Abstract
Plants experience a highly variable light environment over the course of the day. To reveal the molecular mechanisms of their photosynthetic response to fluctuating light, we examined the role of two cyclic electron flows around photosystem I (CEF-PSI)—one depending on PROTON GRADIENT REGULATION 5 (PGR5) and one on NADH dehydrogenase-like complex (NDH)—in photosynthetic regulation under fluctuating light in rice (Oryza sativa L.). The impairment of PGR5-dependent CEF-PSI suppressed the photosynthetic response immediately after sudden irradiation, whereas the impairment of NDH-dependent CEF-PSI did not. However, the impairment of either PGR5-dependent or NDH-dependent CEF-PSl reduced the photosynthetic rate under fluctuating light, leading to photoinhibition at PSI and consequently a reduction in plant biomass. The results highlight that (1) PGR5-dependent CEF-PSI is a key regulator of rapid photosynthetic responses to high light intensity under fluctuating light conditions after constant high light; and (2) both PGR5-dependent and NDH-dependent CEF-PSI have physiological roles in sustaining photosynthesis and plant growth in rice under repeated light fluctuations. The highly responsive regulatory system managed by CEF-PSI appears able to optimize photosynthesis and plant growth under naturally fluctuating light conditions.
Similar content being viewed by others
Introduction
Plants experience a highly variable light environment over the course of the day on timescales of seconds, minutes, or hours owing to changes in leaf angle, cloud cover and canopy cover1. When light intensity exceeds the ability of leaves to use the light in photosynthesis, the excess light energy can lead to the formation of reactive oxygen species (ROS) and eventually to photoinhibition. Thus, plants need a highly responsive regulatory system to keep photosynthetic light reactions in balance with the needs and restrictions of the downstream metabolism. Efficient utilization of light energy through an optimized photosynthetic response under fluctuating light conditions is of ecological and agronomic interest.
Photosynthesis starts with the absorption of light by the light-harvesting systems, which drive photosynthetic electron transport through the thylakoid membranes of the chloroplasts2. Electrons derived from the splitting of water in photosystem II (PSII) ultimately reduce NADP+ to NADPH via photosystem I (PSI). This linear electron transport passes through the cytochrome (Cyt) b6/f complex, generating a proton gradient across the thylakoid membrane (ΔpH). Together with the protons deposited in the thylakoid lumen by the water-splitting complex associated with PSII, the protons translocated at the Cyt b6/f complex into the lumen enable ATP production by chloroplastic ATP synthase. NADPH and ATP generated by light reactions are then utilized in the Calvin–Benson cycle and other assimilatory reactions. The cyclic electron flow around PSI (CEF-PSI), which also passes through the Cyt b6/f complex, can also generate a ΔpH across the thylakoid membrane and also produces ATP but without any accumulation of NADPH in chloroplasts2.
CEF-PSI consists of two partly redundant pathways: the main pathway depends on the PGR5 (PROTON GRADIENT REGULATION 5) and PGRL1 (PGR5-LIKE PHOTOSYNTHETIC PHENOTYPE 1) proteins3,4, whereas the minor pathway is mediated by the chloroplast NADH dehydrogenase-like (NDH) complex5,6. Most likely, CEF-PSI involving PGR5 and PGRL1 proteins mainly mediates electron transport from ferredoxin to plastoquinone, contributes to ΔpH formation in photosynthesis and contributes to ATP supply for CO2 fixation2,4. The Arabidopsis pgr5 mutant grows similarly to the wild-type (WT) plants under constant low-light conditions, but is sensitive to constant high-light conditions, especially under low CO2 concentration7. Recent studies showed that fluctuating light induced photodamage in PSI and severely retarded the growth in pgr5 plants8,9,10. These results indicate that PGR5 is essential for survival under field conditions in Arabidopsis. In contrast, PGR5-knockdown (KD) rice lines showed a mild decline in CO2 assimilation and biomass production, although levels of ΔpH formation were decreased and the redox state of P700 were severely reduced11. Although the molecular function of PGR5 is likely conserved among plants, its physiological contribution to the regulation of photosynthesis might differ between Arabidopsis and rice.
On the other hand, chloroplast NDH forms a supercomplex with PSI and recycles electrons from ferredoxin to plastoquinone and subsequently to PSI2. NDH-defective tobacco and Arabidopsis mutants did not show any decrease in photosynthesis or growth under moderate growth conditions5,12. In addition, NDH-knockout Arabidopsis mutants did not show any growth suppression or reduction of photosynthetic performance even under fluctuating light, leading to the conclusion that NDH dependent CEF-PSI does not contribute to the response to fluctuating light9,10. However, NDH-deficient mutants showed mild sensitivity to various stresses, including strong light13, high temperature14 and low temperature15. These suggest that chloroplast NDH could act to alleviate oxidative stress in chloroplasts under excessive light2. However, the mutant phenotypes tested are mild and the mechanism by which chloroplast NDH might alleviate oxidative stresses is unclear because of the low rate of electron transport monitored in vivo and in isolated thylakoids16,17. The clear phenotype of NDH-deficient mutants is observed only when the PGR5-PGRL1 protein-dependent pathway is also impaired in double mutants12, indicating that chloroplast NDH may act as a safety valve when the stroma is highly reduced. However, the physiological function of NDH-dependent CEF around PSI remains to be clarified.
