Abstract
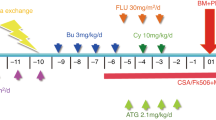
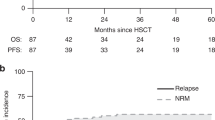
Allogeneic stem cell transplant is curative for haemophagocytic lymphohistiocytosis (HLH) and refractory Langerhans cell histiocytosis (LCH). However, patients frequently have significant pre-transplant morbidity and there is high TRM. Because HLH is caused by immune dysregulation, we surmised that a reduced-intensity conditioned (RIC) regimen might be sufficient for cure, while decreasing the TRM. In 2006, we reported the outcome of 12 patients treated with RIC SCT from a matched family/unrelated or haploidentical donor. Here we discuss the update of these patients, including a total of 25 patients treated with RIC SCT for HLH and three for LCH. Twenty-one of the twenty-five patients with HLH (84%) are alive and well with remission at a median of 36 months from SCT. Mortality included pneumonitis (n=3) and hepatic rupture (n=1). All three patients treated with RIC SCT for LCH remain alive and in remission at a median of 5.1 years from SCT. Seven of twenty-four survivors (one with LCH) have mixed chimerism but remain disease-free. These data are supported by other groups including 100% survival in seven patients with HLH and 78% survival of nine patients with LCH. In summary, RIC compares favourably with conventional SCT with long-term disease control in surviving patients with both HLH and LCL, despite a significant incidence of mixed chimerism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Janka GE . Familial hemophagocytic lymphohistiocytosis: diagnostic problems and differential diagnosis. Pediatr Hematol Oncol 1989; 6: 219–225.
Arico M, Janka G, Fischer A, Henter JI, Blanche S, Elinder G et al. Hemophagocytic lymphohistiocytosis. Report of 122 children from the International Registry. FHL Study Group of the Histiocyte Society. Leukemia 1996; 10: 197–203.
Eife R, Janka GE, Belohradsky BH, Holtmann H . Natural killer cell function and interferon production in familial hemophagocytic lymphohistiocytosis. Pediatr Hematol Oncol 1989; 6: 265–272.
Osugi Y, Hara J, Tagawa S, Takai K, Hosoi G, Matsuda Y et al. Cytokine production regulating Th1 and Th2 cytokines in hemophagocytic lymphohistiocytosis. Blood 1997; 89: 4100–4103.
Egeler RM, Shapiro R, Loechelt B, Filipovich A . Characteristic immune abnormalities in hemophagocytic lymphohistiocytosis. J Pediatr Hematol Oncol 1996; 18: 340–345.
Arico M, Imashuku S, Clementi R, Hibi S, Teramura T, Danesino C et al. Hemophagocytic lymphohistiocytosis due to germline mutations in SH2D1A, the X-linked lymphoproliferative disease gene. Blood 2001; 97: 1131–1133.
Ericson KG, Fadeel B, Andersson M, Gudmundsson GH, Gürgey A, Yalman N et al. Sequence analysis of the granulysin and granzyme B genes in familial hemophagocytic lymphohistiocytosis. Hum Genet 2003; 112: 98–99.
Feldmann J, Callebaut I, Raposo G, Certain S, Bacq D, Dumont C et al. Munc13-4 is essential for cytolytic granules fusion and is mutated in a form of familial hemophagocytic lymphohistiocytosis (FHL3). Cell 2003; 115: 461–473.
Goransdotter Ericson K, Fadeel B, Nilsson-Ardnor S, Söderhäll C, Samuelsson A, Janka G et al. Spectrum of perforin gene mutations in familial hemophagocytic lymphohistiocytosis. Am J Hum Genet 2001; 68: 590–597.
Henter JI . Biology and treatment of familial hemophagocytic lymphohistiocytosis: importance of perforin in lymphocyte-mediated cytotoxicity and triggering of apoptosis. Med Pediatr Oncol 2002; 38: 305–309.
Stepp SE, Dufourcq-Lagelouse R, Le Deist F, Certain S, Mathew PA, Henter JI et al. Perforin gene defects in familial hemophagocytic lymphohistiocytosis. Science 1999; 286: 1957–1959.
Stephan JL, Donadieu J, Ledeist F, Blanche S, Griscelli C, Fischer A et al. Treatment of familial hemophagocytic lymphohistiocytosis with antithymocyte globulins, steroids, and cyclosporin A. Blood 1993; 82: 2319–2323.
