quantification by high-content imaging as an additional read-out. In these experiments, RelA (p65) translocated to the nucleus to a much higher degree (P<0.0001) in BI-1-deficient splenocytes, as suggested by the immunoblotting experiments (Figure 2b).
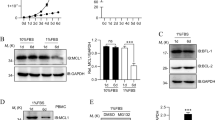
Increased expression and nuclear translocation of class II NF-κB proteins in BI-1 KO splenocytes. (a) Immunoblots with cytosolic and nuclear fractions obtained from WT and KO splenocytes incubated with antibodies against class II NF-κB proteins show increased cytosolic and nuclear abundance in BI-1 KO splenocytes. Loading controls, α/β-tubulin for the cytosolic fraction and HDAC for the nuclear fraction, are shown on the top. Molecular weight is indicated. Bar graphs show mean intensity values normalized to WT±S.E.M., n=8. (b) Increased nuclear localization of RelA (p65) shown by quantitative immunocytochemistry in BI-1 KO splenocytes. Splenocytes were stained with an α-RelA (p65) antibody and analyzed by confocal microscopy. Quantification was carried out on a high-content imaging microscope by measuring the fluorescence intensity in the nucleus defined by DAPI (4',6-diamidino-2-phenylindole) staining and in the cytosol defined as an area around the nucleus. The scatter plot shows the ratio of nuclear to cytosolic RelA (p65) (300 WT and 432 KO cells, n=3 independent experiments, mean is indicated). (c) Increased ratio of RelA (p65) and IκBα and (d) downregulated phosphorylated IκBα in BI-1 KO splenocytes. Actin served as a loading control on the same blot, size is indicated. Bar graphs show mean intensity values normalized to WT±S.E.M., n=6. Data were analyzed by two-tailed t-tests, *P<0.05
In unstimulated cells, the Rel/NF-κB dimers associate with members of the family of inhibitor proteins called IκBs and remain inactive in the cytoplasm. In NF-κB not bound by IκBα in the cytosol, the nuclear localization signal of p65 is liberated, which allows its translocation into the nucleus. The NF-κB/IκB ratio thus determines the amount of nuclear-translocated NF-κB and this ratio was indeed increased in BI-1 KO splenocytes (Figure 2c). IκB is usually removed from NF-κB by activation of the IκB kinase complex, which leads to phosphorylation of IκBα, resulting in ubiquitination and degradation of IκBα. Phosphorylated IκBα was downregulated in BI-1 KO splenocytes (Figure 2d). We conclude that NF-κB proteins harboring a transcriptional activation domain are upregulated and show an increased nuclear localization in the absence of BI-1, which may explain the increase in MZ B cells in these animals compared with controls.
BI-1 KO splenocytes have increased ER and cytosolic Ca2+ levels and a compensatory downregulation of store-operated Ca2+ entry
NF-κB is activated in a pleiotropic manner including changes in the amplitude and duration of Ca2+ signals in lymphocytes.20, 21 Also, treatment with inhibitors of sarcoplasmic/ER Ca2+-ATPases (SERCA), which increase cytosolic Ca2+, causes nuclear NF-κB translocation.22 NF-κB can also be activated by a pathological mechanism known as the ER overload reaction. This condition is caused by retention of proteins within the ER lumen, and in contrast to the UPR does not induce Grp78/BiP (glucose-regulated protein 78 kDa/binding immunoglobulin protein),23 which is also unchanged in the absence of BI-1.12, 13 Notably, NF-κB induction by the ER overload reaction can be attenuated by intracellular Ca2+ chelators.24, 25
Therefore, both major functions of BI-1, its role in ER stress and in the maintenance of the intracellular Ca2+ homeostasis, might be implicated in the observed phenotype. We first investigated the relative abundance of UPR proteins by immunoblotting of splenocyte lysates from BI-1 KO and WT mice and detected no evidence of ongoing ER stress in BI-1-deficient splenocytes. Rather, several of the UPR proteins were downregulated, especially ERp57. Importantly, BiP was unchanged (Supplementary Figure 3a). Despite the role of BI-1 as a direct inhibitor of IRE1α,14 we observed decreased XBP-1 splicing in KO cells (Supplementary Figure 3b) in line with the decreased expression of ER stress-regulated chaperones. We concluded that the UPR and the ER overload reactions are not constitutively active in splenocytes isolated directly from BI-1 KO mice.
The other major role of BI-1 is the regulation of cellular Ca2+ homeostasis. Using flow cytometry of freshly isolated splenocytes, we measured resting Ca2+ levels in WT and KO splenocytes with Fluo8-AM and found increased cytosolic Ca2+ levels in KO cells (Figure 3a). We next quantified the ER Ca2+ content and store-operated Ca2+ entry (SOCE) from the extracellular space, which is activated by a decrease in [Ca2+]ER using the ratiometric probe Fura2. The cells were attached to plates to allow a kinetic analysis of (1) Ca2+ passively leaking from the ER after inhibition of the Ca2+ reuptake pump SERCA in the absence of extracellular Ca2+ and then (2) Ca2+ re-entry from SOCE after re-exposure of the cells to large amounts of extracellular Ca2+. BI-1 KO splenocytes had an increased baseline Ca2+ concentration (corroborating the results obtained with Fluo8), an increased thapsigargin-releasable ER Ca2+ pool and reduced SOCE (Figure 3b).