Since abiotic stresses limit crop productivity, understanding the physiological processes that underlie stress injury and the mechanisms of plant tolerance to the stresses is of immense importance to agriculture. The photosynthetic characteristics brought about by acclimation or adaptation to growth light levels have been studied intensively. Many plants acclimate to their growth light environment and alter their biochemical composition and morphology of leaves and whole plants18,19,20. Dynamic fluctuation of light intensity has recently become a subject of experimental research. However, the molecular mechanisms of photosynthetic responses to fluctuating light remain to be clarified. Here, we examined the physiological role of PGR5-dependent and NDH-dependent CEF-PSI in photosynthetic regulation and plant growth under fluctuating light in rice (Oryza sativa L.), a high-light-adaptive major crop. We measured gas exchange, chlorophyll fluorescence and P700+ reduction rate under fluctuating light. The results clearly show that both PGR5-defective and NDH-defective mutants suffered under fluctuating light, with PSI as the primary target of photodamage and had reduced plant growth. Therefore, not only PGR5-dependent but also NDH-dependent cyclic electron transport is essential for photoprotection of PSI under fluctuating light in rice.
Results
Photosynthetic components in PGR5-defective and NDH-defective mutants
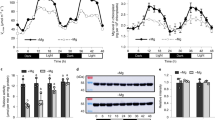
When plants were grown at a constant light intensity of 500 μmol photons m–2 s–2 with a 14-h photoperiod, contents of leaf nitrogen, rubisco and chlorophyll per unit leaf area and the chlorophyll-a/b ratio were similar among WT plants, PGR5 KD plants (defective in PGR5-dependent CEF-PSI pathway) and crr6 mutant plants (defective in NDH-dependent CEF-PSI pathway) (Table 1). A previous study confirmed that levels of PsaA (PSI reaction center), PsbA (PSII reaction center) and PsbS (pH sensor for qE induction) were unaffected in the PGR5 KD lines in rice11. We therefore consider that alterations in photosynthetic properties as a result of light treatments would primarily be the result of the stress due to defects in PGR5 and CRR6, rather than general growth defects.
Role of PGR5- and NDH-dependent cyclic electron transport in photosynthetic response under fluctuating light after constant high light
We simultaneously measured gas exchange, chlorophyll fluorescence and P700 redox state in WT plants under the initial cycle of fluctuating light after constant high light (Fig. 1). The electron transport rate through PSI (ETR I) and through PSII (ETR II) were estimated from Y(I) and Y(II), respectively, on the assumption that there are no changes in the accumulating ratio of PSI to PSII or their antenna sizes (see, Materials & Methods). High light after 10 min at low light increased ETR I and transiently induced non-photochemical quenching (NPQ), probably via the activation of CEF-PSI. Subsequently, ETR II was increased as NPQ relaxed. Then the CO2 assimilation rate increased with stomatal conductance.
Responses of photosynthetic parameters after changes in light intensity in rice WT plants (Oryza sativa cv. Nipponbare).
We simultaneously measured the electron transport rate around photosystem I (ETR I) and around photosystem II (ETR II), CO2 assimilation rate at a CO2 concentration of 400 μmol mol–1, stomatal conductance and non-photochemical quenching (NPQ). Leaves were first allowed to reach a steady rate of photosynthesis at 1500 μmol photons m–2 s–1 for 30 min. The photosynthetic parameters were recorded for 5 min, then the light intensity was reduced to 200 μmol photons m–2 s–1 for 10 min and then returned to 1500 μmol photons m–2 s–1 for 10 min. Values are means, n = 5 or 6.
Next, we analyzed the contribution of the two CEF-PSI pathways to photosynthetic response under the first two cycles of fluctuating light after constant high light in PGR5 KD plants and crr6 mutant plants. During the high-light phase, the responses of ETR I, ETR II and CO2 assimilation rate at a CO2 concentration of 400 μmol mol–1 were virtually identical between the crr6 mutants and the WT, whereas the fraction of PSI reaction centers that are closed due to acceptor side limitation, Y(NA), was significantly higher in the crr6 mutants (Fig. 2, Supplemental Fig. 1). On the other hand, during the low-light phase, the crr6 mutants had a lower NPQ and increased reduction (redox state) of the plastoquinone pool (high 1-qL) compared to the WT plants. ETR I, ETR II and CO2 assimilation rate responded to an increase in light intensity much more slowly in the PGR5 KD plants than in the WT plants. The PGR5 KD plants could not build up NPQ during the high-light phase and showed higher Y(NA) and 1-qL during both high-light and low-light phases. These results indicate that PGR5-dependent CEF-PSI is a key regulator of the rapid photosynthetic response to high light intensity under fluctuating light after constant high light, but the contribution of NDH-dependent CEF-PSI is small.
Time course of photosynthetic responses after changes in light intensity in PGR5-knockdown plants, crr6 mutant plants and WT plants.
