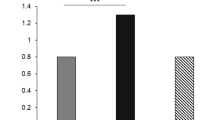Abstract
It has been reported that subclinical hypothyroidism (SCH) is closely related to subclinical atherosclerosis. According to the impact of SCH on noninvasive markers of cardiovascular risk, we fulfilled a meta-analysis of included studies to provide an integrated overview. We searched electronic databases and included all relevant studies involving SCH and epicardial adipose tissue (EAT), carotid intima-media thickness (CIMT), pulse wave velocity (PWV), flow-mediated dilation (FMD) and glyceryl trinitrate-induced dilation (GNT- induced dilation). The result was calculated in a meta-analysis to assess the impact of SCH on these markers. A total of 27 studies were entered in the final analysis. Compared with euthyroid subjects, SCH patients exhibited a significantly increased CIMT (SMD: 0.369 mm; 95%CI: 0.038, 0.700; P = 0.029) and EAT (SMD: 1.167 mm; 95%CI: 0.869, 1.466; P = 0.000) and increased PWV (SMD: 3.574 m/s; 95%CI: 0.935, 6.213, P = 0.008). We also found significantly lower FMD (SMD: −1.525%, 95%CI: −2.156, −0.894, P = 0.000) and lower GNT-induced dilation (SMD: −0.384%, 95%CI: −0.625, −0.142, P = 0.002). Sensitivity analysis and subgroup analysis confirmed the above results. Our meta-analysis confirmed a significant association of SCH and cardiovascular risk with arterial wall thickening and stiffening and endothelial dysfunction. These findings will help to establish detailed cardiovascular prevention strategies for SCH patients.
Similar content being viewed by others
Introduction
Subclinical hypothyroidism (SCH) is a disease characterized by the absence of distinct clinical symptoms and signs; laboratory examination of patients with SCH have revealed elevated serum levels of thyroid-stimulating hormone (TSH) with normal serum free thyroxine concentrations1. According to the level of TSH, SCH can be divided into two types: TSH levels less than 10 mIU/L (normal thyroxine) is considered mild SCH, whereas TSH levels greater than or equal to 10 mIU/L is considered severe SCH2,3. The United States National Health and Nutrition Survey noted that among the population >12 years of age, considering 4.5 mIU/L as the upper limit of normal TSH levels corresponded to a SCH prevalence rate of 4.3%4. According to the Colorado survey, considering 5.0 mIU/L as the upper limit of normal TSH levels corresponded to a SCH prevalence of 8.5%1.
Most patients with SCH do not exhibit clinical symptoms, but SCH can exert a detrimental effect on the cardiovascular (CV) system. The major adverse effects of SCH include reduce cardiac diastolic function and accelerated atherosclerosis5, impaired endothelial dysfunction6, increased carotid artery intima-media thickness (CIMT)7, induced coronary artery disease and increased mortality of coronary heart disease8,9. Studies have shown that abnormal lipid metabolism and oxidative stress are involved in the occurrence of these adverse effects7,10. To further understand the adverse effects induced by SCH, a growing number of studies have focused on CV with disease-related indicators to evaluate the correlation between SCH and atherosclerosis.
CIMT can be measured by noninvasive ultrasound imaging to evaluate subclinical atherosclerosis11,12. CIMT has been widely accepted as a formidable predictor of adverse CV disease events (shock and myocardial infarction)13,14. Similarly, flow-mediated dilation (FMD), pulse wave velocity (PWV), and glyceryl trinitrate-induced dilation (GNT-induced dilation) as independent predictors of CV events are considered to be surrogate markers of subclinical atherosclerosis15,16,17,18. FMD and GNT-induced dilation are widely used to detect vascular endothelial function as a noninvasive and accurate detection technique19. In addition, PWV has been used to detect peripheral and central arterial stiffness20. The epicardial adipose tissue (EAT) thickness surveyed by echocardiography is also considered an important independent predictor of an increased incidence of CV events for evaluating subclinical coronary atherosclerosis21.
For the past few years, many case-control studies have announced that SCH patients exhibit accelerated atherosclerosis, damaged endothelial function, and increased arterial stiffness. However, small sample sizes and potential confounding factors influence the strength of previous evidence. According to the impact of SCH on noninvasive markers of CV risk, we fulfilled a systematic review and meta-analysis of included studies to provide an integrated overview.
Methods
We strictly followed the PRISMA reporting specifications for this meta-analysis.