Increased ER and cytosolic Ca2+ levels cause increased constitutive nuclear translocation of the Ca2+-dependent transcription factor NFAT in BI-1 KO splenocytes. (a) Increased cytosolic Ca2+ in BI-1 KO splenocytes measured by flow cytometry of splenocytes stained with Fluo8-AM. Fluo8-AM fluorescence at 488 nm is proportional to the amount of bound Ca2+ and is quantified as mean fluorescence intensity (MFI). The histogram shows representative data of unstained (black), WT (blue) and BI-1 KO (red) splenocytes where each cell is plotted against its relative fluorescence intensity on a log scale. Box and whiskers show the quantification of data normalized to WT animals (n=12, median, box from the 25th quartile to the 75th quartile, whiskers minimum to maximum). (b) BI-1 KO splenocytes have increased baseline Ca2+ and an increased thapsigargin-releasable Ca2+ pool but decreased store-operated Ca2+ entry (SOCE). Cells stained with Fura2-AM were attached to poly-l-lysine-coated plates and the intracellular Ca2+ concentration recorded in response to the indicated SOCE protocol on a high-content imaging microscope. The traces show the mean of the calibrated Ca2+ concentration from three pooled independent experiments. The box and whisker plot represent the statistical distribution of the absolute Ca2+ concentration at baseline, the baseline-corrected thapsigargin-releasable pool and the amount of Ca2+ that entered the cell during SOCE. (c) Orai1 is downregulated in BI-1 KO splenocytes. Immunoblots were incubated with α-STIM1, α-STIM2, α-ORAI1 and α-pan IP3R antibodies. Actin served as a loading control on the same blot; size is indicated. Bar graphs show mean intensity values normalized to WT±S.E.M., n=6. (d) Increased nuclear localization of NFATc1 in BI-1 KO splenocytes. Splenocytes were stained with α-NFATc1 and analyzed by confocal microscopy. Quantification was carried out on a high-content imaging microscope by measuring the fluorescence intensity in the nucleus defined by DAPI (4',6-diamidino-2-phenylindole) staining and in the cytosol defined as an area around the nucleus. The scatter plot shows the ratio of nuclear-to-cytosolic NFAT (275 WT and 312 KO cells from n=3 independent experiments. The mean is indicated. (e) The expression of all three NFATc1 isoforms is not regulated in BI-1 KO splenocytes. Immunoblots were incubated with an α-NFATc1 antibody. Actin served as a loading control on the same blot; size is indicated. Bar graphs show mean intensity values normalized to WT±S.E.M., n=6. Data were analyzed by two-tailed t-tests, *P<0.05
This SOCE reduction is mediated by a downregulation of Orai1, the plasma membrane channel mediating SOCE,26, 27 and probably serves as a compensatory mechanism to limit filling of the already Ca2+-full ER (Figure 3c). Taken together, these data corroborate that BI-1 deficiency causes profound alterations of the intracellular Ca2+ homeostasis in freshly isolated splenocytes, which manifests as increased Ca2+ concentrations in the ER, and, surprisingly, also in the cytoplasm. It is thus possible that the alterations in the intracellular Ca2+ homeostasis are implicated in the observed increase in NF-κB nuclear translocation.
To test this assumption, we investigated the cellular localization of another transcription factor, NFAT, which translocates to the nucleus upon the pronounced Ca2+ release from the ER in response to activation of the antigen receptors.21 We observed that NFATc1 was also already translocated to a much higher degree in BI-1-deficient splenocytes even without stimulation, similar to NF-κB (Figure 3d) but unchanged at the expression level (Figure 3e). These data corroborate results obtained in BI-1-deficient hepatocytes, which also showed an increased constitutive translocation of NFATc1,12 and support the assumption that the changes in Ca2+ signaling are responsible for the increased abundance and nuclear translocation of NF-κB in BI-1-deficient lymphocytes and thus for the changes in MZ cell abundance.
We next investigated alterations of the Ca2+ homeostasis and NF-κB translocation in purified T and B cells. Fluo8-AM staining revealed higher resting cytosolic Ca2+ concentrations in B cells compared with T cells (Figure 4a) matching nuclear translocation levels of RelA (p65) (Figure 4b). To study physiological ligand-activated Ca2+ release, we incubated Fluo8-stained splenocytes with biotinylated anti-CD3 antibodies and measured Fluo8 intensity by flow cytometry for 60 s to establish a baseline. Then, streptavidin was added to crosslink the anti-CD3 antibodies on the surface, which activates the T-cell receptor and elicits inositol 1,4,5-trisphosphate (IP3)-dependent Ca2+ release from the ER and subsequent SOCE. B cells were similarly stimulated by the addition of anti-IgM. Both KO T and B cells showed increased baseline Ca2+ concentrations and B cells also showed an increased Ca2+ release after stimulation (Figure 4c). To further substantiate this, we then quantified IgM-mediated increases in the mitochondrial electron transfer capacity of B cells from WT and KO animals. IgM-mediated Ca2+ release from the ER elevates mitochondrial matrix [Ca2+], thereby stimulating Krebs’ cycle dehydrogenases and elevating mitochondrial [NADH],28 which is then fed into the oxidative phosphorylation pathway. This indeed revealed an increased stimulation in KO cells as a functional readout of the increased ER Ca2+ release (Figure 4d).
T and B cells are affected by BI-1 deficiency. (a) Basal cytosolic Ca2+ is increased in BI-1 KO T and B cells. Magnetically sorted and negatively selected T and B cells were incubated with Fluo8-AM and the fluorescence measured by flow cytometry. Bar graphs represent the average mean fluorescence intensity as the median±S.D., n=6. (b) Increased RelA (p65) nuclear localization in BI-1 KO B and T cells. B and T cells were magnetically sorted and negatively selected and stained with an RelA (p65) antibody and DAPI (4',6-diamidino-2-phenylindole). Quantification was carried out on a high-content imaging microscope by measuring the fluorescence intensity in the nucleus defined by DAPI staining and in the cytosol defined as the area around the nucleus. The scatter plot shows the ratio of nuclear to cytosolic RelA (p65) (B cells: 1931 WT and 1968 KO cells; T cells: 971 WT and 941 KO cells) from three independent experiments. The mean is indicated. Data were analyzed by two-tailed t-tests, *P<0.05. (c) Ligand-induced Ca2+ release in BI-1 KO T and B cells quantified by flow cytometry. Fluo8-AM fluorescence at 488 nm is proportional to the amount of bound Ca2+ and is quantified as mean fluorescence intensity (MFI). Splenocytes were incubated with biotinylated anti-CD3 antibody and stimulated with streptavidin or directly stimulated with anti-IgM after 60 s of baseline. Streptavidin causes the biotinylated anti-CD3 to cluster and thereby activates T cells, whereas anti-IgM selectively activates B cells. Representative traces of Fluo8 kinetics are shown for WT (black) and BI-1 KO (red) T and B cells. (d) Increased electron transfer system (ETS) capacity in BI-1 KO B cells treated with anti-IgM measured by high-resolution respirometry. Routine respiration was measured under basal conditions, leak respiration after inhibition of ATP synthase using oligomycin and the ETS capacity at maximum uncoupling with FCCP and oxygen flow. All measurements were corrected for residual oxygen consumption after inhibition of complex I with rotenone and complex III with antimycin A. Shown is the mean±S.D., n=4 mice. Data were analyzed by two-tailed t-tests, *P<0.05
Increased spontaneous B-cell death in culture is associated with exhausted mitochondrial Ca2+ buffering capacity and caspase activation
When we took purified T and B cells into culture, we noted that after 24 h, KO cells were much more prone to spontaneous cell death. This was again more pronounced in B cells (Figure 5a) and we focused on these cells for further investigations of the impact of BI-1 deficiency. An analysis by electron microscopy of purified B cells revealed alterations in the texture of the cytoplasm, defective organelles (e.g., mitochondria) and the presence of nuclear inclusions in nearly all KO cells (Supplementary Figure 4). Quantification of B-cell death in a time-course analysis by flow cytometry revealed a statistically significant difference in viability after only 4 h in culture (WT 11.0±1.2% and KO 15.6±0.3%) that increased over time. After 24 h in culture, >68±4.8% of KO cells were dead compared with 28±5.9% WT cells (Figure 5b).