The same fluctuating light regime as in Fig. 1 was used. We simultaneously measured the electron transport rate around PSI (ETR I) and around PSII (ETR II), CO2 assimilation rate at a CO2 concentration of 400 μmol mol–1, the fraction of PSI reaction centers that are closed owing to acceptor side limitation (Y(NA)), the redox state of the plastoquinone pool (1-qL) and non-photochemical quenching (NPQ). The graphs compare PGR5 KD plants with their WT (Oryza sativa cv. Nipponbare) and crr6 mutant plants with their WT (Oryza sativa cv. Hitomebore). Values are means ± SE, n = 5 or 6.
Role of PGR5- and NDH-dependent cyclic electron transport in photosynthesis under repeated light fluctuations
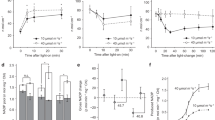
In WT plants, under constant high light, ETR I, ETR II and CO2 assimilation rate were constant over 5 h (Figs 3 and 4). On the other hand, under fluctuating light, ETR I, ETR II and CO2 assimilation rate during both low-light and high-light phases gradually decreased over 5 h. This decrease in both cultivars (‘Hitomebore’ and ‘Nipponbare’) indicates that the light fluctuation is stressful even for WT plants (Supplemental Fig. 2). The saturating pulses applied every 20 s during the measurements may have enhanced the sensitivity to high light to some extent.
Responses of photosynthetic parameters to either constant high-intensity or fluctuating light in PGR5-knockdown and WT (‘Nipponbare’) plants.
Photosynthetic parameters were monitored at a CO2 concentration of 400 μmol mol–1 under either constant high-intensity light (1500 μmol photons m–2 s–1) or fluctuating light (200 μmol m–2 s–1 for 10 min and 1500 μmol m–2 s–1 for 10 min) for 5 h. Abbreviations are the same as in Fig. 2. The responses of the quantum yield of PSI (Y(I)) and PSII (Y(II)) to fluctuating light were presented in Supplemental Fig. 3. Values are means ± SE, n = 5 or 6.
Responses of photosynthetic parameters to either constant high-intensity or fluctuating light in crr6 mutant and WT (‘Hitomebore’) plants.
Photosynthetic parameters were monitored at a CO2 concentration of 400 μmol mol–1 under either constant high-intensity light (1500 μmol photons m–2 s–1) or fluctuating light (200 μmol m–2 s–1 for 10 min and 1500 μmol m–2 s–1 for 10 min) for 5 h. Abbreviations are the same as in Fig. 2. The responses of the quantum yield of PSI (Y(I)) and PSII (Y(II)) to fluctuating light were presented in Supplemental Fig. 4. Values are means ± SE, n = 5 or 6.
Next, we evaluated the impact of the defects in PGR5- and NDH-dependent CEF-PSI on whole-chain electron transport under fluctuating light. Under constant high light, the PGR5 KD caused a large reduction in ETR I but a smaller reduction in ETR II and concomitant increases in 1-qL and Y(NA) and a reduction in NPQ (Fig. 3). Consequently, CO2 assimilation rate was decreased to the same extent as ETR II in the PGR5 KD plants. Under constant light, ETR I, ETR II and CO2 assimilation rate gradually decreased in the PGR5 KD plants over 5 h. Under fluctuating light, they decreased in a stepwise manner at every transition to low light, a pattern not seen in the WT plants (Fig. 3). The redox state of the plastoquinone pool (1-qL) of the PGR5 KD plants was increased, whereas NPQ was suppressed, during both low-light and high-light phases. The quantum yield of PSI (Y(I)) and PSII (Y(II)) showed a similar trend with ETR I and ETR II, respectively (Supplemental Fig. 3). The results indicate that during the high-light phases, the photosynthetic electron transport system accumulates reducing power that cannot be dissipated during the subsequent low-light phases, thus progressively decreasing the PSII yield and increasing the redox state of the plastoquinone pool in PGR5 KD plants.
Under constant high light in the crr6 mutants, ETR I, ETR II, CO2 assimilation rate and the other photosynthetic parameters (1-qL, NPQ, Y(NA)) were constant over 5 h and were virtually identical to those in the WT plants (Fig. 4). Under fluctuating light, ETR I, ETR II and CO2 assimilation rate decreased in a stepwise manner at every transition to low light in the crr6 mutant (Fig. 4), as in the PGR5 KD plants (Fig. 3). In contrast, in the crr6 mutants, 1-qL and Y(NA) increased in a stepwise manner at every transition to low light and were higher than in WT plants. As a result, photosynthetic capacity (i.e., ETR I, ETR II, CO2 assimilation rate) progressively decreased as the redox state of the plastoquinone pool increased, because the photosynthetic electron transport system of the crr6 plants accumulated reducing power that could not be dissipated before the subsequent high-light phases. This was supported by the data that Y(I) and Y(II) also showed a similar trend with ETR I and ETR II, respectively (Supplemental Fig. 4).