Search strategy
According to the PRISMA guidelines22, we formulated a detailed search strategy, including SCH and noninvasive markers of CV risk (i.e., IMT, EAT, PWV, FMD, and GNT-induced dilation). We systematically searched online databases (PubMed, Embase, Web of Science) through 30 November 2017. All possible combinations of search terms included (‘Subclinical hypothyroidism’) AND (‘atherosclerosis’) AND (‘arterial stiffness’) AND (epicardial adipose tissue OR carotid intima-media thickness OR pulse wave velocity OR flow-mediated dilation OR glyceryltrinitrate-induced dilation). No language or publication date restrictions were imposed in this study.
Furthermore, manual audits were conducted on all retrieved articles. If data were missing from an article, the authors communicated via e-mail to try to obtain the raw data. Two independent authors (YAO K and ZOU X) analysed each paper and independently extracted the data. In the case of differences in opinion, the two authors consulted with a third investigator (ZHAO T). The differences were settled by accordance. The selected results were determined by unanimous agreement of each author and were reported according to the PRISMA guidelines.
Inclusion criteria
Studies adhering to the following inclusion criteria were included: 1) case-control studies; 2) including subjects with normal thyroid function as a control group, 3) CIMT, EAT, PWV, FMD and GNT-induced dilation indicators of patients with SCH compared with euthyroid (EU) subjects reported; and 4) 95% confidence intervals (CIs) were reported.
Exclusion criteria
During the literature screening process, studies characterized by the following criteria were excluded: 1) including participants diagnosed with severe SCH or hyperthyroidism; 2) including participants being treated with medicine; 3) including patients with abnormal thyroid function who were diagnosed thyroid disease; 4) studies without a control group, animal studies, and reviews; 5) studies that did not provide the value of at least one variable (mean and standard deviation) among the predictors.
Data extraction and quality assessment
For each included study, we extracted the data corresponding to the demographic variables, the number of cases and controls, and the major clinical variables including CIMT, FMD, GNT-induced dilation, PWV and EAT.
Next, the methodological quality of each study was evaluated based on the characteristics of the study. The Newcastle Ottawa scale (NOS) was used to assess the quality of nonrandomized observational studies23. The scoring system consisted of three major areas (selection, comparability, exposure), and the results ranged from 0 to 8 with higher scores representing better methodological quality. The quality evaluation results of the NOS are reported in Table 1.
Statistical analysis and risk of bias assessment
Statistical analysis was performed by STATA 12.0 software. Differences among cases and controls were presented as the standardized mean difference (SMD) with the associated 95%CI for continuous variables24.
All reported P values were two-sided with a significance level set at P < 0.05. Heterogeneity between studies was calculated by I2 statistics; an I2 value of 0% indicates no heterogeneity, and I2 values of 25%, 25–50%, and 50% indicate low, moderate, and high heterogeneity, respectively25.
Begg’s test, Egger’s test and funnel plots were used to test publication bias. We visually examined the symmetry of the funnel plots to evaluate possible small sample effects, and we used Begg’s test and Egger’s test to evaluate publication bias of the included studies. Statistical significance was considered for P < 0.1026. When publication bias occurred, the adjusted effect scale was assessed using the Duval and Tweedie’s trim and fill method with the random-effect model27.
Considering the variability among the studies, we used a random-effect method for all analyses.
Sensitivity analyses
Sensitivity analysis is used to investigate the reliability of a meta-analysis. We evaluated the reliability of the conclusions by examining the impact of individual studies on the total merged effect. For each study, a new meta-analysis was performed upon deleting that study to assess the stability of the results compared with the total effect.
Subgroup analysis
Taking into account the potential impact of SCH confounding factors on the outcomes of the study, we conducted subgroup analyses for smoking, obesity, NOS score, and national factors.
Meta-regression analyses
Meta-regression is often used to explore the sources and sizes of heterogeneity among studies and to further explain the effects of heterogeneity in the meta-analysis. We hypothesized that the included studies could exhibit differences in the demographic variability (sample size) and the combined traditional CV risk factors (smoking habit, diabetes mellitus, obesity, hyperlipidaemia and hypertension). To assess the possible effects of these variables in explaining different results observed across studies, a regression model with changes in CIMT, FMD, PWV and EAT values as dependent variables (y) and the abovementioned co-variates as independent variables (x) was constructed.
Availability of data and materials
All the data we get was from public sources.
Results
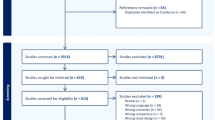
After excluding the repeated citations, we retrieved 423 articles. Among these studies, 217 were excluded because they deviated from the theme according to the title and/or abstract, 15 because they were animal/non-case-control studies or because they lacked the data of interest.
