Abstract
Inflammatory myofibroblastic tumors are rare mesenchymal neoplasms frequently harboring oncogenic chromosomal rearrangements, most commonly, involving the ALK (anaplastic lymphoma kinase) gene. Treatment of this molecularly defined subgroup with the anaplastic lymphoma kinase inhibitor crizotinib has shown to be effective. However, comparable to lung adenocarcinoma, resistance inevitably develops. Second generation anaplastic lymphoma kinase inhibitors such as ceritinib are able to overcome acquired resistance to crizotinib. Here, we report the case of a patient with an inflammatory myofibroblastic tumors harboring a DCTN1-ALK fusion who developed resistance to crizotinib treatment. Next-generation sequencing of a rebiopsy sample revealed the acquisition of the ALK G1269A mutation as a mechanism of resistance. Therapy with ceritinib resulted in a short but profound clinical, metabolic and morphologic response. This case illustrates that (i) different tumor entities may share similar oncogenic driver mechanisms, rendering them vulnerable for the same therapeutic substances and (ii) likewise, the same mode of resistance may occur under targeted therapy among different tumor entities.
Similar content being viewed by others
Introduction
Inflammatory myofibroblastic tumors (IMTs) are rare mesenchymal neoplasms.1 IMTs predominantly affect young adults and prognosis is favorable upon surgical resection. Metastastic or recurrent disease is rare but is associated with poor prognosis most notably due to the lack of an effective systemic standard treatment.
IMTs are molecularly characterized by chromosomal rearrangements, most notably fusions of ALK (50% of cases), ROS1 (ROS proto-oncogene 1) or PDGFRβ (platelet derived growth factor receptor beta).2 Efficacy of the kinase inhibitor crizotinib is proven in ALK-positive non-small cell lung cancer (NSCLC) and IMTs.3, 4 Second-generation ALK inhibitors are effective in crizotinib-resistant ALK-positive NSCLC, including cases with acquired ALK G1269A mutations.5,6,7
Report
A 36-year-old female patient was first diagnosed in December 2009 with an ALK-rearranged pulmonary IMT (UICC stage: pT2 pN0 cM0 G2). Left pneumonectomy was performed (R0) but recurrence occurred in July 2011. Crizotinib treatment was initialized in October 2011 and resulted in a good radiologic response lasting until March 2014.
At time of progression the patient presented with chills, night sweats, thoracic pain and significant elevation of serum C-reactive protein (CRP). Infection was ruled out. A rebiopsy confirmed the ALK fusion. Massively parallel (MPS) and hybrid-capture sequencing identified dynactin subunit 1 gene (DCTN1) as fusion partner and revealed the acquisition of an ALK G1269A mutation (confirmation by Sanger sequencing; Fig. 1). No other mutations were detected. Baseline 18FDG-PET/CT scan showed local progression and the appearance of new thoracic lesions (sum of target lesions: 122 mm; SUVmax: 29.8; Fig. 2). Treatment with ceritinib was initiated in June 2014 (750 mg daily). Tumor-related symptoms resolved rapidly and serum CRP levels diminished. PET/CT at treatment day 17 revealed a metabolic response with a decrease of SUVmax by 68% and a reduction of the sum of target lesions by 25% (91 mm; SD). PET/CT at day 63 showed stable SUVmax and a reduction of target lesions by 30% (86 mm; unconfirmed partial response; Fig. 2). Subsequently CRP levels rose and tumor related symptoms recurred. Restaging revealed a progression according to RECIST with new lesions and an increased FDG-uptake (day 110).
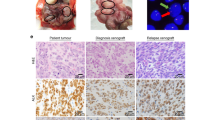
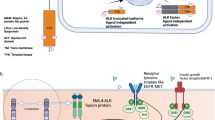
a1–a3 Hematoxylin and eosin stained section of IMT. b Fluorescence in situ hybridization of post-crizotinib sample tissue, confirming rearrangement of ALK in 2p23: Isolated red signals (arrows) indicate loss of the 5' region of ALK. Orange fusion signals of the red 3' probe and the green 5' probe indicate normal ALK alleles. c Sanger sequencing of ALK of the initial tumor sample (C1) and the post-crizotinib sample (C2) identifying the ALK G1269A resistance mutation. d Schematic illustration of the DCTN1-ALK fusion. Exons 20–29 of the ALK gene, which contain the tyrosine kinase domain, are fused with exons 1–26 of the DCTN1 gene. Both genes are located on chromosome 2p. The 5' region of ALK was apparently lost as indicated by the absence of the green FISH signal
Metabolic response evaluation and CRP levels. a Baseline 18FDG-PET/CT and follow-up scans showing metabolic response with decreasing uptake. b Non-linear chart of serum CRP levels (mg/L) (blue graph) and change of SUVmax in the single hottest lesion (red columns). Gray arrows: ceritinib treatment at daily doses indicated (mg); blue graph: CRP levels (mg/L); red columns: SUVmax of the hottest lesion
The patient was discontinued from ceritinib at day 111 and deceased in February 2015.
Discussion
To the best of our knowledge, this is the first report describing a molecular mechanism of acquired resistance to crizotinib in ALK-rearranged IMTs. DNA sequencing revealed the acquisition of ALK G1269A as the potential driver of resistance. All other investigated genes showed wild-type DNA sequences, highlighting the pivotal role of the ALK rearrangement in this case.
In our patient, treatment with ceritinib proved to be effective, however, for a short time only. Nevertheless, our case represents proof-of-concept for efficacy of second-generation ALK inhibitors in ALK-rearranged IMTs with acquired resistance to crizotinib. Several mutations, including ALK G1269A, confer resistance to crizotinib in ALK-positive NSCLC and the next-generation ALK inhibitors ceritinib, alectinib, lolartinib, and brigatinib are able to overcome ALK G1269A-driven resistance.5,6,7 At time of treatment of our patient, only ceritinib was available within an individual IND. Rebiopsy upon progression to ceritinib was not feasible and at that time third-generation inhibitors were not available.