Stomatal conductance under fluctuating light was not significantly affected in either mutant line (Supplemental Fig. 5). These results suggest that the reduction in CO2 assimilation rate under the light fluctuations was due to the reduction in photosynthetic capacity due to the lack of PGR5- and NDH-dependent CEF-PSI.
Role of PGR5- and NDH-dependent cyclic electron transport in the alleviation of photoinhibition under fluctuating light conditions
We analyzed the effects of PGR5- and NDH-dependent CEF-PSI on photoinhibition. The maximum level of the P700 signal (Pm, full oxidation of P700) in the dark and the maximum quantum yield of PSII (Fv/Fm) were measured before and after treatment with constant high light (1500 μmol photons m–2 s–1) or fluctuating light (1500 μmol m–2 s–1 for 10 min and 200 μmol m–2 s–1 for 10 min) for 5 h. Before treatment, Pm and Fv/Fm were similar among PGR5 KD, crr6 mutant and WT plants. In the WT plants, constant high light significantly reduced only Fv/Fm, whereas fluctuating light reduced Fv/Fm and Pm, indicating that PSII is susceptible to constant high light, whereas PSI is more susceptible to fluctuating light than to constant high light (Fig. 5). In the PGR5 KD plants, constant high light decreased Fv/Fm by 16% and Pm by 48%. Fluctuating light decreased Fv/Fm by 17% and Pm by 67%. In the crr6 mutant plants, constant high light decreased Pm and Fv/Fm only slightly, indicating that chloroplast NDH does not function in the tolerance to constant high light. Fluctuating light decreased Fv/Fm by 21% and Pm by 39%. Like PGR5-dependent CEF-PSI, NDH-dependent CEF-PSI is required for alleviating photodamage of both photosystems by fluctuating light.
Effect of PGR5 knockdown or crr6 defect on alleviation of photoinhibition.
The maximum level of the P700 signal of PSI (Pm, full oxidation of P700) and the maximum quantum yield of PSII (Fv/Fm) were measured before and after treatment with constant high-intensity or fluctuating light for 5 h. Values were subsequently measured after dark incubation for 30 min. Light treatments were the same as in Figs 3 and 4. Values are means ± SE, n = 5 or 6. Significant differences among PGR5 KD plants and the WT plants are examined by Tukey–Kramer multiple comparison test (P < 0.05).
Role of PGR5- and NDH-dependent cyclic electron transport in plant growth under fluctuating light
We examined the effects of defects in PGR5- and NDH-dependent CEF-PSI on plant growth under fluctuating light. Constant light slightly suppressed growth of the PGR5 KD plants, but fluctuating light greatly suppressed it (Fig. 6). Constant light produced similar growth in crr6 mutant and WT plants, but fluctuating light reduced the final dry weight of crr6 mutant plants more than that of WT plants (Fig. 6).
Effect of PGR5 knockdown or crr6 defect on plant biomass production.
Plants were first grown under a constant PPFD of 500 μmol m–2 s–1 for 30 days after germination. They were then grown under either constant high-intensity light (800 μmol photons m–2 s–1) or fluctuating light (150 μmol m–2 s–1 for 10 min and 800 μmol m–2 s–1 for 10 min) for ≥50 days. The graphs compare PGR5-knockdown plants with their WT (‘Nipponbare’) and crr6 mutant plants with their WT (‘Hitomebore’). Values are means ± SE, n = 5 or 6. Significant differences among PGR5 KD plants, crr6 mutant and the WT plants are examined by Tukey–Kramer multiple comparison test (P < 0.05).
Discussion
Genetic analyses using Arabidopsis thaliana revealed that CEF-PSI consists of two redundant pathways2: the main pathway, which depends on PGR5-PGRL1 proteins and the minor pathway, which is mediated by the chloroplast NDH complex. Mutant phenotypes for CEF-PSI have been characterized only in Arabidopsis. However, the physiological role of CEF-PSI could depend on species, since evolution would have adapted the photosynthetic system to different light environments. Here, we examined the physiological consequences of defects in both PGR5 and NDH under fluctuating light in rice. The results clearly show that (1) PGR5-dependent CEF-PSI is a key regulator of a rapid photosynthetic response to high light under fluctuating light conditions and (2) although the contribution of NDH-dependent CEF-PSI is small, both PGR5-defective and NDH-defective mutants suffer from fluctuating light, with PSI as the primary target of photodamage and have reduced growth. Therefore, we conclude that both PGR5-dependent and NDH-dependent CEF-PSI have physiological roles in sustaining photosynthesis and growth of rice under fluctuating light (Fig. 7).
Role of PGR5-dependent and NDH-dependent CEF-PSI in photoprotection of PSI from photodamage under fluctuating light.
(A) In WT plants, under fluctuating light, the electron transport system is over-reduced and NPQ is developed via the activation of CEF-PSI, resulting in thermal dissipation of excess light energy. (B and C) In contrast, in both PGR5-defective and NDH-defective mutants, the electron transport system accumulates excess reducing power because it cannot dissipate heat. The cumulative strong reduction of the entire electron transport system under fluctuating light for a couple of hours would cause a strong reducing burst at the acceptor side of PSI, leading to photodamage at PSI. Thus, both PGR5-dependent and NDH-dependent CEF-PSI are essential for photoprotection of PSI under fluctuating light. Abbreviations: PSI/II, photosystem I/II; PQ, plastoquinone; Cyt b6/f, cytochrome b6/f complex; Fd, ferredoxin.
