Summary:
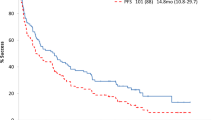
A prospective randomized trial was conducted to study the timing of high-dose intravenous melphalan and autologous stem cell transplantation (HDM/SCT) in AL amyloidosis. In all, 100 newly diagnosed patients were randomized to receive HDM/SCT, either as initial therapy (Arm-1) or following two cycles of oral melphalan and prednisone (Arm-2). The objectives of the trial were to compare survival and hematologic and clinical responses. With a median follow-up of 45 months (range 24–70), the overall survival was not significantly different between the two treatment arms (P=0.39). The hematologic response and organ system improvements after treatment did not differ between the two groups. Fewer patients received HDM/SCT in Arm-2 because of disease progression during the oral chemotherapy phase of the study, rendering them ineligible for subsequent high-dose therapy. This affected patients with cardiac involvement particularly, and led to a trend for an early survival disadvantage in Arm-2. Hence, newly diagnosed patients with AL amyloidosis eligible for HDM/SCT did not benefit from initial treatment with oral melphalan and prednisone, and there was a survival disadvantage for patients with cardiac involvement if HDM/SCT was delayed by initial oral chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kyle RA, Linos A, Beard CM et al. Incidence and natural history of primary systemic amyloidosis in Olmsted County, Minnesota, 1950 through 1989 (see comments). Blood 1992; 79: 1817–1822.
Simms RW, Prout MN, Cohen AS . The epidemiology of AL and AA amyloidosis. Baillieres Clin Rheumatol 1994; 8: 627–634.
Kyle RA, Bayrd ED . Amyloidosis: review of 236 cases. Medicine 1975; 54: 271–299.
Kyle RA, Greipp PR . Amyloidosis (AL). Clinical and laboratory features in 229 cases. Mayo Clin Proc 1983; 58: 665–683.
Kyle RA, Gertz MA . Primary systemic amyloidosis: clinical and laboratory features in 474 cases. Semin Hematol 1995; 32: 45–59.
Falk RH, Comenzo RL, Skinner M . The systemic amyloidoses (see comments). N Engl J Med 1997; 337: 898–909.
Dubrey SW, Cha K, Anderson J et al. The clinical features of immunoglobulin light-chain (AL) amyloidosis with heart involvement. Q J Med 1998; 91: 141–157.
Kyle RA, Greipp PR, Garton JP, Gertz MA . Primary systemic amyloidosis. Comparison of melphalan/prednisone vs colchicines. Am J Med 1985; 79: 708–716.
Kyle RA, Gertz MA, Greipp PR et al. A trial of three regimens for primary amyloidosis: colchicine alone, melphalan and prednisone, and melphalan, prednisone, and colchicine (see comments). N Engl J Med 1997; 336: 1202–1207.
Skinner M, Anderson J, Simms R et al. Treatment of 100 patients with primary amyloidosis: a randomized trial of melphalan, prednisone, and colchicine vs colchicine only. Am J Med 1996; 100: 290–298.
Merlini G . Treatment of primary amyloidosis. Semin Hematol 1995; 32: 60–79.
Gianni L, Bellotti V, Gianni AM, Merlini G . New drug therapy of amyloidoses: resorption of AL-type deposits with 4′-iodo-4′-deoxydoxorubicin. Blood 1995; 86: 855–861.
Gertz MA, Lacy MQ, Lust JA et al. Phase II trial of high-dose dexamethasone for previously treated immunoglobulin light-chain amyloidosis. Am J Hematol 1999; 61: 115–119.
Gertz MA, Lacy MQ, Lust JA et al. Prospective randomized trial of melphalan and prednisone vs vincristine, carmustine, melphalan, cyclophosphamide, and prednisone in the treatment of primary systemic amyloidosis. J Clin Oncol 1999; 17: 262–267.
Lokhorst HM, Sonneveld P, Wijermans P et al. Intermediate-dose melphalan (IDM) combined with G-CSF (filgrastim) is an effective and safe induction therapy for autologous stem cell transplantation in multiple myeloma. Br J Haematol 1995; 92: 44–48.
Attal M, Harousseau J-L, Stoppa A-M et al. A prospective, randomized trial of autologous bone marrow transplantation and chemotherapy in multiple myeloma. N Engl J Med 1996; 335: 91–97.
Barlogie B, Jagannath S, Vesole D, Tricot G . Autologous and allogeneic transplants for multiple myeloma. Semin Hematol 1995; 32: 31–44.
Barlogie B, Jagannath S, Vesole DH et al. Superiority of tandem autologous transplantation over standard therapy for previously untreated multiple myeloma. Blood 1997; 89: 789–793.
Sanchorawala V, Wright D, Seldin D et al. Use of high dose melphalan with autologous stem cell transplantation for the treatment of AL amyloidosis. Bone Marrow Transplant 2001; 28: 637–642.
Comenzo RL, Vosburgh E, Simms RW et al. Dose-intensive melphalan with blood stem cell support for the treatment of AL amyloidosis: one-year follow-up in five patients. Blood 1996; 88: 2801–2806.
Comenzo RL, Vosburgh E, Falk RH et al. Dose-intensive melphalan with blood stem-cell support for the treatment of AL (amyloid light-chain) amyloidosis: survival and responses in 25 patients. Blood 1998; 91: 3662–3670.