In pre-clinical assays different ALK fusion partners and EML4-ALK fusion variants have shown to influence sensitivity to crizotinib.8 Whether, DCTN1 which we identified as a novel translocation partner of ALK in IMTs has a negative impact on efficacy or duration of ALK inhibition is unclear.
CRP levels correlated inversely with tumor response and the elevation of inflammatory serum markers and their normalization following resection has regularly been observed in IMTs.9 CRP may therefor be a potential serum marker to follow treatment response.
This case illustrates how similar modes of resistance to ALK inhibitor treatment may occur in epithelial and mesenchymal malignancies, rendering them vulnerable to the same drugs. Although identical molecular targets do not generally confer equal sensitivity to the appropriate inhibitors in different tumors, our case raises the hope that the growing repertoire of targeted therapeutics will be effective against classes of malignancies defined by molecular alterations.
Patient and methods
The patient was treated with ceritinib within an individual patient treatment program after the collection of the written informed consent.
Metabolic response was assessed using 18FDG-PET/CT according to the PET response criteria in solid tumors (PERCIST) v1.0 guideline, comparing SUVmax of the hottest single lesion in each consecutive scan (metabolic response definition: reduction in SUVmax of ≥30%). Morphologic response was assessed according to RECIST v1.1.
Fluorescence in situ hybridization was performed using ALK-specific dual-color break-apart probes (TriCheck®, Zytovision, Bremerhaven, Germany). MPS for ALK exons 21–25 and 13 more genes was performed on a MiSeq platform (Illumina, San Diego, USA).10 The hybrid-capture sequencing panel 'NEOplus' was used to test for point mutations, small insertions/deletions, copy number alterations, and fusions in 72 genes and to characterize the ALK rearrangement (NEO New Oncology AG, Cologne, Germany).
References
Coffin, C., Watterson, J., Priest, J. & Dehner, L. P. Extrapulmonary inflammatory myofibroblastic tumor: a clinicopathologic and immunohistochemical study of 84 cases. Am. J. Surg. Pathol. 19, 859–872 (1995).
Lovly, C. M. et al. Inflammatory myofibroblastic tumors harbor multiple potentially actionable kinase fusions. Cancer Discov. 4, 889–895 (2014).
Shaw, A. et al. Crizotinib versus chemotherapy in advanced ALK-positive lung cancer. N. Engl. J. Med. 368, 2385–2394 (2013).
Butrynski, J. et al. Crizotinib in ALK-rearranged inflammatory myofibroblastic tumor. N. Engl. J. Med. 363, 1727–1733 (2010).
Shaw, A. T. et al. Ceritinib in ALK-rearranged non-small-cell lung cancer. N. Engl. J. Med. 370, 1189–1197 (2014).
Ou, S. H. I. et al. Alectinib in crizotinib-refractory ALK-rearranged non–small-cell lung cancer: a phase II global study. J. Clin. Oncol. 34, 661–668 (2016).
Zou, H. Y. et al. PF-06463922, an ALK/ROS1 inhibitor, overcomes resistance to first and second generation ALK inhibitors in preclinical models. Cancer Cell 28, 70–81 (2015).
Heuckmann, J. M. et al. Differential protein stability and ALK inhibitor sensitivity of EML4-ALK fusion variants. Clin. Cancer Res. 18, 4682–4690 (2012).
Mergan, F. et al. Inflammatory myofibroblastic tumor in children: clinical review with anaplastic lymphoma kinase, Epstein-Barr virus, and human herpesvirus 8 detection analysis. J. Ped. Surg. 40, 1581–1586 (2005).
König, K. et al. Implementation of amplicon parallel sequencing leads to improvement of diagnosis and therapy of lung cancer patients. J. Thorac. Oncol. 10, 1049–1057 (2015).
Acknowledgements
Ceritinib was kindly proved by Novartis within an individual IND.
Author information
Authors and Affiliations
Contributions
S.M., A.H.S., T.W., R.B., J.W. contributed equally to this work and wrote the manuscript. S.M., J.W., T.W., M.S., R.F. were involved in patient treatment and interpretation of the treatment data. A.H.S., A.M.S., R.B., S.M.B., L.H., R.M., H.P., C.H. performed the molecular and pathological analyses and interpretation of the data. M.P. and C.K. performed imaging acquisition and interpretation of data.
Corresponding author
Ethics declarations
Competing interests
S.M. and J.W. received honoraria for consultancy by Novartis and Pfizer. R.M. is employee at NEO New Oncology AG.J.M.H. and L.H. are shareholders and full-time and part-time employees at NEO New Oncology. All other authors have declared no competing interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Michels, S., Scheel, A.H., Wündisch, T. et al. ALK G1269A mutation as a potential mechanism of acquired resistance to crizotinib in an ALK-rearranged inflammatory myofibroblastic tumor. npj Precision Onc 1, 4 (2017). https://doi.org/10.1038/s41698-017-0004-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-017-0004-3
This article is cited by
-
New perspectives for targeting therapy in ALK-positive human cancers
Oncogene (2023)
-
Update of Diagnosis and Targeted Therapy for ALK+ Inflammation Myofibroblastic Tumor
Current Treatment Options in Oncology (2023)
-
Spindle cell/sclerosing rhabdomyosarcoma with DCTN1::ALK fusion: broadening the molecular spectrum with potential therapeutic implications
Virchows Archiv (2022)
-
ALK expressed in a gastrointestinal stromal tumor harboring PDGFRA p. D842V mutation:a case report
Diagnostic Pathology (2020)