Photosynthetic induction occurring upon a sudden increase in light intensity after a prolonged period of low light or darkness has two main components based on the time scale of the processes1. The fast induction component is due mostly to the rapid light activation of RuBP regeneration, which is affected by photosynthetic electron transport. The second slower component is due to the light activation required for the primary carboxylation enzyme, Rubisco, combined with an increase in stomatal conductance. The mechanism of photosynthetic induction may depend on the period of low light or darkness before a sudden increase in light intensity. Our results clearly show that a sudden increase in light intensity during the fluctuating light conditions promoted ETR I and induced the rapid formation of ΔpH (i.e., NPQ) across the thylakoid membranes within 1 min (Fig. 1). Subsequently, ETR II was enhanced, with increases in CO2 assimilation rate and stomatal conductance. These processes need 5–10 min to be activated after illumination. Thus, it is obvious that the increase in ETR I during the first cycle of induction was faster than that of ETR II and CO2 assimilation rate (Fig. 1). Most likely, CEF-PSI functions immediately after a sudden increase in light intensity. In this period of photosynthetic induction, the build-up of ΔpH and the formation of NPQ would be essential not only for preventing photoinhibition, but also for activating Calvin cycle enzymes, including Rubisco activase21, because some enzymes of the Calvin cycle (i.e., fructose-1,6-bisphosphate phosphatase, sedoheptulose-1,7-bisphosphate phosphatase, ribulose-5-phosphate kinase, NADP-glyceraldehyde-3-phosphate dehydrogenase and Rubisco activase) are redox-regulated by thioredoxins using reduced ferredoxin produced by the electron transport reactions22.
Our results show that the crr6 mutation did not affect the response of ETR I, ETR II, or CO2 assimilation rate during the high-light phase under fluctuating light, leading to the conclusion that NDH-dependent CEF-PSI does not affect the photosynthetic response to an increase in light intensity under fluctuating light (Fig. 2). On the other hand, the PGR5 KD plants showed a much slower response of ETR II and CO2 assimilation rate following an increase in light intensity, since it caused a lower ETR I rate and thus NPQ was suppressed immediately after irradiation. Analysis of the direct impact of NPQ on photosynthetic CO2 assimilation during the induction phase in transgenic rice with altered levels of PsbS showed that the accumulation of PsbS and the resulting NPQ exerts control over photosynthesis under fluctuating light23. Thus, we conclude that PGR5-dependent CEF-PSI and the resulting lower NPQ control the photosynthetic response during the high-light phase under fluctuating light (Fig. 2). Alternative pathways, including the water–water cycle and CEF-PSI, may kick-start photosynthesis immediately after sudden light irradiation24. We propose that the PGR5-dependent CEF-PSI functions mainly to start photosynthesis during the high-light phase under fluctuating light via the rapid build-up of ΔpH across the thylakoid membranes and formation of NPQ.
In WT plants, during the high-light phase of fluctuating light (on the order of minutes), the electron transport system is over-reduced (high 1-qL) and NPQ is developed (Fig. 2), resulting in the thermal dissipation of excess light energy. On the other hand, during the low-light phase, the electron transport system is oxidized (low 1-qL) and NPQ is relaxed within minutes to allow the maximum photosynthetic electron transport during subsequent high light (Fig. 2). Nevertheless, ETR I, ETR II and CO2 assimilation rate showed gradual decreases over 5 h in WT plants after fluctuating light treatment (Figs 3 and 4). These plants also showed slight photoinhibition of PSI under the fluctuating light treatment (Fig. 5). Thus, PSI photoinhibition under fluctuating light could occur even in WT plants.
In contrast, the PGR5 KD plants are deficient in the development of ΔpH across the thylakoid membrane, since ETR I was greatly suppressed (Fig. 3). The PGR5 KD plants showed a stepwise increase in the redox state of the plastoquinone pool (high 1-qL) at every transition to the low-light phase. This increase indicates that during every high-light phase, the electron transport system of the PGR5 KD plants accumulates excess reducing power, which the system is not capable of dissipating as heat. The cumulative strong reduction of the entire electron transport system under fluctuating light would cause a strong reducing burst at the acceptor side of PSI, as indicated by Y(NA) (Fig. 3), eventually leading to the photoinhibition of PSI (Figs 5 and 7). As a result, the PGR5 KD plants decreased their CO2 assimilation rate via reductions in ETR I and ETR II (Fig. 3) and their growth was subsequently retarded under the fluctuating light conditions (Fig. 6). The similarity of these observations in rice PGR5 KD to those in Arabidopsis pgr5 mutants shows that plants with defects in PGR5 suffered from fluctuating light, with PSI as the primary target of photodamage and a stunted phenotype8,9. Therefore, the physiological function of the PGR5-dependent CEF pathway seems to be conserved among higher plants.