Thus, 27 studies (on 1065 SCH cases and 866 subjects) were included in the final analysis28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54 (Fig. 1). Thirteen studies compared the CIMT between 494 SCH patients and 390 EU subjects. PWV was evaluated in 5 studies (248 cases and 240 controls), 9 studies reported FMD (230 patients and 204 controls), 7 studies reported EAT (297 cases and 239 controls), and 6 studies reported GNT-induced dilation (149 cases and 125 controls).
Study characteristics
All the included studies were case-control designs. The main characteristics of the studies are shown in Table 1.
The number of patients varied from 10 to 67, the mean age ranged from 8.1 to 65.2 years. Smoking habit was reported by 4 studies28,36,41,52, obesity by 4 studies28,32,40,52, hypertensive by only 1 study36 and hyperlipidaemia by 4 studies30,41,42,52.
The NOS for quality assessment of the included studies yielded a median value of 6. There were 23 high-quality articles in our meta-analysis28,29,31,32,33,34,35,36,37,40,42,43,44,45,46,47,48,49,50,51,52,53,54.
Subclinical hypothyroidism versus euthyroidism
Analysis of 13 studies, revealed a significantly higher CIMT in 494 SCH patients than in 390 EU subjects (SMD: 0.369 mm; 95%CI: 0.038, 0.700; P = 0.029, Fig. 2A), with significant heterogeneity among studies (I2 = 82.4%; P = 0.000).
Analysis of 5 studies, evaluating a total of 248 cases and 240 controls, indicated that SCH patients have a significantly higher PWV than EU subjects (SMD: 3.574 m/s; 95%CI: 0.935, 6.213, P = 0.008, Fig. 2B), and the heterogeneity among studies was significant (I2 = 99%, P = 0.000).
Nine studies, evaluating a total of 230 SCH patients and 204 controls, indicated that SCH patients have a significantly lower FMD than EU subjects (SMD: −1.525%, 95%CI: −2.156, −0.894, P = 0.000, Fig. 2C), with significant heterogeneity among studies (I2 = 87.8%; P = 0.000).
Seven studies, evaluating a total of 297 SCH patients and 239 EU subjects, revealed a significantly higher EAT in SCH patients than in EU subjects (SMD: 1.167 mm; 95%CI: 0.869, 1.466; P = 0.000, Fig. 2D), with significant heterogeneity among studies (I2 = 60%; P = 0.02).
Six studies, evaluating a total of 149 SCH patients and 125 controls, showed that SCH patients have a significantly lower GNT-induced dilation than EU subjects (SMD: −0.384%, 95%CI: -0.625, −0.142, P = 0.002, Fig. 2E), without heterogeneity among studies (I2 = 0%; P = 0.78).
Publication bias in included studies may affect the results of meta-analyses. Therefore, we used funnel plots to assess potential publication bias among our included studies.Visual evaluation of the funnel plot for the included studies on SCH patients and EU subjects indicated low publication bias for CIMT (Fig. 3A), confirmed by Begg’s test (P = 0.127, Fig. 4A) and Egger’s test (P = 0.196). Similarly, no publication bias was found for EAT and GNT-induced dilation by inspection of the funnel plots (Fig. 3D,E), confirmed by Begg’s test (P = 0.548, Fig. 4D; P = 1.0, Fig. 4E, respectively) and Egger’s test (P = 0.328, P = 0.291, respectively). In comparison, an asymmetric distribution of studies was detected among those that assessed PWV (Fig. 3B) and FMD (Fig. 3C), and Begg’s test (P = 0.086, Fig. 4B; P = 0.029, Fig. 4C, respectively) and Egger’s test (P = 0.000, P = 0.004) confirmed significant publication bias.
We performed sensitivity analysis by deleting single studies one-by-one and performing an additional meta-analysis for each study removed. For CIMT, PWV, FMD, EAT and GNT-induced dilation, the influence of the omission of each individual study on pooled SMD was assessed. However, no single study had an impact on the overall effect according to the sensitivity analysis, and thus, the meta-analysis was statistically stable.