Increased spontaneous B-cell death in culture is caused by an exhausted mitochondrial Ca2+ buffer capacity leading to caspase activation. (a) B and T cells were magnetically sorted, negatively selected and put into culture. Cell death was assessed 24 h later by flow cytometry using a live/dead stain and demonstrates increased spontaneous cell death in KO splenocytes with B cells being more susceptible compared with T cells. (b) A time-course analysis of B-cell death with significant differences already evident after 4 h in culture. Cell death was assessed as described in (a). The graphs in (a and b) show the mean percentage of dead cells±S.E.M. from six mice at each time point. (c) Caspase-3/7 activity in both WT and BI-1 KO B cells increases in a time-dependent manner and becomes significantly increased in KO cells at 4 h in culture. (d) Caspase-8 and -9 are significantly increased in BI-1 KO B cells after 3 h in culture, with caspase-9 being more prominent. All caspase activity was assessed in magnetically sorted and negatively selected B cells using a luminescent caspase assay. Caspase-8 and -9 were assessed in parallel. Bar graphs represent the mean average relative luminescence±S.E.M. from six mice measured in triplicates (e–g) After 4 h in culture, the mitochondrial Ca2+ is significantly increased in KO B cells. Magnetically sorted and negatively selected B cells were stained with (e) Rhod2-AM, (f) Fluo8-AM or (g) CellROX at the indicated time points. The graph represents the mean fluorescence intensity±S.D., n=3–6. Data were analyzed by two-tailed t-tests, *P<0.05. (h) XBP-1 splicing was assessed by PCR in magnetically sorted and negatively selected B cells at the indicated time points in culture. Bar graphs show the mean percentage±S.D. of spliced XBP-1/(spliced XBP-1+unspliced XBP-1) from six mice. Data were analyzed by two-tailed t-tests, *P<0.05. (i) Magnetically sorted and negatively selected B cells were stained with a live/dead stain, Fluo8-AM or Rhod2-AM after 4 h in culture. In the pH experiments, the cells were cultured as normally and put into HBSS buffer with the indicated pH immediately after staining and directly processed; in the Ca2+ experiments, the culturing medium was adjusted with CaCl2 (mM) and EGTA and processed as described above. Bar graphs represent the mean fluorescence intensity as the median±S.D., n=6–24 mice
Because caspases are principal effectors of apoptosis, we measured the activity of the effector caspases 3 and 7 after placing B cells into culture. We detected a significant difference between KO and WT cells already after 4 h in culture, with higher relative levels of effector caspase activity in the KO cells (Figure 5c). We quantified the activity of initiator caspases 8 and 9, which represent the apical proteases in the ‘extrinsic’ and ‘intrinsic’ apoptosis pathways, respectively, after 3 h in culture (thus before effector caspase activation became different). Activity of both of these initiator caspases was significantly higher in BI-1 KO compared with WT B cells (Figure 5d), with relative levels of caspase-9 activity considerably higher compared with caspase-8 in both KO and WT B cells placed into culture.
Caspase-9 activation classically involves cytochrome c release from the mitochondria,29 which can be stimulated by a Ca2+-dependent mechanism that impacts inner membrane permeability, followed later by rupture of the outer membrane.2 BI-1 is capable of regulating Ca2+ transport between ER and mitochondria,30 leading us to hypothesize that the altered ER and cytoplasmic Ca2+ levels in BI-1-deficient cells might be reflected by similar alterations of mitochondria. Measuring mitochondrial [Ca2+] using Rhod2-AM in freshly isolated B cells from KO versus WT animals demonstrated that Rhod2-AM fluorescence was only slightly increased at baseline but much more significantly after 4 h in culture (Figure 5e), whereas the difference between KO and WT in cytosolic [Ca2+] declined (Figure 5f). Levels of reactive oxygen species (Figure 5g) and XBP-1 splicing (Figure 5h), in contrast, did not differ in WT and KO cells at baseline or after 4 h. Exposure of these cells to different pH or extracellular [Ca2+] did not profoundly affect cell death or cytosolic and mitochondrial Ca2+ levels (Figure 5i).
These data suggest that mitochondria of BI-1-deficient B cells exceed their buffering capacity after a few hours in culture, and we envisaged that clamping the intracellular Ca2+ with the cell-permeable Ca2+ buffer BAPTA-AM should inhibit this Ca2+-dependent cell death. However, this experimental approach was not possible because BAPTA-AM treatment rapidly killed primary B cells (not shown), as has previously been shown for leukemic cells.31 Thus, the observed defects in mitochondrial Ca2+ buffering capacity seen in cultured BI-1 KO B cells are correlated with caspase activation and cell death, but a direct cause and effect relationship was not demonstrable.
BI-1 deficiency attenuates T- and B-cell function in vivo
We finally aimed to clarify the effect of the identified changes in T and B cells on immune system function in vivo using two different experimental paradigms. First, in experimental autoimmune encephalitis (EAE), the immune system is challenged by immunization with myelin proteins emulsified in complete Freund’s adjuvant, causing a mainly T-cell-driven autoimmunity against the central nervous system and the development of a multiple sclerosis-like disease that can be scored for severity. KO mice displayed a later onset of clinical symptoms and an overall lower clinical score (Figure 6a), despite the fact that the brain of BI-1 KO mice appears to be more vulnerable to exogenous stress, as shown by larger infarcts in a stroke model.6 The attenuated EAE disease score was associated with reduced infiltration of microglia as well as a decreased number of lesions detectable by immunohistochemistry (Figures 6b and c). Second, to assess B-cell function, we immunized the mice with 4-hydroxy-3-nitrophenylacetyl coupled to chicken globulin (NPCG), which is a standard T-cell-independent immunogen,32 and then quantified the resulting antibody response. BI-1 KO animals generated significantly lower amounts of IgG1 and IgG2b in response to this antigen (Figure 6d). We conclude that both T- and B-cell functions are attenuated in vivo, thereby corroborating the in vitro changes. To clarify whether this is because of defective cell signaling or the reduced number of lymphocytes in vivo, we investigated in vitro isotype switching of splenic B cells. In these experiments, splenocytes stimulated ex vivo with LPS and IL-4 start producing IgG1, which can be quantified intracellularly by flow cytometry. Upon this treatment, significantly less BI-1 KO cells switched to IgG1 compared with control splenocytes, suggesting that defective signaling is involved in the observed phenotype (Figure 6e).