On the other hand, the NDH-defective mutant showed photosynthetic reactions almost identical to those in WT plants under constant high light (Fig. 4). However, the plastoquinone pool was reduced more (high 1-qL) at low light intensity than in the WT plants (Figs 2 and 4). The mutant showed a stepwise increase in the redox state of the plastoquinone pool at every transition to the low-light phase, as in the PGR5 KD plants. This increase indicates that during every high-light phase, the electron transport system of the NDH defective mutant accumulates excess reducing power (Fig. 4), which would cause photoinhibition of PSI (Figs 5 and 7), as in the PGR5 KD plants (Figs 3,5 and 7). As a result, the NDH-defective mutant decreased its CO2 assimilation rate via reductions in ETR I and ETR II (Fig. 4) and thus its growth, under fluctuating light (Fig. 6). Previous studies showed that even the complete absence of the NDH complex in Arabidopsis did not suppress growth under fluctuating light9,10. Thus, there is an interspecific difference between Arabidopsis and rice in the physiological function of NDH-dependent CEF-PSI. Although substantial progress has been made in understanding the structure and enzyme activity of the chloroplast NDH complex, the physiological significance of the complex remains to be clarified. Disturbed electron transfer parameters and reduced plant growth and grain production of an NDH-defective mutant of rice highlight the physiological significance of the NDH-dependent CEF-PSI under non-optimal growth conditions, including low temperature15 and low light25. Our results highlight the importance of NDH-dependent CEF-PSI under fluctuating light in rice.
In both the PGR5 KD plants and the NDH-defective mutant, the PSI reaction center remained fully reduced during the high-light phase, whereas in the WT plants, it became normally oxidized at transition to the high-light phase (Figs 3 and 4). This change implies that the photosynthetic machinery in the WT plants is capable of decreasing the electron flow to PSI during the high-light phase, which seems to be essential to preventing the over-reduction of PSI and to keeping the reduction level of the entire electron transport chain low enough during the subsequent low-light phase. In addition, when the low-light phase is followed by a high-light phase, rapid oxidation of the highly reduced electron transport chain (i.e., mainly the plastoquinone pool) in the two mutants exceeds the capacity of immediate PSI electron acceptors, as indicated by the PSI acceptor side limitation in these mutants (Figs 3 and 4). Such an excess of electrons could damage PSI under fluctuating light. Our results emphasize the importance of the PGR5-dependent and NDH-dependent CEF-PSI for sustaining photosynthesis and plant growth under fluctuating light in rice.
The physiological functions of state transition and PGR5-PGRL1-dependent CEF-PSI have been clarified in Arabidopsis under fluctuating light to represent natural conditions8,9,10. In particular, the PGR5-PGRL1-dependent CEF-PSI plays a central role in the regulation of linear electron transport via the downregulation of the Cyt b6/f complex26,27,28. It is still open for discussion how the PGR5-PGRL1 proteins regulate the rate of linear electron transport, since the exact molecular function of PGR5 and PGRL1 has been a topic of debate29. Here, we revealed similar phenotypes in the rice crr6 mutant and PGR5 KD rice under fluctuating light. This discrepancy with the phenotype in the Arabidopsis NDH-knockout mutants could be explained by the different physiological significance of NDH-dependent CEF-PSI in rice and Arabidopsis. Notably, a similar phenotype under fluctuating light was observed in two rice mutants defective in different CEF-PSI pathways. The most straightforward explanation for this observation is that both PGR5-PGRL1 and chloroplast NDH regulate the rate of linear electron transport by controlling the activity of the Cyt b6/f complex via CEF-PSI, as both pathways contribute to creating the proton motive force in the light30. It is also likely that both CEF-PSI pathways prevent the over-reduction of the stroma by balancing the ATP/NADPH production ratio in photosynthesis.
Materials & Methods
Plant materials and growth conditions
The rice crr6 mutant (defective in the OsCRR6 gene15) and its WT (Oryza sativa cv. Hitomebore) and the knockdown (KD) rice of the OsPGR5 gene11 and its WT (Oryza sativa cv. Nipponbare) were grown in soil in an environmentally controlled growth chamber. Each seedling was planted in a 1.3-L plastic pot with 1.0 g of a slow-release fertilizer (Temairazu; Co-op Chemical Co., Ltd., Tokyo, Japan). The chamber was operated with a day/night temperature of 28/23 °C, a relative humidity of 65%, a PPFD of 500 μmol m–2 s–1, a 14-h photoperiod and a CO2 concentration of 400 μmol mol–1.
For the analysis of growth under fluctuating light, plants grown as above for 30 days were transferred to either constant high light (800 μmol m–2 s–1) or fluctuating light (150 μmol m–2 s–1 for 10 min and 800 μmol m–2 s–1 for 10 min) for another 40 days.
