Abstract
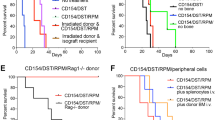
We examined the effects of intra-BM-BMT (IBM-BMT) plus adult thymus transplantation (ATT) from the same donor after 5.5 Gy sublethal irradiation (SubLI) or low-dose (3 × 106) BM cell injection (LDBMCI). With SubLI, BALB/c mice that had received 1 × 107 bone marrow cells by IBM-BMT plus ATT from B6 mice showed 73% donor chimerism, whereas those treated with IBM-BMT alone showed 45% chimerism. In the LDBMCI with 7Gy irradiation, IBM-BMT plus ATT resulted in a 90% survival rate with 90% chimerism, whereas IBM-BMT alone resulted in a 55% survival rate with 44% chimerism. Although the number of CD4 T cells was higher in IBM-BMT plus ATT than in IBM-BMT alone, the percentages of FoxP3+/CD4+ T cells and lymphocyte functions in the former were almost identical to those in the latter. When treated with IBM-BMT plus donor lymphocyte infusion (DLI), the mice showed a reduced survival time as a result of GVHD, with low numbers of FoxP3+CD4 T cells under either condition, although 100% chimerism was induced. These results suggest that IBM-BMT plus ATT is effective in reconstituting the recipients with donor-derived cells even after SubLI or LDBMCI.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- ATT:
-
adult thymus transplantation
- BMCs:
-
bone marrow cells
- BMT:
-
bone marrow transplantation
- DLI:
-
donor lymphocyte infusion
- GVHD:
-
graft-versus-host disease
- IBM:
-
intra-bone marrow
- LDBMCI:
-
low–dose bone marrow cell injection
- SubLI:
-
sublethal irradiation
References
Ikehara S . Bone marrow transplantation: a new strategy for intractable disease. Drugs Today 2002; 38: 103–111.
Ikehara S, Ohtsuki H, Good RA, Asamoto H, Nakamura T, Sekita K et al. Prevention of type I diabetes in non-obese diabetic mice by allogeneic bone marrow transplantation. Proc Natl Acad Sci USA 1985; 22: 7743–7747.
Yasumizu R, Sugiura K, Iwai H, Inaba M, Makino S, Ida T et al. Treatment of type 1 diabetes mellitus in non-obese diabetic mice by transplantation of allogeneic bone marrow and pancreatic tissue. Proc Natl Acad Sci USA 1987; 84: 6555–6557.
Than S, Ishida H, Inaba M, Fukuba Y, Seino Y, Adachi M et al. Bone marrow transplantation as a strategy for treatment of non-insulin-dependent diabetes mellitus in KK-Ay mice. J Exp Med 1992; 176: 1233–1238.
Ishida T, Inaba M, Hisha H, Sugiura K, Adachi Y, Nagata N et al. Requirement of donor-derived stromal cells in the bone marrow for successful allogeneic bone marrow transplantation. Complete prevention of recurrence of autoimmune diseases in MRL/MP-lpr/lpr mice by transplantation of bone marrow plus bones (stromal cells) from the same donor. J Immunol 1994; 152: 3119–3127.
Nakagawa T, Nagata N, Hosaka N, Ogawa R, Nakamura K, Ikehara S . Prevention of autoimmune inflammatory polyarthritis in male New Zealand black/KN mice by transplantation of bone marrow cells plus bone (stromal cells). Arthritis Rheum 1993; 36: 263–268.
Kane AB, Kumar V . Environmental and nutritional pathology. In: Kumar V, Abbas AK, Fausto N (eds). Pathologic Basis of Disease, 7th edn. Elsevier Saunders: Philadelphia, PA, 2004, pp. 415–468.
Petersdorf EW, Mickelson EM, Anasetti C, Martin PJ, Woolfrey AE, Hansen JA . Effect of HLA mismatches on the outcome of hematopoietic transplants. Curr Opin Immunol 1999; 11: 521–526.
Bachar-Lustig E, Rachamim N, Li HW, Lan F, Reisner Y . Megadose of T cell-depleted bone marrow overcomes MHC barriers in sublethally irradiated mice. Nat Med 1995; 1: 1268–1273.
Or R, Hadar E, Bitan M, Resnick IB, Aker M, Ackerstein A et al. Safety and efficacy of donor lymphocyte infusions following mismatched stem cell transplantation. Biol Blood Marrow Transplant 2006; 12: 1295–1301.
Kushida T, Inaba M, Takeuchi K, Sugiura K, Ogawa R, Ikehara Y . Treatment of intractable autoimmune disease in MRL/lpr mice using a new strategy for allogeneic bone marrow cells transplantation. Blood 2000; 95: 1862–1868.
Kushida T, Inaba M, Hisha H, Ichioka N, Esumi T, Ogawa R et al. Intra-bone marrow injection of allogeneic bone marrow cells: a powerful new strategy for treatment of intractable autoimmune diseases in MRL/lpr mice. Blood 2001; 97: 3292–3299.
Hosaka N, Nose M, Kyogoku M, Nagata N, Miyashima S, Good RA et al. Thymus transplantation, a critical factor for correction of autoimmune disease in aging MRL/+mice. Proc Natl Acad Sci USA 1996; 93: 8558–8562.
Hosaka N, Ryu T, Miyake T, Cui W, Nishida T, Takaki T et al. Treatment of autoimmune diseases in MRL/lpr mice by allogeneic bone marrow transplantation plus adult thymus transplantation. Clin Exp Immunol 2007; 147: 555–563.