To eliminate the effects of confounding factors on the outcomes of the study, we conducted a subgroup analysis for high-heterogeneity markers. Subgroup analysis using the high-quality studies (n = 10) indicated that CIMT was significantly increased in patients with SCH (SMD: 0.35 mm; 95%CI: 0.10, 0.59; P = 0.005), and the heterogeneity of the result was markedly reduced (I2 = 59%; P = 0.009). For PWV, subgroup analysis revealed that smoking habit (n = 3) was significantly increased in patients with SCH (SMD: 6.138 m/s; 95%CI: 4.493, 7.783; P = 0.005), with significant heterogeneity among studies (I2 = 87.9%; P = 0.00). For FMD, we assessed the variability of heterogeneity and effects by region classification, and we found that the heterogeneity of China studies (n = 3) (SMD: −2.607%; 95%CI: −3.044, −2.170; P = 0.000) (I2 = 0%; P = 0.925) and other national studies (n = 5) (SMD: −0.745%; 95%CI: −1.146, −0.344; P = 0.000) (I2 = 62.3%; P = 0.031) was significantly decreased; the pooled effects were statistically significant, and the heterogeneity of the results was significantly decreased. For EAT, we assessed the variability of heterogeneity and effects of non-obesity (n = 2), and we found that SCH was increased (SMD: 1.538 mm; 95%CI: 1.202, 1.874; P = 0.000) without heterogeneity among studies (I2 = 0%; P = 0.978).
Regression models for studies comparing SCH patients and EU subjects revealed that small-size studies significantly affected CIMT, indicating that small-sample-size studies are the source of heterogeneity (p = 0.075, tau2 = 0.2467, Adj R-squared = 23.39%, I-squared_res = 78.41%, Fig. 5A). Similarly, a small sample size was also associated with FMD heterogeneity (p = 0.041, tau2 = 0.5246, Adj R-squared = 44.21%, I-squared_res = 82.69%, Fig. 5C).According to the regression models for studies of SCH patients and EU subjects, smoking habit (p < 0.01, tau2 = 0.9931, Adj R-squared = 92.41%, I-squared_res = 82.44%, Fig. 5B) as a confounding factor significantly affected PWV. No other demographic or clinical factors influenced the assessment results.
Meta-regression analyses: impact of demographic and clinical variables on effect size assessed with regression techniques. Subclinical hypothyroidism versus euthyroidism: effect of sample size on carotid intima-media thickness (A), smoking habit on pulse wave velocity (B), and sample size on flow-mediated dilation (C).
Discussion
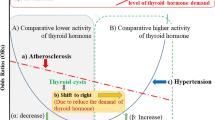
This meta-analysis encompassing 27 studies and including 1065 SCH patients and 866 EU subjects provides compelling evidence that SCH was associated with increased arterial stiffness. We also found a marked increase in EAT and impaired endothelial function (as expressed by a reduced FMD and GNT-induced dilation) in SCH patients compared with EU controls. The sensitivity analysis results strongly confirmed our findings. In addition, the regression models provide evidence that smoking habits and small sample sizes have significant effects on the results of assessments. A former published meta-analysis reported an increase in CIMT in patients with SCH55. Our meta-analysis confirms this result and further reveals significant adverse effects of PWV, EAT, FMD and GNT-induced dilation as other CV risk factors.
There is frequent concern regarding the long-term adverse effects of various factors on increasing the risk of CV disease in SCH patients. SCH is associated with increased levels of total cholesterol and low-density lipoprotein cholesterol. In a population-based study of the Health Aging and Body Composition, the authors prospectively investigated the adverse effects of TSH levels on CV outcomes through a 4-year follow-up. The study found that when the patient’s TSH levels exceeded 7 mIU/L and 10 mIU/L, the risk ratios for developing heart failure were 2.58 and 3.26, respectively56. Another study conducted by Rodondi et al. revealed that more than 55,000 individuals exhibited a positive association of elevated TSH levels with CV events rates and mortality8.
Many CV risk factors are deemed to play causal roles in the atherosclerotic process57. SCH contributes to vascular endothelial dysfunction by promoting lipid metabolism disorder. A study carried out in DaDong district of Shenyang city (China) revealed that the elevated serum TSH levels are positively correlated with serum total cholesterol, triglyceride, and low-density lipoprotein cholesterol and negatively correlated with high-density lipoprotein cholesterol58. Although the majority of SCH patients are hyperlipidaemic, the correlation between subclinical atherosclerosis and SCH appear to be more complicated, and dyslipidaemia may not fully explain the accelerating atherosclerosis in this clinical setting10,59. There is increasing evidence that long-term exposure to high levels of TSH has a deleterious effect on the CV system beyond the mechanism of dyslipidaemia9,33,49,60,61,62. In addition, chronic inflammation may initiate and promote atherosclerosis and its complications through adverse effects on the vascular endothelium, which may be one of the causes of endothelial dysfunction. The clinical importance of SCH in CV disease and mortality remains controversial, and many studies offer conflicting results. This can be explained by the choice of heterogeneous patient groups, arbitrary definitions of TSH reference limits when defining SCH, lack of stratification based on elevated TSH levels, and different study designs.