Analysis of gas exchange, chlorophyll fluorescence and P700 measurements
Gas exchange, chlorophyll fluorescence and P700 redox state were measured simultaneously with a GFS-3000 and a Dual-PAM-100 measuring systems (Walz, Effeltrich, Germany) in the uppermost, fully expanded new leaves of 60- to 80-day-old plants as described15. After leaves were dark-adapted for 30 min, a saturating pulse was applied to obtain the maximum fluorescence and the maximum change in P700. Several photosynthetic parameters were measured every 20 s at a CO2 concentration of 400 μmol mol–1 under either constant high light or fluctuating light. The quantum yield of PSI (Y(I)) was calculated as Y(I) = 1 – Y(ND) – Y(NA), where Y(ND) corresponds to the fraction of P700 that is already oxidized by actinic light and Y(NA) corresponds to the fraction of P700 that are closed owing to acceptor side limitation. Since it has been reported that the signal of P700 is slightly affected by plastocyanin-dependent signal31, the parameters of P700 may be slightly affected although it should not be so significant. The quantum yield of photosystem II [Y(II) = (Fm′–F′)/Fm′], photochemical quenching [qP = (Fm′–F′)/(Fm′–Fo′)], non-photochemical quenching [NPQ = (Fm–Fm′)/Fm′] and the fraction of PSII centers in the open state (with plastoquinone oxidized) [qL = qP × (Fo′/F′)] were calculated. The electron transport rate (ETR) was calculated as ETR I (or ETR II) = 0.5 × abs I × Y(I) (or Y(II)), where 0.5 is the fraction of absorbed light reaching PSI or PSII and abs I is absorbed irradiance taken as 0.84 of incident irradiance.
Analysis of photoinhibition
The leaves were placed in a temperature-controlled chamber at a CO2 concentration of 400 μmol mol–1 and a relative humidity of 65% in the Dual-PAM-100 and GFS-3000 measuring systems. Photoinhibition was analyzed immediately after the measurements of photosynthesis under constant high light or fluctuating light (Figs 3 and 4). The leaves were exposed to constant high light (1500 μmol photons m–2 s–1) for 5 h or fluctuating light (high light at 1500 μmol m–2 s–1 for 10 min and low light at 200 μmol m–2 s–1 for 10 min) for 5 h. The maximum level of the P700 signal (full oxidation of P700) in the dark and the maximum quantum yield of PSII (Fv/Fm) after dark incubation for 30 min were measured before and after the 5 h light treatment.
Quantifications of photosynthetic components and Immunoblot analysis
Immediately after the measurements of gas exchange, leaf samples were taken, immersed in liquid nitrogen and stored at –80 °C. The frozen leaf samples were ground in liquid nitrogen and homogenized in an extraction buffer 12. Contents of leaf nitrogen, chlorophyll and rubisco were quantified12,25.
Additional Information
How to cite this article: Yamori, W. et al. A physiological role of cyclic electron transport around photosystem I in sustaining photosynthesis under fluctuating light in rice. Sci. Rep. 6, 20147; doi: 10.1038/srep20147 (2016).
References
Pearcy, R. W. Sunflecks and photosynthesis in plant canopies. Ann. Rev. Plant Physiol. Plant Mol. Biol. 41, 421–453 (1990).
Shikanai, T. Cyclic electron transport around photosystem I: genetic approaches. Annu. Rev. Plant. Biol. 58, 199–217 (2007).
Munekage, Y. et al. PGR5 is involved in cyclic electron flow around photosystem I and is essential for photoprotection in Arabidopsis. Cell 110, 361–371 (2002).
Hertle A. P. et al. PGRL1 is the elusive ferredoxin-plastoquinone reductase in photosynthetic cyclic electron flow. Mol. Cell 49, 511–523 (2013).
Shikanai, T. et al. Directed disruption of the tobacco ndhB gene impairs cyclic electron flow around photosystem I. Proc. Natl Acad. Sci. USA 95, 9705–9709 (1998).
Peng, L., Fukao, Y., Fujiwara, M. & Shikanai, T. Multistep assembly of chloroplast NADH dehydrogenase-like subcomplex A requires several nucleus-encoded proteins, including CRR41 and CRR42, in Arabidopsis. Plant Cell 24, 202–214 (2012).
Munekage, Y. N., Genty, B. & Peltier, G. Effect of PGR5 impairment on photosynthesis and growth in Arabidopsis thaliana. Plant Cell Physiol. 49, 1688–1698 (2008).
Tikkanen, M., Grieco, M., Kangasjärvi, S. & Aro, E. M. Thylakoid protein phosphorylation in higher plant chloroplasts optimizes electron transfer under fluctuating light. Plant Physiol. 152, 723–735 (2010).
Suorsa, M. et al. PROTON GRADIENT REGULATION5 is essential for proper acclimation of Arabidopsis photosystem I to naturally and artificially fluctuating light conditions. Plant Cell 24, 2934–2948 (2012).
Kono, M., Noguchi, K. & Terashima, I. Roles of the cyclic electron flow around PSI (CEF-PSI) and O2-dependent alternative pathways in regulation of the photosynthetic electron flow in short-term fluctuating light in Arabidopsis thaliana. Plant Cell Physiol. 55, 990–1004 (2014).
Nishikawa, Y. et al. PGR5-dependent cyclic electron transport around PSI contributes to the redox homeostasis in chloroplasts rather than CO2 fixation and biomass production in rice. Plant Cell Physiol. 53, 2117–2126 (2012).
