Abstract
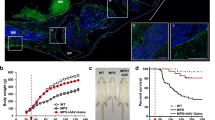
Patients with sialidosis (mucolipidosis type I) type I typically present with myoclonus, seizures, ataxia, cherry-red spots, and blindness because of mutations in the neuraminidase 1 (NEU1) gene. Currently, there is no treatment for sialidosis. In this study, we developed an adeno-associated virus (AAV)-mediated gene therapy for a Neu1 knockout (Neu1−/−) mouse model of sialidosis. The vector, AAV9-P3-NP, included the human NEU1 promoter, NEU1 cDNA, IRES, and CTSA cDNA. Untreated Neu1−/− mice showed astrogliosis and microglial LAMP1 accumulation in the nervous system, including brain, spinal cord, and dorsal root ganglion, together with impaired motor function. Coexpression of NEU1 and protective protein/cathepsin A (PPCA) in neonatal Neu1−/− mice by intracerebroventricular injection, and less effective by facial vein injection, decreased astrogliosis and LAMP1 accumulation in the nervous system and improved rotarod performance of the treated mice. Facial vein injection also improved the grip strength and survival of Neu1−/− mice. Therefore, cerebrospinal fluid delivery of AAV9-P3-NP, which corrects the neurological deficits of mice with sialidosis, could be a suitable treatment for patients with sialidosis type I.

After intracerebroventricular or facial vein injection of AAV vectors, NEU1 and PPCA are expressed together. PPCA-protected NEU1 is then sent to lysosomes, where β–Gal binds to this complex to form a multienzyme complex in order to execute its function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Data supporting the findings of this study are available from the corresponding authors upon reasonable request.
References
Khan A, Sergi C. Sialidosis: a review of morphology and molecular biology of a rare pediatric disorder. Diagnostics. 2018;8:29.
Bonten E, van der Spoel A, Fornerod M, Grosveld G, d’Azzo A. Characterization of human lysosomal neuraminidase defines the molecular basis of the metabolic storage disorder sialidosis. Genes Dev. 1996;10:3156–69.
Caciotti A, Melani F, Tonin R, Cellai L, Catarzi S, Procopio E, et al. Type I sialidosis, a normosomatic lysosomal disease, in the differential diagnosis of late-onset ataxia and myoclonus: an overview. Mol Genet Metab. 2020;129:47–58.
Arora V, Setia N, Dalal A, Vanaja MC, Gupta D, Razdan T, et al. Sialidosis type II: expansion of phenotypic spectrum and identification of a common mutation in seven patients. Mol Genet Metab Rep. 2020;22:100561.
Bonten EJ, Annunziata I, d’Azzo A. Lysosomal multienzyme complex: pros and cons of working together. Cell Mol Life Sci. 2014;71:2017–32.
Bonten EJ, Campos Y, Zaitsev V, Nourse A, Waddell B, Lewis W, et al. Heterodimerization of the sialidase NEU1 with the chaperone protective protein/cathepsin A prevents its premature oligomerization. J Biol Chem. 2009;284:28430–41.
Mosca R, van de Vlekkert D, Campos Y, Fremuth LE, Cadaoas J, Koppaka V, et al. Conventional and unconventional therapeutic strategies for sialidosis type I. J Clin Med. 2020;9:695.
Wang D, Bonten EJ, Yogalingam G, Mann L, d’Azzo A. Short-term, high dose enzyme replacement therapy in sialidosis mice. Mol Genet Metab. 2005;85:181–9.
Bonten EJ, Wang D, Toy JN, Mann L, Mignardot A, Yogalingam G, et al. Targeting macrophages with baculovirus-produced lysosomal enzymes: implications for enzyme replacement therapy of the glycoprotein storage disorder galactosialidosis. FASEB J. 2004;18:971–3.
Cadaoas J, Hu H, Boyle G, Gomero E, Mosca R, Jayashankar K, et al. Galactosialidosis: preclinical enzyme replacement therapy in a mouse model of the disease, a proof of concept. Mol Ther Methods Clin Dev. 2021;20:191–203.
Samulski RJ, Muzyczka N. AAV-mediated gene therapy for research and therapeutic purposes. Annu Rev Virol. 2014;1:427–51.