BI-1 deficiency attenuates T- and B-cell function. (a) BI-1 KO mice are less susceptible to EAE elicited by immunization with the MOG35–55 peptide. Individual clinical disease scores (1, limp tail; 2, weakness of hind legs; 3, hind leg paralysis; 4, hind and front leg paralysis; 5, death) were assessed daily. Shown is the mean±S.E.M. of the daily average score for the indicated number of WT and BI-1 KO mice from one representative experiment of three. (b) Histopathology of cryosectioned brain stained with Iba1 (microglia, green), MBP (myelin, red) and Hoechst (nuclei, blue) shows less demyelination in BI-1 KO mice. (c) Cuffs and lesions caused by intruding microglia were counted 15 days after immunization. Cuffs are inflammatory vessel-centered lesions without tissue infiltration, undemyelinated (undem) is defined as parenchymal lesions without demyelination and demyelinated (demyel) is defined as parenchymal lesions with demyelination. Three to eight sections from seven mice were quantified and BI-1 KO mice show a significantly reduced number of undemyelinated lesions per section (WT 0.7±0.4 versus BI-1 KO 0.05±0.09). Bar graphs show the average number of lesions per section of each genotype as the mean±S.E.M. (d) Attenuated immunoglobulin production quantified by ELISA 10 days after immunization with NPCG precipitated in Alum in BI-1 KO mice. (e) Class switching of BI-1 KO and control splenocytes and CD19+ B cells at day 4 of in vitro stimulation with LPS+IL-4. IgG1 of 200 000 events was quantified by intracellular staining as indicated. Bar graphs show the mean±S.E.M., n=3. The box and whiskers represent the median amount of the indicated immunoglobulins with the box extending from the 25th quartile to the 75th quartile and the whiskers represent the minimum and maximum values (n=10). EAE data were analyzed by two-way ANOVA with Bonferroni post hoc correction; all other data were analyzed by two-tailed t-tests, *P<0.05
Discussion
Our observations indicate that BI-1 has a major role in the correct functioning of the adaptive immune system. B and T cells malfunction apparently because of profound changes in intracellular Ca2+ homeostasis and dynamics. All aspects of the cellular Ca2+ machinery are altered in the absence of functioning BI-1 and this correlates with an enhanced nuclear translocation of Ca2+-dependent transcription factors and an overload of the mitochondrial Ca2+ buffer capacity leading to spontaneous cell death in culture. These results are well in line with previously reported effects of BI-1 in the control of the ER Ca2+ content.6, 7, 8, 10 Also, structural evidence obtained from a bacterial homolog of BI-1 just demonstrated that BI-1 is indeed a Ca2+ leak channel with an evolutionarily conserved pH sensor.11
We observed an increased ER Ca2+ concentration as expected for cells lacking an important ER Ca2+ leak channel and a reduced SOCE caused by downregulation of the plasma membrane Ca2+ channel Orai1. It is clear that the already overfilled ER does not need further filling, but why and how does this result in the observed increase in cytosolic Ca2+ concentration?
In BI-1 KO, there is less Ca2+ leak from the ER, and, hence, more Ca2+ is available for IP3-induced Ca2+ release, which can explain the higher Ca2+ transients. It is possible that even under resting conditions, constitutive activation of B- or T-cell receptors provokes basal Ca2+ release, which is expected to be higher in KO cells. Cytosolic Ca2+ transients are normally cleared efficiently by extrusion to the extracellular space or immediate refilling of the ER by SERCA, which (i) is apparently less effective in immune cells in general, thereby explaining why this phenotype is most pronounced in the immune system33 and (ii) is directly inhibited by the Ca2+ load of the store.34 It was also shown previously that a high ER Ca2+ load negatively regulates Ca2+ reuptake from the cytosol,35 which is probably mediated by the oxidoreductase ERp57. In conditions of high luminal Ca2+, like in the absence of functional BI-1, ERp57 binds SERCA2b (the ubiquitously expressed isoform) and inhibits its activity through the facilitation of disulfide bond formation in the longest ER facing loop of SERCA2b.36 As we observed an unexpected downregulation of the UPR target protein ERp57 in BI-1 KO splenocytes, we speculate that this represents a compensatory mechanism to remove the breaks from SERCA2b and relieve the cell from the excessive cytosolic Ca2+ load.
Finally, how can we resolve our findings with the proposed function of BI-1 as a direct inhibitor of IRE1α?14 IRE1α not only interacts with BI-1 but also with the proapoptotic proteins BAX and BAK (Bcl-2 homologous antagonist/killer), which in contrast to BI-1 activate IRE1α signaling.37 Cells lacking both proteins have a reduced ER Ca2+ concentration38 because of an increased Ca2+ leak39 and therefore behave like BI-1-overexpressing cells.7 As BI-1 does not directly interact with BAX/BAK5, 14 but attenuates the interaction of BAX with IRE1α,14 it was speculated that BI-1 acts upstream of these proteins in the control of IRE1α inactivation.14 However, maybe the proposed inactivation of IRE1α by BI-1 is reciprocal, meaning that IRE1α also inhibits BI-1 channel activity. To the best of our knowledge, this has not yet been studied. In this scenario, the lack of BAX/BAK in double KO cells would inhibit IRE1α, thereby activating BI-1 and causing a reduction in the ER Ca2+ concentration, which is in accordance with reported results.
In summary, our findings present in vivo evidence that the Ca2+ channel activity of BI-1 is critically important for immune cell function and survival.
Materials and Methods
Animals and laboratory tests
BI-1 KO mice first described by Chae et al.6 were identified by genotyping according to the published protocol and backcrossed to WT C57/Bl6 mice for more than generations. All mice were maintained in the local animal facility. All animal experiments were fully approved by local authorities for animal experimentation. Mice were age and sex matched and 8–12 weeks old. The blood count and liver enzyme analysis were carried out by the clinical laboratory of the University Hospital Düsseldorf. A differential blood count was carried out manually in stained blood streaks.