To fully understand the correlation between SCH and subclinical atherosclerosis, our meta-analysis involves major CV risk markers. In addition, to prevent SCH-related dyslipidaemia from affecting the results of our assessment, we compared hyperlipidaemic and non-dyslipidaemic SCH patients, and the results were consistent. In addition, a meta-regression analysis was conducted to assess whether clinical data and demographic variables affect the outcome. As expected, the regression models revealed that the combination of traditional CV risk factors (e.g., smoking habits, hyperlipidaemia) and small-sample-size studies affect the outcomes. In addition, FMD and GNT-induced dilation were significantly impaired and PWV was significantly increased in SCH patients compared with EU subjects. However, given the small number of studies included in the PWV results, these results should be interpreted with caution. Studies have shown that when CIMT increases by 0.163 mm, the risk of myocardial infarction increases by 43%63,64; furthermore, the risk of CV events increases by 14% when PWV increases by 1 m/s16. These data illustrate the clinical significance of our assessment of these indicators and the need for regular examination of the relevant indicators of subclinical atherosclerosis in patients with SCH.
Some potential limitations of this study need to be considered. First, among the studies included in this meta-analysis, each study had their own inclusion and exclusion criteria. Some of these patients were characterized by CV risk factors (smoking, obesity, diabetes mellitus, hyperlipidaemia and hypertension). Although the results of the regression analysis could be improved by assessing the impact of most clinical and demographic variables on the outcomes of the observation, caution is needed in interpreting the overall outcome. Finally, the evaluation of heterogeneity among studies is very important. Although all sources of possible heterogeneity could not be conclusively determined, the stability of the outcomes was confirmed after adjusting for potential publication bias.
In conclusion, SCH has a significant association with arterial wall thickening and stiffening and endothelial dysfunction and increased risk of CV events. Therefore, Whether SCH patients can benefit from early assessments of measures of CV risk markers that may require large-scale, long-term clinical studies to further confirmation.
References
Canaris, G. J., Manowitz, N. R., Mayor, G. & Ridgway, E. C. The Colorado thyroid disease prevalence study. Arch Intern Med 160, 526–534 (2000).
Evered, D. C., Ormston, B. J., Smith, P. A., Hall, R. & Bird, T. Grades of hypothyroidism. Br Med J 1, 657–662 (1973).
Duntas, L. H. Subclinical hypothyroidism: a misnomer in search of a new name. Thyroid 11, 361–362, https://doi.org/10.1089/10507250152039091 (2001).
Hollowell, J. G. et al. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab 87, 489–499, https://doi.org/10.1210/jcem.87.2.8182 (2002).
Masaki, M. et al. Elevated arterial stiffness and diastolic dysfunction in subclinical hypothyroidism. Circ J 78, 1494–1500 (2014).
Razvi, S. et al. The beneficial effect of L-thyroxine on cardiovascular risk factors, endothelial function, and quality of life in subclinical hypothyroidism: randomized, crossover trial. J Clin Endocrinol Metab 92, 1715–1723, https://doi.org/10.1210/jc.2006-1869 (2007).
Monzani, F. et al. Effect of levothyroxine replacement on lipid profile and intima-media thickness in subclinical hypothyroidism: a double-blind, placebo- controlled study. J Clin Endocrinol Metab 89, 2099–2106, https://doi.org/10.1210/jc.2003-031669 (2004).
Rodondi, N. et al. Subclinical hypothyroidism and the risk of coronary heart disease and mortality. JAMA 304, 1365–1374, https://doi.org/10.1001/jama.2010.1361 (2010).
Singh, S. et al. Impact of subclinical thyroid disorders on coronary heart disease, cardiovascular and all-cause mortality: a meta-analysis. Int J Cardiol 125, 41–48, https://doi.org/10.1016/j.ijcard.2007.02.027 (2008).
Iacobellis, G., Malavazos, A. E. & Corsi, M. M. Epicardial fat: from the biomolecular aspects to the clinical practice. Int J Biochem Cell Biol 43, 1651–1654, https://doi.org/10.1016/j.biocel.2011.09.006 (2011).
Bots, M. L. & Grobbee, D. E. Intima media thickness as a surrogate marker for generalised atherosclerosis. Cardiovasc Drugs Ther 16, 341–351 (2002).
de Groot, E. et al. Measurement of arterial wall thickness as a surrogate marker for atherosclerosis. Circulation 109, III33–38, https://doi.org/10.1161/01.CIR.0000131516.65699.ba (2004).
