Abstract
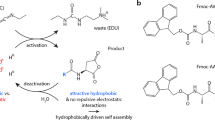
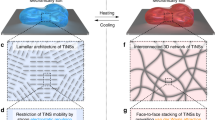
Dissipative self-assembly is common in biological systems, where it serves to maintain a far-from-equilibrium functional state through fuel consumption. Synthetic dissipative systems have been prepared that can mimic some of the properties of biological systems, but they often show poor mechanical performance. Here, we report a shear-induced transient hydrogel that is highly stretchable. The system is constructed by adding Cu(ii) into the aqueous solution of a pseudopolyrotaxane, which is itself formed by threading molecular tubes on polyethylene glycol chains. Vigorous shaking transforms the solution into a gel, which gradually relaxes back to the sol state over time. This cycle can be repeated at least five times. A mechanism is proposed that relies on a shear-induced transition from intrachain to interchain coordination and subsequent thermal relaxation. The far-from-equilibrium hydrogel is highly stretchable, which is probably due to ‘frictional’ sliding of the molecular tubes on the polyethylene glycol chains. On shaking, the hydrogel undergoes fast self-healing.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre under deposition no. CCDC 1563693 (7@2b). Copies of the data can be obtained free of charge from www.ccdc.cam.ac.uk/structures/. All other data supporting the findings of this study are available within the Article and its Supplementary Information and/or from the corresponding author upon reasonable request.
References
Philp, D. & Stoddart, J. F. Self-assembly in natural and unnatural systems. Angew. Chem. Int. Ed. 35, 1154–1196 (1996).
Steed, J. W. & Gale, P. A. (eds) in Supramolecular Chemistry: from Molecules to Nanomaterials Vol. 5, 1965 (Wiley, Hoboken, 2012).
Fialkowski, M. et al. Principles and implementations of dissipative (dynamic) self-assembly. J. Phys. Chem. B 110, 2482–2496 (2006).
Mattia, E. & Otto, S. Supramolecular systems chemistry. Nat. Nanotechnol. 10, 111–119 (2015).
van Rossum, S. A., Tena-Solsona, M., van Esch, J. H., Eelkema, R. & Boekhoven, J. Dissipative out-of-equilibrium assembly of man-made supramolecular materials. Chem. Soc. Rev. 46, 5519–5535 (2017).
Grzybowski, B. A. & Huck, W. T. The nanotechnology of life-inspired systems. Nat. Nanotechnol. 11, 585–592 (2016).
Soejima, T., Morikawa, M. A. & Kimizuka, N. Holey gold nanowires formed by photoconversion of dissipative nanostructures emerged at the aqueous–organic interface. Small 5, 2043–2047 (2009).
Eelkema, R. et al. Molecular machines: nanomotor rotates microscale objects. Nature 440, 163–163 (2006).
Klajn, R., Wesson, P. J., Bishop, K. J. M. & Grzybowski, B. A. Writing self-erasing images using metastable nanoparticle ‘inks’. Angew. Chem. Int. Ed. 48, 7035–7039 (2009).
Ragazzon, G., Baroncini, M., Silvi, S., Venturi, M. & Credi, A. Light-powered autonomous and directional molecular motion of a dissipative self-assembling system. Nat. Nanotechnol. 10, 70–75 (2015).
Kundu, P. K. et al. Light-controlled self-assembly of non-photoresponsive nanoparticles. Nat. Chem. 7, 646–652 (2015).
Grzybowski, B. A., Stone, H. A. & Whitesides, G. M. Dynamic self-assembly of magnetized, millimetre-sized objects rotating at a liquid–air interface. Nature 405, 1033–1036 (2000).
Krabbenborg, S. O., Veerbeek, J. & Huskens, J. Spatially controlled out-of-equilibrium host–guest system under electrochemical control. Chem. Eur. J. 21, 9638–9644 (2015).
Nakanishi, H. et al. Dynamic internal gradients control and direct electric currents within nanostructured materials. Nat. Nanotechnol. 6, 740–746 (2011).
Boekhoven, J., Hendriksen, W. E., Koper, G. J. M., Eelkema, R. & van Esch, J. H. Transient assembly of active materials fueled by a chemical reaction. Science 349, 1075–1079 (2015).
Pappas, C. G., Sasselli, I. R. & Ulijn, R. V. Biocatalytic pathway selection in transient tripeptide nanostructures. Angew. Chem. Int. Ed. 54, 8119–8123 (2015).