Flow cytometry
Single-cell suspensions were prepared from different organs and the erythrocytes lysed with tris-ammonium chloride, pH 7.2. Cells were treated with Fc-block (Affymetrix eBioscience, San Diego, CA, USA), washed and surface stained with antibodies against CD5, CD19, CD21 (7G6), CD23, CD90.2, and IgD, B220 and anti-IgM (BD Bioscience, Franklin Lakes, NJ, USA). All samples were measured on a FACSCanto II (Becton, Dickinson, Franklin Lakes, NJ, USA) and analyzed with FlowJo, LLC (Ashland, OR, USA).
Immunohistochemistry
Animals were perfused and tissues fixed with 4% paraformaldehyde (Carl Roth, Karlsruhe, Germany). Thick slices (20 μM) of the spinal cord and 8 μm sections from spleens were cut in a cryotome (Leica Mikrosysteme Vertrieb GmbH, Wetzlar, Germany) and stained with primary antibodies against CD1d (Affymetrix eBioscience), MOMA-1 (Affymetrix eBioscience), Iba1 (Wako Chemicals GmbH, Neuss, Germany; 1 : 800) and MBP (EMD Millipore, Billerica, MA, USA; 1 : 500). Iba1 and MBP were detected with secondary anti-Cy2 and anti-Cy3 antibodies (EMD Millipore; 1 : 500) and CD1d and MOMA-1 with a biotinylated secondary antibody (DIANOVA Vertriebs-Gesellschaft mbH, Hamburg, Germany), treated with streptavidin-horseradish peroxidase and stained with tyramide (Cy3) and fluorescein (FITC). Nuclei were counterstained with Hoechst 33258 (Life Technologies GmbH, Darmstadt, Germany). Slices were mounted with Immuno Mount (Fisher Scientific GmbH, Schwerte, Germany) and analyzed on an Olympus BX51 (Olympus, Hamburg, Germany).
Transmission electron microscopy
Cells were pelleted, fixed in 2.5% glutaraldehyde, 2% PFA and 0.05% tannic acid and later treated with 2% osmium tetroxide. After staining with 1.5% uranylacetate and 1.5% phosphotungstic acid, pellets were embedded in epoxide resin (Spurr) and dissected in 70- to 80-nm-thick slices on an ultramicrotome (Reichert Ultracut; Reichert-Jung, Vienna, Austria). Images (30 KO and 32 WT) were taken on a Hitachi H 600 transmission electron microscope (Hitachi High Technologies America, Schaumburg, IL, USA).
Ca2+ measurements
Flow cytometry
Cells were incubated in culture medium consisting of RPMI 1640 (Fisher Scientific GmbH) supplemented with 10% FCS (Fisher Scientific GmbH), 100 U/ml penicillin and 100 μg/ml streptomycin (Invitrogen), 1% glutamax (Invitrogen) and 0.1% β-mercaptoethanol (Fisher Scientific GmbH), and the Ca2+ concentration was adjusted with 1M CaCl2 and 1M EGTA. A total of 1 × 106 cells per ml were loaded with either 5 μM Fluo8-AM (Santa Cruz Biotechnology, Inc., Dallas, TX, USA), 2 μM Rhod2-AM (AAT Bioquest, Sunnyvale, CA, USA) or 5 μM CellROX (Fisher Scientific GmbH), washed, measured on a BD FACSCalibur or BD FACS Canto II (BD Bioscience) and analyzed with FlowJo, LLC. For pH-dependent measurements, cells were resuspended in HBSS (Fisher Scientific GmbH) adjusted to the indicated values with 1M HCl and 1M NaOH, directly after staining and measured immediately.
Ca2+ kinetics
Fura2 Ca2+ imaging experiments were conducted on a BD Pathway 855 High Content Imaging System (BD Bioscience). The 96-well imaging plates (BD Bioscience) were coated with 5 mg/ml BSA (Carl Roth) and 0.01% (w/v) poly-l-lysine (Sigma-Aldrich, St. Louis, MO, USA), splenocytes seeded at a density of 150 000 cells per well and loaded with 3 μM Fura2-AM (Life Technologies GmbH) in HBSS at RT for 20 min before the experiment. SOCE was quantified by a high-throughput method previously validated using STIM1−/− mouse embryonic fibroblasts and shown to be reliable.40 Ratiometric Fura2-AM values were converted to absolute Ca2+ values as described41 and analyzed with Attovision (BD Bioscience). For measuring the Ca2+ kinetics of B and T cells, splenocytes were incubated with 3 μM Fluo8 for 30 min at RT. Cells were washed once in DMEM without phenol red (Fisher Scientific GmbH) and resuspended at 40 × 106 cells per ml in the washing medium. A total of 2 × 106 cells were additionally incubated with 3 μg/ml biotinylated anti-CD3 antibody (Affymetrix eBioscience) for 10 min at RT, and before the measurement, 2 × 106 cells were transferred into 500 μl Ringer solution (Fresenius Kabi, Bad Homburg, Germany). After recording the baseline for 1 min in a FACS Canto II flow cytometer, either 3 μg/ml streptavidin (Fisher Scientific GmbH) were added to the cells, which were previously incubated with biotinylated anti-CD3 antibody or 10 μg/ml anti-IgM (Jackson ImmunoResearch Laboratories, Inc., West Grove, PA, USA). Data were recorded for 5 min and analyzed with the kinetics module of FlowJo, LLC.