O’Leary, D. H. et al. Carotid-artery intima and media thickness as a risk factor for myocardial infarction and stroke in older adults. Cardiovascular Health Study Collaborative Research Group. N Engl J Med 340, 14–22, https://doi.org/10.1056/NEJM199901073400103 (1999).
Chambless, L. E. et al. Association of coronary heart disease incidence with carotid arterial wall thickness and major risk factors: the Atherosclerosis Risk in Communities (ARIC) Study, 1987–1993. Am J Epidemiol 146, 483–494 (1997).
Calabia, J. et al. Doppler ultrasound in the measurement of pulse wave velocity: agreement with the Complior method. Cardiovasc Ultrasound 9, 13, https://doi.org/10.1186/1476-7120-9-13 (2011).
Vlachopoulos, C., Aznaouridis, K. & Stefanadis, C. Prediction of cardiovascular events and all-cause mortality with arterial stiffness: a systematic review and meta-analysis. J Am Coll Cardiol 55, 1318–1327, https://doi.org/10.1016/j.jacc.2009.10.061 (2010).
Ras, R. T., Streppel, M. T., Draijer, R. & Zock, P. L. Flow-mediated dilation and cardiovascular risk prediction: a systematic review with meta-analysis. Int J Cardiol 168, 344–351, https://doi.org/10.1016/j.ijcard.2012.09.047 (2013).
Bettencourt, N. et al. Epicardial adipose tissue is an independent predictor of coronary atherosclerotic burden. Int J Cardiol 158, 26–32, https://doi.org/10.1016/j.ijcard.2010.12.085 (2012).
Corretti, M. C. et al. Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: a report of the International Brachial Artery Reactivity Task Force. J Am Coll Cardiol 39, 257–265 (2002).
Mackenzie, I. S., Wilkinson, I. B. & Cockcroft, J. R. Assessment of arterial stiffness in clinical practice. QJM 95, 67–74 (2002).
Yerramasu, A. et al. Increased volume of epicardial fat is an independent risk factor for accelerated progression of sub-clinical coronary atherosclerosis. Atherosclerosis 220, 223–230, https://doi.org/10.1016/j.atherosclerosis.2011.09.041 (2012).
Moher, D., Liberati, A., Tetzlaff, J., Altman, D. G. & Group, P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6, e1000097, https://doi.org/10.1371/journal.pmed.1000097 (2009).
Wells, G. A. et al Ottawa Hospital Research Institute. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in meta-analysis, http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp.
Takeshima, N. et al. Which is more generalizable, powerful and interpretable in meta-analyses, mean difference or standardized mean difference? BMC Med Res Methodol 14, 30, https://doi.org/10.1186/1471-2288-14-30 (2014).
Higgins, J. P., Thompson, S. G., Deeks, J. J. & Altman, D. G. Measuring inconsistency in meta-analyses. BMJ 327, 557–560, https://doi.org/10.1136/bmj.327.7414.557 (2003).
Sterne, J. A., Egger, M. & Smith, G. D. Systematic reviews in health care: Investigating and dealing with publication and other biases in meta-analysis. BMJ 323, 101–105 (2001).
Duval, S. & Tweedie, R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56, 455–463 (2000).
Akbaba, G. et al. Changes in the before and after thyroxine treatment levels of adipose tissue, leptin, and resistin in subclinical hypothyroid patients. Wien Klin Wochenschr 128, 579–585, https://doi.org/10.1007/s00508-015-0865-9 (2016).
Almeida, C. A. et al. Carotid intima-media thickness as a marker of cardiovascular risk in patients with subclinical hypothyroidism. Arq Bras Endocrinol Metabol 51, 472–477 (2007).
Altay, M. et al. Serum Total Sialic Acid Level is Elevated in Hypothyroid Patients as an Atherosclerotic Risk Factor. J Clin Lab Anal 31, https://doi.org/10.1002/jcla.22034 (2017).
Arpaci, D. et al. Epicardial Adipose Tissue Thickness in Patients With Subclinical Hypothyroidism and the Relationship Thereof With Visceral Adipose Tissue Thickness. J Clin Med Res 8, 215–219, https://doi.org/10.14740/jocmr2460w (2016).
Asik, M. et al. Evaluation of epicardial fat tissue thickness in patients with Hashimoto thyroiditis. Clin Endocrinol (Oxf) 79, 571–576, https://doi.org/10.1111/cen.12176 (2013).