Blanche S, Caniglia M, Girault D, Landman J, Griscelli C, Fischer A et al. Treatment of hemophagocytic lymphohistiocytosis with chemotherapy and bone marrow transplantation: a single-center study of 22 cases. Blood 1991; 78: 51–54.
Imashuku S, Hibi S, Todo S, Inoue M, Kawa K, Koike K et al. Allogeneic hematopoietic stem cell transplantation for patients with hemophagocytic syndrome (HPS) in Japan. Bone Marrow Transplant 1999; 23: 569–572.
Horne A, Janka G, Maarten Egeler R, Gadner H, Imashuku S, Ladisch S et al. Haematopoietic stem cell transplantation in haemophagocytic lymphohistiocytosis. Br J Haematol 2005; 129: 622–630.
Minkov M, Grois N, Heitger A, Pötschger U, Westermeier T, Gadner H et al. Response to initial treatment of multisystem Langerhans cell histiocytosis: an important prognostic indicator. Med Pediatr Oncol 2002; 39: 581–585.
Broadbent V, Gadner H . Current therapy for Langerhans cell histiocytosis. Hematol Oncol Clin North Am 1998; 12: 327–338.
Gadner H, Grois N, Arico M, Broadbent V, Ceci A, Jakobson A et al. A randomized trial of treatment for multisystem Langerhans' cell histiocytosis. J Pediatr 2001; 138: 728–734.
Akkari V, Donadieu J, Piguet C, Bordigoni P, Michel G, Blanche S et al. Hematopoietic stem cell transplantation in patients with severe Langerhans cell histiocytosis and hematological dysfunction: experience of the French Langerhans Cell Study Group. Bone Marrow Transplant 2003; 31: 1097–1103.
Rao K, Amrolia PJ, Jones A, Cale CM, Naik P, King D et al. Improved survival after unrelated donor bone marrow transplantation in children with primary immunodeficiency using a reduced-intensity conditioning regimen. Blood 2005; 105: 879–885.
Cooper N, Rao K, Gilmour K, Hadad L, Adams S, Cale C et al. Stem cell transplantation with reduced-intensity conditioning for hemophagocytic lymphohistiocytosis. Blood 2006; 107: 1233–1236.
Vaughn G, Bleesing J, Jordan M, Marsh R, Fillipovich A . Hematopoietic cell transplantation with reduced intensity conditioning (RIC HCT) for hemophagocytic lymphohistiocytosis (HLH) and X-linked lymphoproliferative syndrome. Proceedings of the Histiocyte Society Meeting, Cambridge, September 2007, p 20 (abstract).
Steiner M, Matthes-Martin S, Attarbaschi A, Minkov M, Grois N, Unger E et al. Improved outcome of treatment-resistant high-risk Langerhans cell histiocytosis after allogeneic stem cell transplantation with reduced-intensity conditioning. Bone Marrow Transplant 2005; 36: 215–225.
Ardeshna KM, Hollifield J, Chessells JM, Veys P, Webb DK . Outcome for children after failed transplant for primary haemophagocytic lymphohistiocytosis. Br J Haematol 2001; 115: 949–952.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cooper, N., Rao, K., Goulden, N. et al. The use of reduced-intensity stem cell transplantation in haemophagocytic lymphohistiocytosis and Langerhans cell histiocytosis. Bone Marrow Transplant 42 (Suppl 2), S47–S50 (2008). https://doi.org/10.1038/bmt.2008.283
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2008.283
Keywords
This article is cited by
-
Sequential haplo-identical conditioning transplant regimen for pediatric patients with relapsed or refractory hemophagocytic lymphohistiocytosis
Bone Marrow Transplantation (2024)
-
Treatment Strategies for Central Nervous System Effects in Primary and Secondary Haemophagocytic Lymphohistiocytosis in Children
Current Treatment Options in Neurology (2022)
-
Reduced-Intensity/Reduced-Toxicity Conditioning Approaches Are Tolerated in XIAP Deficiency but Patients Fare Poorly with Acute GVHD
Journal of Clinical Immunology (2022)
-
Hematopoietic stem cell transplantation in children with Griscelli syndrome type 2: a single-center report on 35 patients
Bone Marrow Transplantation (2020)