Maiti, S., Fortunati, I., Ferrante, C., Scrimin, P. & Prins, L. J. Dissipative self-assembly of vesicular nanoreactors. Nat. Chem. 8, 725–731 (2016).
Wang, G. et al. The fabrication of a supra-amphiphile for dissipative self-assembly. Chem. Sci. 7, 1151–1155 (2016).
Kariyawasam, L. S. & Hartley, C. S. Dissipative assembly of aqueous carboxylic acid anhydrides fueled by carbodiimides. J. Am. Chem. Soc. 139, 11949–11955 (2017).
Dambenieks, A. K., Vu, P. H. Q. & Fyles, T. M. Dissipative assembly of a membrane transport system. Chem. Sci. 5, 3396–3403 (2014).
Dhiman, S., Jain, A. & George, S. J. Transient helicity: fuel-driven temporal control over conformational switching in a supramolecular polymer. Angew. Chem. Int. Ed. 56, 1329–1333 (2017).
Semenov, S. N. et al. Rational design of functional and tunable oscillating enzymatic networks. Nat. Chem. 7, 160–165 (2015).
Yashin, V. V. & Balazs, A. C. Pattern formation and shape changes in self-oscillating polymer gels. Science 314, 798–801 (2006).
Che, H., Buddingh, B. C. & van Hest, J. C. M. Self-regulated and temporal control of a ‘breathing’ microgel mediated by enzymatic reaction. Angew. Chem. Int. Ed. 56, 12581–12585 (2017).
Cheng, C. et al. An artificial molecular pump. Nat. Nanotechnol. 10, 547–553 (2015).
Wood, C. S., Browne, C., Wood, D. M. & Nitschke, J. R. Fuel-controlled reassembly of metal–organic architectures. ACS Cent. Sci. 1, 504–509 (2015).
Erbas-Cakmak, S. et al. Rotary and linear molecular motors driven by pulses of a chemical fuel. Science 358, 340–343 (2017).
Kumar, M. et al. Amino-acid-encoded biocatalytic self-assembly enables the formation of transient conducting nanostructures. Nat. Chem. 10, 696–703 (2018).
Brown, E. et al. Generality of shear thickening in dense suspensions. Nat. Mater. 9, 220–224 (2010).
Chen, H. et al. Blood-clotting-inspired reversible polymer-colloid composite assembly in flow. Nat. Commun. 4, 1333 (2013).
Zebrowski, J., Prasad, V., Zhang, W., Walker, L. M. & Weitz, D. A. Shake-gels: shear-induced gelation of laponite-PEO mixtures. Colloids Surf. A 213, 189–197 (2003).
Witten, T. A. Associating polymers and shear thickening. J. Phys. France 49, 1055–1063 (1988).
Winnik, M. A. & Yekta, A. Associative polymers in aqueous solution. Curr. Opin. Colloid Interface Sci. 2, 424–436 (1997).
Aubry, T. & Moan, M. Rheological behavior of a hydrophobically associating water soluble polymer. J. Rheol. 38, 1681–1692 (1994).
Huang, G.-B., Wang, S.-H., Ke, H., Yang, L.-P. & Jiang, W. Selective recognition of highly hydrophilic molecules in water by endo-functionalized molecular tubes. J. Am. Chem. Soc. 138, 14550–14553 (2016).
Yao, H. et al. Molecular recognition of hydrophilic molecules in water by combining the hydrophobic effects with hydrogen bonding. J. Am. Chem. Soc. 140, 13466–13477 (2018).
Wang, L.-L. et al. Molecular recognition and chirality sensing of epoxides in water using endo-functionalized molecular tubes. J. Am. Chem. Soc. 139, 8436–8439 (2017).
Shriver, D. F. & Atkins, P. W. Inorganic Chemistry 4th edn, 227–236 (Oxford Univ. Press, 2001).
Naota, T. & Koori, H. Molecules that assemble by sound: an application to the instant gelation of stable organic fluids. J. Am. Chem. Soc. 127, 9324–9325 (2005).
Ramirez, A. L. B. et al. Mechanochemical strengthening of a synthetic polymer in response to typically destructive shear forces. Nat. Chem. 5, 757–761 (2013).
Harada, A., Li, J. & Kamachi, M. Preparation and properties of inclusion complexes of polyethylene glycol with α-cyclodextrin. Macromolecules 26, 5698–5703 (1993).