Comenzo RL, Sanchorawala V, Fisher C et al. Intermediate-dose intravenous melphalan and blood stem cells mobilized with sequential GM+G-CSF or G-CSF alone to treat AL (amyloid light chain) amyloidosis. Br J Haematol 1999; 104: 553–559.
Gillmore JD, Apperley JF, Craddock C et al. High-dose melphalan and stem cell rescue for AL amyloidosis. In: Kyle RA, Gertz MA (eds.). Amyloid and Amyloidosis 1998. Parthenon Publishing Group: New York, 1999, pp 102–104.
Majolino I, Marceno R, Pecoraro G et al. High-dose therapy and autologous transplantation in amyloidosis-AL. Haematologica 1993; 78: 68–71.
Gertz MA, Lacy MQ, Dispenzieri A . Myeloablative chemotherapy with stem cell rescue for the treatment of primary systemic amyloidosis: a status report. Bone Marrow Transplant 2000; 25: 465–470.
Gertz MA, Lacy MA, Gastineau DA et al. Blood stem cell transplantation as therapy for primary systemic amyloidosis (AL). Bone Marrow Transplant 2000; 26: 963–969.
Moreau P, Leblond V, Bourquelot P et al. Prognostic factors for survival and response after high-dose therapy and autologous stem cell transplantation in systemic AL amyloidosis: a report on 21 patients. Br J Haematol 1998; 101: 766–769.
Moreau P, Milpied N, de Faucal P et al. High-dose melphalan and autologous bone marrow transplantation for systemic AL amyloidosis with cardiac involvement (letter). Blood 1996; 87: 3063–3064.
Desikan KR, Dhodapkar MV, Hough A et al. Incidence and impact of light chain associated (AL) amyloidosis on the prognosis of patients with multiple myeloma treated with autologous transplantation. Leuk Lymphoma 1997; 27: 315–319.
van Gameren II, Hazenberg BP, Jager PL et al. AL amyloidosis treated with induction chemotherapy with VAD followed by high dose melphalan and autologous stem cell transplantation. Amyloid 2002; 9: 165–174.
Dember LM, Sanchorawala V, Seldin DC et al. Effect of dose-intensive intravenous melphalan and autologous blood stem-cell transplant on AL amyloidosis-associated renal disease. Ann Int Med 2001; 134: 746–753.
Choufani EB, Sanchorawala V, Ernst T et al. Acquired factor X deficiency in patients with AL amyloidosis: incidence, bleeding manifestations, and response to high-dose chemotherapy. Blood 2001; 97: 1885–1887.
Carr-Smith HD, Showell P, Bradwell AR . Antigen excess assessment of free light chain assays on the Dade–Behring BN™II nephelometer. Clin Chem 2002; 48: A-71.
Comenzo RL, Gertz MA . Autologous stem cell transplantation for primary systemic amyloidosis. Blood 2002; 99: 4276–4282.
Seldin DC, Choufani EB, Dember LM et al. Tolerability and efficacy of thalidomide for treatment of patients with AL amyloidosis. Clin Lymphoma 2003; 3: 241–246.
Rajkumar SV, Hayman S, Gerta MA et al. Combination therapy with thalidomide plus dexamethasone for newly diagnosed myeloma. J Clin Oncol 2002; 20: 4319–4323.
Weber D, Rankin K, Gavino M et al. Thalidomide alone or with dexamethasone for previously untreated multiple myeloma. J Clin Oncol 2003; 21: 16–19.
Acknowledgements
This work was supported by grants from the Food and Drug Administration (FD-R-001346), the National Institute of Health (HL 68705), the Gerry Foundation, and the Amyloid Research Fund at Boston University. Dr Seldin is a scholar of the Leukemia and Lymphoma Society of America. Results of this study were presented at the 43rd Annual Meeting of the American Society of Hematology. We gratefully acknowledge the numerous current and former colleagues in the Amyloid Treatment and Research Program, Clinical Trials Office and the Center for Cancer and Blood Disorders at Boston University Medical Center, who assisted with the multidisciplinary evaluation and treatment of the patients with AL amyloidosis. We thank Dr Michael LaValley for assistance with study design and randomization of participants.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sanchorawala, V., Wright, D., Seldin, D. et al. High-dose intravenous melphalan and autologous stem cell transplantation as initial therapy or following two cycles of oral chemotherapy for the treatment of AL amyloidosis: results of a prospective randomized trial. Bone Marrow Transplant 33, 381–388 (2004). https://doi.org/10.1038/sj.bmt.1704346
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704346
Keywords
This article is cited by
-
Clinical trials: design, endpoints and interpretation of outcomes
Bone Marrow Transplantation (2022)
-
Echocardiographic assessment of cardiac amyloidosis
Heart Failure Reviews (2022)
-
State-of-the-art review on management of end-stage heart failure in amyloidosis: transplant and beyond
Heart Failure Reviews (2022)
-
Depth of response prior to autologous stem cell transplantation predicts survival in light chain amyloidosis
Bone Marrow Transplantation (2021)
-
High-dose melphalan and stem cell transplantation in systemic AL amyloidosis in the era of novel anti-plasma cell therapy: a comprehensive review
Bone Marrow Transplantation (2019)