Ryu T, Hosaka N, Miyake T, Cui W, Nishida T, Takaki T et al. Transplantation of newborn thymus plus hematopoietic stem cells can rescue supralethal irradiated mice. Bone Marrow Transplant 2008; 41: 659–666.
Miyake T, Hosaka N, Cui W, Nishida T, Takaki T, Inaba M et al. Adult thymus transplantation with allogeneic intra-bone marrow-bone marrow transplantation from same donor induces high thymopoiesis, mild graft-versus-host reaction and strong graft-versus-tumour effects. Immunology, (in press).
Markert ML, Boeck A, Hale LP, Kloster AL, McLaughlin TM . Transplantation of thymus tissue in complete DiGeorge syndrome. N Engl J Med 1999; 341: 1180–1189.
Markert ML, Hicks CB, Bartlett JA, Harmon JL . Effect of highly active antiretroviral therapy and thymic transplantation on immunoreconstitution in HIV infection. AIDS Res Hum Retroviruses 2000; 16: 403–413.
Hoffmann P, Ermann J, Edinger M, Fathman CG, Strober S . Donor-type CD4(+)CD25(+) regulatory T cells suppress lethal acute graft-versus-host disease after allogeneic bone marrow transplantation. J Exp Med 2002; 196: 389–399.
Nakamura K, Inaba M, Sugiura K, Yoshimura T, Kwon AH, Kamiyama Y et al. Enhancement of allogeneic hematopoietic stem cell engraftment and prevention of GVHD by intra-bone marrow bone marrow transplantation plus donor lymphocyte infusion. Stem Cells 2004; 22: 125–134.
Suzuki Y, Adachi Y, Minamino K, Zhang Y, Iwasaki M, Nakano K et al. A new strategy for treatment of malignant tumor: intra-bone marrow-bone marrow transplantation plus CD4- donor lymphocyte infusion. Stem Cells 2005; 23: 365–370.
Nguyen VH, Shashidhar S, Chang DS, Ho L, Kambham N, Bachmann M et al. The impact of regulatory T cells on T-cell immunity following hematopoietic cell transplantation. Blood 2008; 111: 945–953.
Miura Y, Thoburn CJ, Bright EC, Phelps ML, Shin T, Matsui EC et al. Association of Foxp3 regulatory gene expression with graft-versus-host disease. Blood 2004; 104: 2187–2193.
Miyara M, Sakaguchi S . Natural regulatory T cells: mechanisms of suppression. Trends Mol Med 2007; 13: 108–116.
Watanabe N, Wang YH, Lee HK, Ito T, Wang YH, Cao W et al. Hassall's corpuscles instruct dendritic cells to induce CD4+CD25+ regulatory T cells in human thymus. Nature 2005; 436: 1181–1185.
Nomura T, Sakaguchi S . Foxp3 and Aire in thymus-generated Treg cells: a link in self-tolerance. Nat Immunol 2007; 8: 333–334.
Abbas AK, Lichtman AH, Pillai S (eds). Cellular and Blood Immunology, 6th edn. Saunders: Philadelphia, PA, 2007, pp 153–188.
Cui W, Hosaka N, Miyake T, Wang X, Guo K, Cui Y et al. Analysis of Tolerance Induction using Triple Chimeric Mice: MHC-Disparate Thymus, Hemopoietic Cells and Microenvironment. Transplantation 2008; 85: 1151–1158.
Blackburn CC, Manley NR, Palmer DB, Boyd RL, Anderson G, Ritter MA . One for all and all for one: thymic epithelial stem cells and regeneration. Trends Immunol 2002; 23: 391–395.
Zhang L, Sun L, Zhao Y . Thymic epithelial progenitor cells and thymus regeneration: an update. Cell Res 2007; 17: 50–55.
Acknowledgements
This study was supported by a grant from ‘Haiteku Research Center’ of the Ministry of Education, a grant from the ‘Millennium’ program of the Ministry of Education, Culture, Sports, Science and Technology, a grant from the ‘Science Frontier’ program of the Ministry of Education, Culture, Sports, Science and Technology, a grant from the ‘The 21st Century Center of Excellence (COE)’ program of the Ministry of Education, Culture, Sports, Science and Technology, a grant-in-aid for scientific research (B) 11470062, grants-in-aid for scientific research on priority areas (A)10181225 and (A)11162221 and Health and Labor Sciences research grants (Research on Human Genome, Tissue Engineering Food Biotechnology) and also a grant from the Department of Transplantation for Regeneration Therapy (Sponsored by Otsuka Pharmaceutical Company, Ltd), a grant from Molecular Medical Science Institute, Otsuka Pharmaceutical Co., Ltd., a grant from Japan Immunoresearch Laboratories Co., Ltd (JIMRO) and a research grant B from Kansai Medical University.We thank Ms Y Tokuyama and Ms A Kitajima for their technical assistance, and Mr Hilary Eastwick-Field and Ms K Ando for their help in the preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nishida, T., Hosaka, N., Takaki, T. et al. Allogeneic intra-BM-BMT plus adult thymus transplantation from same donor has benefits for long-term survival even after sublethal irradiation or low-dose BM cell injection. Bone Marrow Transplant 43, 829–837 (2009). https://doi.org/10.1038/bmt.2008.396
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2008.396
Keywords
This article is cited by
-
Going straight to the point: intra-BM injection of hematopoietic progenitors
Bone Marrow Transplantation (2010)
-
Prolonged survival in mice with advanced tumors treated with syngeneic or allogeneic intra-bone marrow–bone marrow transplantation plus fetal thymus transplantation
Cancer Immunology, Immunotherapy (2010)