Immunoblotting
Splenocytes were lysed in ice-cold CelLytic M buffer (Sigma-Aldrich) containing Mini Complete Protease Inhibitor Cocktail (Sigma-Aldrich) and centrifuged for 30 min at 16 000 × g. Subcellular fractionations were prepared as described previously.42 Fifty micrograms of protein lysates were blotted and incubated with primary antibodies (diluted 1 : 1000) as follows: IP3R Pan (Rbt475),43 ERp57 (Cell Signaling; no. 2881), Ero1-Lα (Cell Signaling; no. 3264), BIP (Cell Signaling; no. 3177), ERp72 (Cell Signaling; no. 5033), ERp44 (Cell Signaling; no. 3798), GrP94 (Cell Signaling; no. 2104), IκBα (Cell Signaling; no. 4814), phospho-IκBα (Cell Signaling; no. 2859), NF-κB p65 (Cell Signaling; no. 8242), phospho-NF-κB p65 (Cell Signaling; no. 3033), ubiquitin (Dako Deutschland GmbH, Hamburg, Germany; no. Z0458), STIM1 (stromal interaction molecule 1; Abnova (Taipei, Taiwan); no. h0000678-m01), ORAI1 (1 : 500; Alomone Labs, Jerusalem, Israel; no. ACC-062), NFATc1 (7A6) (1:200; Santa Cruz Biotechnology, Inc.; no. msc-7294), NF-κB2 p100/p52 (Cell Signaling; no. 4882), HDAC1 (Cell Signaling; no. 2062), αβ-tubulin (Cell Signaling; no. 2148), RelB (Santa Cruz Biotechnology, Inc.; no. sc-226), RelA (Santa Cruz Biotechnology, Inc.; no. sc-8008), c-Rel (Santa Cruz Biotechnology, Inc.; no. sc-71), NF-κB2 p105/p50 (Santa Cruz Biotechnology, Inc.; no. sc-7178), Serca2b (a kind gift from Frank Wuytack and Peter Vangheluwe), calreticulin (1 : 500), calnexin C20 (1 : 500), calnexin H70 (1 : 500), HSPA8 (Cell Signaling; no. 8444) and actin (1 : 4000; EMD Millipore; no. MAB1501). Antisera without vendor were obtained from the Bultynck Lab (Leuven, Belgium). Secondary antibodies used were as follows: anti-mouse or anti-rabbit IgG (Fc) infrared fluorescence conjugated secondary antibodies (LI-COR, Lincoln, NE, USA; 1 : 30 000). The membranes were scanned for infrared fluorescence at 680 and 800 nm using the Odyssey System (LI-COR) and the signal was analyzed with the image processing software ImageJ (National Institutes of Health, Bethesda, MD, USA).
Immunofluorescence staining and nuclear translocation assay
Splenocytes were seeded at a density of 150 000 cells per well on BSA and poly-l-lysine-coated 96-well imaging plates, fixed, blocked and incubated overnight with anti-NF-κB p65 (Cell Signaling; no. 8242) or anti-NFATc1 (Hybridoma Bank, Iowa City, IA, USA; no. 7A6) diluted 1 : 100 in blocking solution. After incubation with Cy3-labeled secondary antibody (EMD Millipore; 1 : 500) and nuclear staining with 150 nM DAPI (Sigma-Aldrich), pictures were taken on a Leica TSC SP5 confocal microscope (Leica Mikrosysteme Vertrieb GmbH) at x63 magnification and nuclear translocation of NF-κB p65 or NFATc1 was quantified in a BD Pathway 855 high content imaging system (BD Bioscience). Nuclear regions of interest were identified by DAPI signal and a concentric region around the nuclear region of interest was assessed as the cytoplasm. The ratio of the nuclear and cytoplasmic Cy3 signal was quantified with Attovision (BD Bioscience).
Measurement of mitochondrial oxygen consumption
A phosphorylation control protocol was performed for measurements of mitochondrial oxygen consumption of purified B cells using the high-resolution respirometer Oxygraph-2k (Oroboros Instruments Corp., Innsbruck, Austria). Intact B cells were monitored in two closed glass chambers under continuous stirring at 750 r.p.m. at 37 °C in 2 ml growth medium at a density of 3–4 × 106 cells per ml. After measurement of routine respiration, 2 μg/ml oligomycin was added to measure the leak respiration state. The electron transfer system capacity was determined by titration of the uncoupler carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone (FCCP) in 0.5 μM steps until a maximum flow was reached. Respiration was inhibited by application of 0.5 μM rotenone and 2.5 μM antimycin A to determine non-mitochondrial residual oxygen consumption (ROX). All substrates and inhibitors used were obtained from Sigma-Aldrich. Oxygen concentration and oxygen flow per cell were recorded in 2 s intervals using the DatLab Software 5.1 (Oroboros Instruments Corp.). To compare mitochondrial respiratory states, all values measured were corrected for ROX. All experiments were performed using instrumental background correction and after calibration of the polarographic oxygen sensors.
Viability and caspase assays
Splenocytes were magnetically sorted with an AutoMACS (magnetic cell seperation) cell separator (Miltenyi Biotec GmbH, Bergisch Gladbach, Germany), using either the CD43 (Ly-48) or Pan T-Cell Isolation Kit II (Miltenyi Biotec GmbH). Cell viability was assessed with LIVE/DEAD Fixable Dead Cell Stain Kit Green/Violet (Invitrogen). Caspase-3/7, -8 and -9 activity was measured with the Caspase Glo Assay Systems (Promega Corporation, Madison, WI, USA). In short, 50 000 B cells were seeded in 100 μl culture medium (as described above) in white-walled 96-well plates (Fisher Scientific GmbH). One hundred microliters of Caspase Glo reagent (Promega Corporation), containing the proteasome inhibitor MG-132, was added to the wells, the plate was shaken on a plate shaker for 1 min at 300 r.p.m. and then incubated for 40 min at RT. Luminescence was measured in a GENios Pro plate reader (Tecan Group AG, Männedorf, Switzerland).
XBP-1 splicing
PCR primers 5′-ACACGCTTGGGAATGGACAC-3′ and 5′-CCATGGGAAGATGTTCTGGG-3′ encompassing the spliced sequences in xbp-1 mRNA were used for PCR amplification with Platinum Taq Super Mix (Invitrogen). PCR products were separated on a 2.5% agarose gel (Sigma-Aldrich) and visualized with ethidium bromide (Sigma-Adrich). Band density was analyzed with the ImageJ software (National Institutes of Health) and was expressed as the density of spliced XBP-1 divided by the density of spliced XBP-1 plus unspliced XBP-1, where no splicing corresponds to 0%.
Active EAE and immunization
Female WT and KO mice were injected subcutaneously with 200 μg MOG35–55 peptide (Biotrend Chemikalien GmbH, Köln, Germany) emulsified in Complete Freund’s Adjuvant (1 : 1 (v/v)), which consisted of Incomplete Freund’s Adjuvant supplemented with 5 mg/ml Mycobacterium tuberculosis H37RA (Difco Laboratories, Beckton, Dickinson, USA). For boosting, 200 ng Pertussis toxin (Sigma-Aldrich) was injected intraperitoneally on days 0 and 2 after immunization. From day 1, severity of the EAE was monitored and graded on a scale from 0 to 5: 0, no disease; 1, limp tail; 2, hind limb weakness; 3, hind limb paralysis; 4, hind and fore limb paralysis; 5, moribundity and death.
Immunoglobulin measurements
For immunizations, mice were injected intraperitoneally with 100 μg NPCG (Biosearch Technologies, Petaluma, CA, USA). Serum was collected from the peripheral blood and the immunoglobulin concentrations and NPCG-specific antibodies were determined by ELISA. For in vitro class switch experiments, splenocytes were cultured in triplicates of 300 000 treated with LPS (Sigma-Aldrich; 50 μg/ml)+IL4 (R&D Systems, Inc., Minneapolis, MN, USA; 20 ng/ml) for 4 days. The frequency of switched cells was measured by flow cytometry by gating on IgM− IgG1+ events. For each sample 200 000 events were acquired for analysis.