Aydogdu, A. et al. Epicardial fat thickness and oxidative stress parameters in patients with subclinical hypothyroidism. Arch Med Sci 13, 383–389, https://doi.org/10.5114/aoms.2017.65479 (2017).
Belen, E. et al. The Association between Subclinical Hypothyroidism and Epicardial Adipose Tissue Thickness. Korean Circ J 45, 210–215, https://doi.org/10.4070/kcj.2015.45.3.210 (2015).
Cerbone, M. et al. Effects of L-thyroxine treatment on early markers of atherosclerotic disease in children with subclinical hypothyroidism. Eur J Endocrinol 175, 11–19, https://doi.org/10.1530/EJE-15-0833 (2016).
Cikim, A. S. et al. Evaluation of endothelial function in subclinical hypothyroidism and subclinical hyperthyroidism. Thyroid 14, 605–609, https://doi.org/10.1089/1050725041692891 (2004).
G., D. X., J., H. P., H., L. S. & L., S. Z. Alpha-lipoic acid improves endothelial dysfunction in patients with subclinical hypothyroidism. Exp Clin Endocrinol Diabetes 118, 625–629, https://doi.org/10.1055/s-0029-1237702 (2010).
Isik-Balci, Y. et al. Impaired Hemorheological Parameters and Increased Carotid Intima-Media Thickness in Children with Subclinical Hypothyroidism. Horm Res Paediatr 85, 250–256, https://doi.org/10.1159/000444363 (2016).
Kalra, P., Yeragani, V. K. & Prasanna Kumar, K. M. Cardiac autonomic function and vascular profile in subclinical hypothyroidism: Increased beat-to-beat QT variability. Indian J Endocrinol Metab 20, 605–611, https://doi.org/10.4103/2230-8210.190527 (2016).
Kebapcilar, L. et al. Effect of levothyroxine replacement therapy on paraoxonase-1 and carotid intima-media thickness in subclinical hypothyroidism. Med Sci Monit 16, CR41–47 (2010).
Kilic, I. D. et al. Noninvasive indicators of atherosclerosis in subclinical hypothyroidism. Indian J Endocrinol Metab 17, 271–275, https://doi.org/10.4103/2230-8210.109708 (2013).
Kim, S. K., Kim, S. H., Park, K. S., Park, S. W. & Cho, Y. W. Regression of the increased common carotid artery-intima media thickness in subclinical hypothyroidism after thyroid hormone replacement. Endocr J 56, 753–758 (2009).
Korkmaz, L. et al. Epicardial adipose tissue increased in patients with newly diagnosed subclinical hypothyroidism. Med Princ Pract 22, 42–46, https://doi.org/10.1159/000340065 (2013).
Nagasaki, T. et al. Increased pulse wave velocity in subclinical hypothyroidism. J Clin Endocrinol Metab 91, 154–158, https://doi.org/10.1210/jc.2005-1342 (2006).
Nagasaki, T. et al. Central pulse wave velocity is responsible for increased brachial-ankle pulse wave velocity in subclinical hypothyroidism. Clin Endocrinol (Oxf) 66, 304–308, https://doi.org/10.1111/j.1365-2265.2006.02730.x (2007).
Nagasaki, T. et al. Changes in brachial-ankle pulse wave velocity in subclinical hypothyroidism during normalization of thyroid function. Biomed Pharmacother 61, 482–487, https://doi.org/10.1016/j.biopha.2007.04.004 (2007).
Niknam, N., Khalili, N., Khosravi, E. & Nourbakhsh, M. Endothelial dysfunction in patients with subclinical hypothyroidism and the effects of treatment with levothyroxine. Adv Biomed Res 5, 38, https://doi.org/10.4103/2277-9175.178783 (2016).
Sayin, I. et al. Thickening of the epicardial adipose tissue can be alleviated by thyroid hormone replacement therapy in patients with subclinical hypothyroidism. Kardiol Pol 74, 1492–1498, https://doi.org/10.5603/KP.a2016.0053 (2016).
Turemen, E. E., Cetinarslan, B., Sahin, T., Canturk, Z. & Tarkun, I. Endothelial dysfunction and low grade chronic inflammation in subclinical hypothyroidism due to autoimmune thyroiditis. Endocr J 58, 349–354 (2011).
Unal, E. et al. Association of Subclinical Hypothyroidism with Dyslipidemia and Increased Carotid Intima-Media Thickness in Children. J Clin Res Pediatr Endocrinol 9, 144–149, https://doi.org/10.4274/jcrpe.3719 (2017).