Munekage, Y. et al. Cyclic electron flow around photosystem I is essential for photosynthesis. Nature 429, 579–582 (2004).
Endo, T., Shikanai, T., Takabayashi, A., Asada, K. & Sato, F. The role of chloroplastic NAD(P)H dehydrogenase in photoprotection. FEBS Letters 457, 5–8 (1999).
Wang, P. et al. Chloroplastic NAD(P)H dehydrogenase in tobacco leaves functions in alleviation of oxidative damage caused by temperature stress. Plant Physiol. 141, 465–474 (2006).
Yamori, W., Sakata, N., Suzuki, Y., Shikanai, T. & Makino, A. Cyclic electron flow around photosystem I via chloroplast NAD(P)H dehydrogenase (NDH) complex performs a significant physiological role during photosynthesis and plant growth at low temperature in rice. Plant J 68, 966–976 (2011).
Joliot, P. & Joliot, A. Quantification of cyclic and linear flows in plants. Proc. Natl Acad. Sci. USA 102, 4913–4918 (2005).
Okegawa, Y., Kagawa, Y., Kobayashi, Y. & Shikanai, T. Characterization of factors affecting the activity of photosystem I cyclic electron transport in chloroplasts. Plant Cell Physiol. 49, 825–834 (2008).
Smith, H. Light Quality, Photoperception and Plant Strategy. Annu. Rev. Plant. Physiol. 33, 481–518 (1982).
Terashima, I., Araya, T., Miyazawa, S.-I., Sone, K. & Yano, S. Construction and maintenance of the optimal photosynthetic systems of the leaf, herbaceous plant and tree: an eco-developmental treatise. Ann. Bot. 95, 507–19 (2005).
Yamori, W., Evans, J. R. & von Caemmerer, S. Effects of growth and measurement light intensities on temperature dependence of CO2 assimilation rate in tobacco leaves. Plant, Cell & Environ. 33, 332–343 (2010).
Yamori, W., Masumoto, C., Fukayama, H. & Makino, A. Rubisco activase is a key regulator of non-steady-state photosynthesis at any leaf temperature and, to a lesser extent, of steady-state photosynthesis at high temperature. Plant J. 71, 870–880 (2012).
Buchanan, B. B. & Balmer, Y. Redox regulation: a broadening horizon. Annu. Rev. Plant. Biol. 56, 187–220 (2005).
Hubbart, S., Ajigboye, O. O., Horton, P. & Murchie, E. H. The photoprotective protein PsbS exerts control over CO2 assimilation rate in fluctuating light in rice. Plant J. 71, 402–412 (2012).
Makino, A., Miyake, C. & Yokota, A. Physiological functions of the water-water cycle (Mehler reaction) and the cyclic electron flow around PSI in rice leaves. Plant Cell Physiol. 43, 1017–1026 (2002).
Yamori, W., Shikanai, T. & Makino, A. Photosystem I cyclic electron flow via chloroplast NADH dehydrogenease-like complex performs a physiological role for photosynthesis at low light. Sci. Rep. 5 13908 (2015).
Shikanai, T. Central role of cyclic electron transport around photosystem I in the regulation of photosynthesis. Curr. Opin. Biotech. 26, 25–30 (2014).
Tikkanen, M. & Aro, E. M. Integrative regulatory network of plant thylakoid energy transduction. Trends Plant Sci. 19, 10–17 (2014).
Allahverdiyeva, Y., Suorsa, M., Tikkanen, M. & Aro, E.-M. Photoprotection of photosystems in fluctuating light intensities. J. Exp. Bot. 66, 2427–2436 (2015).
Yamori, W. & Shikanai, T. Physiological Functions of Cyclic Electron Transport Around Photosystem I in Sustaining Photosynthesis and Plant Growth. Annu. Rev. Plant. Biol. 67 (2016) (in press).
Wang, C., Yamamoto, H. & Shikanai, T. Role of cyclic electron transport around photosystem I in regulating proton motive force. Biochim. Biophys. Acta 1847, 931–938 (2015).
Klughammer, C. & Schreiber, U. Analysis of light induced absorbance changes in the near-infrared region. I. Characterization of various components in isolated chloroplasts. Z Naturforsch 46c, 233–244 (1991).
Acknowledgements
This work was supported by the Japan Science and Technology Agency, PRESTO (to W.Y.) and in part by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (Scientific Research No. 25891005 to W.Y.) and by an NC-CARP/GRENE project (to A.M. and T.S.).
Author information
Authors and Affiliations
Contributions
W.Y., A.M. and T.S. designed the experiments. W.Y. mainly involved in all the experiments. W.Y. prepared the manuscript and W.Y., A.M. and T.S. contributed extensively to its finalization.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Yamori, W., Makino, A. & Shikanai, T. A physiological role of cyclic electron transport around photosystem I in sustaining photosynthesis under fluctuating light in rice. Sci Rep 6, 20147 (2016). https://doi.org/10.1038/srep20147
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep20147
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.