Statistical analysis
Data were analyzed as mean±S.D., and the statistical significance was analyzed using two-tailed t-tests or analysis of variance (ANOVA) with Tukey’s multiple comparison test as indicated.
Abbreviations
- BAK:
-
Bcl-2 homologous antagonist/killer
- BAX:
-
BCL2-associated X protein
- BI-1:
-
Bax inhibitor-1
- EAE:
-
experimental autoimmune encephalomyelitis
- ER:
-
endoplasmic reticulum
- FACS:
-
fluorescence-activated cell sorting
- FCCP:
-
carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone
- FO:
-
follicular
- Grp78/BiP:
-
glucose-regulated protein 78 kDa/binding immunoglobulin protein
- IP3:
-
inositol 1,4,5-trisphosphate
- IRE1:
-
inositol requiring enzyme 1
- KO:
-
knockout
- MZ:
-
marginal zone
- NADH:
-
nicotinamide adenine dinucleotide hydride
- NFAT:
-
nuclear factor of activated T cell
- NF-κB:
-
nuclear factor-κ light-chain enhancer of activated B cells
- NPCG:
-
4-hydroxy-3-nitrophenylacetyl coupled to chicken globulin
- ROX:
-
residual oxygen consumption
- SERCA:
-
sarcoplasmic/endoplasmic reticulum Ca2+-ATPase
- SOCE:
-
store-operated Ca2+ entry
- STIM1:
-
stromal interaction molecule 1
- UPR:
-
unfolded protein response
- WT:
-
wild type
- XBP-1:
-
X-box-binding protein-1
References
Berridge MJ, Bootman MD, Roderick HL . Calcium: calcium signalling: dynamics, homeostasis and remodelling. Nat Rev Mol Cell Biol 2003; 4: 517–529.
Demaurex N, Distelhorst CW . Cell biology. Apoptosis – the calcium connection. Science 2003; 300: 65–67.
Hetz C . The unfolded protein response: controlling cell fate decisions under ER stress and beyond. Nat Rev Mol Cell Biol 2012; 13: 89–102.
Henke N, Lisak DA, Schneider L, Habicht J, Pergande M, Methner A . The ancient cell death suppressor BAX inhibitor-1. Cell Calcium 2011; 50: 251–260.
Xu Q, Reed JC . Bax inhibitor-1, a mammalian apoptosis suppressor identified by functional screening in yeast. Mol Cell 1998; 1: 337–346.
Chae H-J, Kim H-R, Xu C, Bailly-Maitre B, Krajewska M, Krajewski S et al. BI-1 regulates an apoptosis pathway linked to endoplasmic reticulum stress. Mol Cell 2004; 15: 355–366.
Westphalen BC, Wessig J, Leypoldt F, Arnold S, Methner A . BI-1 protects cells from oxygen glucose deprivation by reducing the calcium content of the endoplasmic reticulum. Cell Death Differ 2005; 12: 304–306.
Bultynck G, Kiviluoto S, Henke N, Ivanova H, Schneider L, Rybalchenko V et al. The C terminus of Bax inhibitor-1 forms a Ca2+-permeable channel pore. J Biol Chem 2012; 287: 2544–2557.
Lisak DA, Schacht T, Enders V, Habicht J, Kiviluoto S, Schneider J et al. The transmembrane Bax inhibitor motif (TMBIM) containing protein family: Tissue expression, intracellular localization and effects on the ER CA(2+)-filling state. Biochim Biophys Acta 2015; 1853: 2104–2114.
Kiviluoto S, Luyten T, Schneider L, Lisak D, Rojas-Rivera D, Welkenhuyzen K et al. Bax Inhibitor-1-mediated Ca(2+) leak is decreased by cytosolic acidosis. Cell Calcium 2013; 54: 186–192.
Chang Y, Bruni R, Kloss B, Assur Z, Kloppmann E, Rost B et al. Structural basis for a pH-sensitive calcium leak across membranes. Science 2014; 344: 1131–1135.
Bailly-Maitre B, Bard-Chapeau E, Luciano F, Droin N, Bruey J-M, Faustin B et al. Mice lacking bi-1 gene show accelerated liver regeneration. Cancer Res 2007; 67: 1442–1450.
Bailly-Maitre B, Fondevila C, Kaldas F, Droin N, Luciano F, Ricci J-E et al. Cytoprotective gene bi-1 is required for intrinsic protection from endoplasmic reticulum stress and ischemia–reperfusion injury. Proc Natl Acad Sci USA 2006; 103: 2809–2814.
Lisbona F, Rojas-Rivera D, Thielen P, Zamorano S, Todd D, Martinon F et al. BAX inhibitor-1 is a negative regulator of the ER stress sensor IRE1alpha. Mol Cell 2009; 33: 679–691.
Kiviluoto S, Schneider L, Luyten T, Vervliet T, Missiaen L, De Smedt H et al. Bax inhibitor-1 is a novel IP3 receptor-interacting and -sensitizing protein. Cell Death Dis 2012; 3: e367.
Gilmore TD, Herscovitch M . Inhibitors of NF-kappaB signaling: 785 and counting. Oncogene 2006; 25: 6887–6899.
Weih DS, Yilmaz ZB, Weih F . Essential role of RelB in germinal center and marginal zone formation and proper expression of homing chemokines. J Immunol 2001; 167: 1909–1919.
Hövelmeyer N, Wunderlich FT, Massoumi R, Jakobsen CG, Song J, Wörns MA et al. Regulation of B cell homeostasis and activation by the tumor suppressor gene CYLD. J Exp Med 2007; 204: 2615–2627.
Guo F, Weih D, Meier E, Weih F . Constitutive alternative NF-kappaB signaling promotes marginal zone B-cell development but disrupts the marginal sinus and induces HEV-like structures in the spleen. Blood 2007; 110: 2381–2389.
Dolmetsch RE, Xu K, Lewis RS . Calcium oscillations increase the efficiency and specificity of gene expression. Nature 1998; 392: 933–936.
Dolmetsch RE, Lewis RS, Goodnow CC, Healy JI . Differential activation of transcription factors induced by Ca2+ response amplitude and duration. Nature 1997; 386: 855–858.