Unubol, M., Eryilmaz, U., Guney, E., Akgullu, C. & Kurt Omurlu, I. Epicardial adipose tissue in patients with subclinical hypothyroidism. Minerva Endocrinol 39, 135–140 (2014).
Valentina, V. N., Marijan, B., Chedo, D. & Branka, K. Subclinical hypothyroidism and risk to carotid atherosclerosis. Arq Bras Endocrinol Metabol 55, 475–480 (2011).
Xiang, G. D. et al. Regular aerobic exercise training improves endothelium-dependent arterial dilation in patients with subclinical hypothyroidism. Eur J Endocrinol 161, 755–761, https://doi.org/10.1530/EJE-09-0395 (2009).
Xiang, G. D., Xiang, L. W., He, H. L. & Zhao, L. S. Postprandial lipaemia suppresses endothelium-dependent arterial dilation in patients with hypothyroidism. Endocrine 42, 391–398, https://doi.org/10.1007/s12020-012-9624-0 (2012).
Gao, N., Zhang, W., Zhang, Y. Z., Yang, Q. & Chen, S. H. Carotid intima-media thickness in patients with subclinical hypothyroidism: a meta-analysis. Atherosclerosis 227, 18–25, https://doi.org/10.1016/j.atherosclerosis.2012.10.070 (2013).
Rugge, B. et al. In Screening and Treatment of Subclinical Hypothyroidism or Hyperthyroidism AHRQ Comparative Effectiveness Reviews (2011 Oct. Report No.: 11(12)-EHC033-EF.).
Martin-Timon, I., Sevillano-Collantes, C., Segura-Galindo, A. & Del Canizo-Gomez, F. J. Type 2 diabetes and cardiovascular disease: Have all risk factors the same strength? World J Diabetes 5, 444–470, https://doi.org/10.4239/wjd.v5.i4.444 (2014).
Lai, Y. et al. The relationship between serum thyrotropin and components of metabolic syndrome. Endocr J 58, 23–30 (2011).
Hassan, A., Altamirano-Ufion, A. & Zulfiqar, B. & Boddu, P. Sub-Clinical Hypothyroidism and Its Association With Increased Cardiovascular Mortality: Call for Action. Cardiol Res 8, 31–35, https://doi.org/10.14740/cr524w (2017).
Floriani, C., Gencer, B., Collet, T. H. & Rodondi, N. Subclinical thyroid dysfunction and cardiovascular diseases: 2016 update. Eur Heart J, https://doi.org/10.1093/eurheartj/ehx050 (2017).
Torun, A. N. et al. Serum total antioxidant status and lipid peroxidation marker malondialdehyde levels in overt and subclinical hypothyroidism. Clin Endocrinol (Oxf) 70, 469–474, https://doi.org/10.1111/j.1365-2265.2008.03348.x (2009).
Cebeci, E., Alibaz-Oner, F., Usta, M., Yurdakul, S. & Erguney, M. Evaluation of oxidative stress, the activities of paraoxonase and arylesterase in patients with subclinical hypothyroidism. J Investig Med 60, 23–28, https://doi.org/10.2310/JIM.0b013e31823581dd (2012).
Ambrosino, P. et al. Subclinical atherosclerosis in patients with rheumatoid arthritis. A meta-analysis of literature studies. Thromb Haemost 113, 916–930, https://doi.org/10.1160/TH14-11-0921 (2015).
van der Meer, I. M. et al. Predictive value of noninvasive measures of atherosclerosis for incident myocardial infarction: the Rotterdam Study. Circulation 109, 1089–1094, https://doi.org/10.1161/01.CIR.0000120708.59903.1B (2004).
Acknowledgements
This work was sponsored by Research Fund for Excellent Dissertation of China Three Gorges University (2017YPY058) and by the Science and Technology Research and Development Project of Yichang City (A11301-15).
Author information
Authors and Affiliations
Contributions
X.Z. and K.Y. conceived and designed the study. K.Y., T.Z., L.Z., J.Y. and Y.L. extracted the data. K.Y., T.Z. and L.Z. analyzed and interpreted the data. K.Y. and T.Z. wrote the paper. Y.K., Z.T. and Q.H. reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yao, K., Zhao, T., Zeng, L. et al. Non-invasive markers of cardiovascular risk in patients with subclinical hypothyroidism: A systematic review and meta-analysis of 27 case control studies. Sci Rep 8, 4579 (2018). https://doi.org/10.1038/s41598-018-22897-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-22897-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.