Biedermann, F., Nau, W. M. & Schneider, H. J. The hydrophobic effect revisited—-studies with supramolecular complexes imply high-energy water as a noncovalent driving force. Angew. Chem. Int. Ed. 53, 11158–11171 (2014).
Benson, S. W. Statistical factors in the correlation of rate constants and equilibrium constants. J. Am. Chem. Soc. 80, 5151–5154 (1958).
Fasting, C. et al. Multivalency as a chemical organization and action principle. Angew. Chem. Int. Ed. 51, 10472–10498 (2012).
Beyer, M. K. & Clausen-Schaumann, H. Mechanochemistry: the mechanical activation of covalent bonds. Chem. Rev. 105, 2921–2948 (2005).
Siegler, M. A., Hao, X., Parkin, S. & Brock, C. P. Five more phases of the structural family [M(H2O)2(15-crown-5)](NO3)2. Acta Crystallogr. B 64, 738–749 (2008).
Sun, J.-Y. et al. Highly stretchable and tough hydrogels. Nature 489, 133–136 (2012).
Imran, A. B. et al. Extremely stretchable thermosensitive hydrogels by introducing slide-ring polyrotaxane cross-linkers and ionic groups into the polymer network. Nat. Commun. 5, 5124 (2014).
Okumura, Y. & Ito, K. The polyrotaxane gel: a topological gel by figure‐of‐eight cross‐links. Adv. Mater. 13, 485–487 (2001).
Zhang, Y. S. & Khademhosseini, A. Advances in engineering hydrogels. Science 356, eaaf3627 (2017).
Acknowledgements
This research was financially supported by the National Natural Science Foundation of China (nos. 21772083 and 21822104), SZSTI (nos. JCYJ20170307105848463 and KQJSCX20170728162528382), the Shenzhen Nobel Prize Scientists Laboratory Project (C17213101) and the Open Project of the State Key Laboratory of Supramolecular Structure and Materials (sklssm201807, Jilin University). The authors thank SUSTech-MCPC for instrumental support and C.A. Schalley, Z.-T. Li, W. Lu and S. Craig for valuable suggestions and comments. This Article is dedicated to Y. Liu and J. Rebek.
Author information
Authors and Affiliations
Contributions
W.J. conceived and designed the experiments. H.K. performed all the experiments with the help of L.-P.Y. and H.Y. Z.C. optimized the synthesis of the molecular tubes. M.X. performed the DFT calculations. W.J. and H.K. analysed the data. W.J. wrote the manuscript and all authors commented on it.
Corresponding author
Ethics declarations
Competing interests
W.J., H.K. and L.-P.Y. are listed as the inventors on a Chinese patent application from Southern University of Science and Technology (patent application no. CN201710569674.7). The patent, currently under substantive examination, comprises the following aspects of this study: binding of the PEGs with the endo-functionalized molecular tubes, the preparation of the hydrogels and the shear-thickening measurements.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Titration data, 1H and 13C NMR spectra, mass spectra, crystallographic data, computational details, and details of control experiments.
Crystallographic data
CIF for compound 7@2b; CCDC reference: 1563693
Crystallographic data
structure-factor file for compound 7@2b; CCDC reference: 1563693
Supplementary Video 1
Demonstration of shear-induced transition from a sol to a gel
Supplementary Video 2
Demonstration of the high elasticity of the shear-induced transient hydrogel.
Supplementary Video 3
Demonstration of the breakage of the merged hydrogel without shaking
Rights and permissions
About this article
Cite this article
Ke, H., Yang, LP., Xie, M. et al. Shear-induced assembly of a transient yet highly stretchable hydrogel based on pseudopolyrotaxanes. Nat. Chem. 11, 470–477 (2019). https://doi.org/10.1038/s41557-019-0235-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0235-8
This article is cited by
-
A general strategy for synthesizing biomacromolecular ionogel membranes via solvent-induced self-assembly
Nature Synthesis (2023)
-
Mechanosensitive non-equilibrium supramolecular polymerization in closed chemical systems
Nature Communications (2023)
-
Switchable bifunctional molecular recognition in water using a pH-responsive Endo-functionalized cavity
Nature Communications (2022)
-
A 2D material–based transparent hydrogel with engineerable interference colours
Nature Communications (2022)
-
Specific behavior of transition metal chloride complexes for achieving giant ionic thermoelectric properties
npj Flexible Electronics (2022)