Pahl HL, Sester M, Burgert HG, Baeuerle PA . Activation of transcription factor NF-kappaB by the adenovirus E3/19K protein requires its ER retention. J Cell Biol 1996; 132: 511–522.
Pahl HL, Baeuerle PA . A novel signal transduction pathway from the endoplasmic reticulum to the nucleus is mediated by transcription factor NF-kappa B. EMBO J 1995; 14: 2580–2588.
Pahl HL, Baeuerle PA . Activation of NF-kappa B by ER stress requires both Ca2+ and reactive oxygen intermediates as messengers. FEBS Lett 1996; 392: 129–136.
Davies SP, Reddy H, Caivano M, Cohen P . Specificity and mechanism of action of some commonly used protein kinase inhibitors. Biochem J 2000; 351: 95–105.
Zhang SL, Yeromin AV, Zhang XH-F, Yu Y, Safrina O, Penna et al. Genome-wide RNAi screen of Ca(2+) influx identifies genes that regulate Ca(2+) release-activated Ca(2+) channel activity. Proc Natl Acad Sci USA 2006; 103: 9357–9362.
Prakriya M, Feske S, Gwack Y, Srikanth S, Rao A, Hogan PG . Orai1 is an essential pore subunit of the CRAC channel. Nature 2006; 443: 230–233.
White C, Li C, Yang J, Petrenko NB, Madesh M, Thompson CB et al. The endoplasmic reticulum gateway to apoptosis by Bcl-X(L) modulation of the InsP3R. Nat Cell Biol 2005; 7: 1021–1028.
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES et al. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 1997; 91: 479–489.
Sano R, Hou Y-CC, Hedvat M, Correa RG, Shu C-W, Krajewska M et al. Endoplasmic reticulum protein BI-1 regulates Ca2+-mediated bioenergetics to promote autophagy. Genes Dev 2012; 26: 1041–1054.
Zhong F, Harr MW, Bultynck G, Monaco G, Parys JB, De Smedt H et al. Induction of Ca2+-driven apoptosis in chronic lymphocytic leukemia cells by peptide-mediated disruption of Bcl-2-IP3 receptor interaction. Blood 2011; 117: 2924–2934.
Smith KG, Light A, Nossal GJ, Tarlinton DM . The extent of affinity maturation differs between the memory and antibody-forming cell compartments in the primary immune response. EMBO J 1997; 16: 2996–3006.
Suzuki J, Kanemaru K, Ishii K, Ohkura M, Okubo Y, Iino M . Imaging intraorganellar Ca2+ at subcellular resolution using CEPIA. Nat Commun 2014; 5: 4153.
Favre CJ, Schrenzel J, Jacquet J, Lew DP, Krause KH . Highly supralinear feedback inhibition of Ca2+ uptake by the Ca2+ load of intracellular stores. J Biol Chem 1996; 271: 14925–14930.
Mogami H, Tepikin AV, Petersen OH . Termination of cytosolic Ca2+ signals: Ca2+ reuptake into intracellular stores is regulated by the free Ca2+ concentration in the store lumen. EMBO J 1998; 17: 435–442.
Li Y, Camacho P . Ca2+-dependent redox modulation of SERCA 2b by ERp57. J Cell Biol 2004; 164: 35–46.
Hetz C, Bernasconi P, Fisher J, Lee A-H, Bassik MC, Antonsson B et al. Proapoptotic BAX and BAK modulate the unfolded protein response by a direct interaction with IRE1alpha. Science 2006; 312: 572–576.
Scorrano L, Oakes SA, Opferman JT, Cheng EH, Sorcinelli MD, Pozzan T et al. BAX and BAK regulation of endoplasmic reticulum Ca2+: a control point for apoptosis. Science 2003; 300: 135–139.
Oakes SA, Scorrano L, Opferman JT, Bassik MC, Nishino M, Pozzan T et al. Proapoptotic BAX and BAK regulate the type 1 inositol trisphosphate receptor and calcium leak from the endoplasmic reticulum. Proc Natl Acad Sci USA 2004; 102: 105–110.
Henke N, Albrecht P, Pfeiffer A, Toutzaris D, Zanger K, Methner A . Stromal interaction molecule 1 (STIM1) is involved in the regulation of mitochondrial shape and bioenergetics and plays a role in oxidative stress. J Biol Chem 2012; 287: 42042–42052.
Williams DA, Fay FS . Intracellular calibration of the fluorescent calcium indicator Fura-2. Cell Calcium 1990; 11: 75–83.
Sasaki Y, Derudder E, Hobeika E, Pelanda R, Reth M, Rajewsky K et al. Canonical NF-kappaB activity, dispensable for B cell development, replaces BAFF-receptor signals and promotes B cell proliferation upon activation. Immunity 2006; 24: 729–739.
Bultynck G, Szlufcik K, Kasri NN, Assefa Z, Callewaert G, Missiaen L et al. Thimerosal stimulates Ca2+ flux through inositol 1,4,5-trisphosphate receptor type 1, but not type 3, via modulation of an isoform-specific Ca2+-dependent intramolecular interaction. Biochem J 2004; 381: 87–96.
Acknowledgements
This work was supported by DFG ME1922/9-1 (to AM) and the Forschungskommission HHU (to AM and HHH).
Author contributions
DL, MG, AW, HHH, NH and AM designed the experiments with advice from GB, JBP and HdS. DL, TS, AG, PA, BK, MG, KZ, PAQ, MH and PA performed the experiments. DL, TS, BK, MG, KZ, NH and AM analyzed the data. DL, JCR and AM wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by C Borner
Supplementary Information accompanies this paper on Cell Death and Differentiation website
Supplementary information
Rights and permissions
About this article
Cite this article
Lisak, D., Schacht, T., Gawlitza, A. et al. BAX inhibitor-1 is a Ca2+ channel critically important for immune cell function and survival. Cell Death Differ 23, 358–368 (2016). https://doi.org/10.1038/cdd.2015.115
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cdd.2015.115
This article is cited by
-
Paradoxical implication of BAX/BAK in the persistence of tetraploid cells
Cell Death & Disease (2021)
-
DNA-PKcs promotes cardiac ischemia reperfusion injury through mitigating BI-1-governed mitochondrial homeostasis
Basic Research in Cardiology (2020)
-
Bax inhibitor-1 deficiency leads to obesity by increasing Ca2+-dependent insulin secretion
Journal of Molecular Medicine (2020)
-
Deoxynivalenol induces structural alterations in epidermoid carcinoma cells A431 and impairs the response to biomechanical stimulation
Scientific Reports (2018)