Hu H, Mosca R, Gomero E, van de Vlekkert D, Campos Y, Fremuth LE, et al. AAV-mediated gene therapy for galactosialidosis: a long-term safety and efficacy study. Mol Ther Methods Clin Dev. 2021;23:644–58.
Bonten EJ, Yogalingam G, Hu H, Gomero E, van de Vlekkert D, d’Azzo A. Chaperone-mediated gene therapy with recombinant AAV-PPCA in a new mouse model of type I sialidosis. Biochim Biophys Acta. 2013;1832:1784–92.
Gupta AO, Patterson MC, Wood T, Eisengart JB, Orchard PJ, Lund TC. Hematopoietic cell transplantation for sialidosis type I. Mol Genet Metab Rep. 2022;30:100832.
d’Azzo A, Machado E, Annunziata I. Pathogenesis, emerging therapeutic targets and treatment in sialidosis. Expert Opin Orphan Drugs. 2015;3:491–504.
Lai SC, Chen RS, Wu Chou YH, Chang HC, Kao LY, Huang YZ, et al. A longitudinal study of Taiwanese sialidosis type 1: an insight into the concept of cherry-red spot myoclonus syndrome. Eur J Neurol. 2009;16:912–9.
Huang YZ, Lai SC, Lu CS, Weng YH, Chuang WL, Chen RS. Abnormal cortical excitability with preserved brainstem and spinal reflexes in sialidosis type I. Clin Neurophysiol. 2008;119:1042–50.
Fan SP, Lee NC, Lin CH. Clinical and electrophysiological characteristics of a type 1 sialidosis patient with a novel deletion mutation in NEU1 gene. J Formos Med Assoc. 2020;119:406–12.
Lin CC, Shen YR, Chang CC, Guo XY, Young YY, Lai TY, et al. Terminal uridyltransferase 7 regulates TLR4-triggered inflammation by controlling Regnase-1 mRNA uridylation and degradation. Nat Commun. 2021;12:3878.
Lee NC, Shieh YD, Chien YH, Tzen KY, Yu IS, Chen PW, et al. Regulation of the dopaminergic system in a murine model of aromatic L-amino acid decarboxylase deficiency. Neurobiol Dis. 2013;52:177–90.
Sleigh JN, West SJ, Schiavo G. A video protocol for rapid dissection of mouse dorsal root ganglia from defined spinal levels. BMC Res Notes. 2020;13:302.
de Geest N, Bonten E, Mann L, de Sousa-Hitzler J, Hahn C, d’Azzo A. Systemic and neurologic abnormalities distinguish the lysosomal disorders sialidosis and galactosialidosis in mice. Hum Mol Genet. 2002;11:1455–64.
Liang F, Seyrantepe V, Landry K, Ahmad R, Ahmad A, Stamatos NM, et al. Monocyte differentiation up-regulates the expression of the lysosomal sialidase, Neu1, and triggers its targeting to the plasma membrane via major histocompatibility complex class II-positive compartments. J Biol Chem. 2006;281:27526–38.
Agarwal S. High-dose AAV gene therapy deaths. Nat Biotechnol. 2020;38:910.
Nguyen GN, Everett JK, Kafle S, Roche AM, Raymond HE, Leiby J, et al. A long-term study of AAV gene therapy in dogs with hemophilia A identifies clonal expansions of transduced liver cells. Nat Biotechnol. 2021;39:47–55.
Weiss C, Ziegler A, Becker LL, Johannsen J, Brennenstuhl H, Schreiber G, et al. Gene replacement therapy with onasemnogene abeparvovec in children with spinal muscular atrophy aged 24 months or younger and bodyweight up to 15 kg: an observational cohort study. Lancet Child Adolesc Health. 2022;6:17–27.
Hinderer C, Bell P, Katz N, Vite CH, Louboutin JP, Bote E, et al. Evaluation of intrathecal routes of administration for adeno-associated viral vectors in large animals. Hum Gene Ther. 2018;29:15–24.
Taghian T, Marosfoi MG, Puri AS, Cataltepe OI, King RM, Diffie EB, et al. A safe and reliable technique for CNS delivery of AAV vectors in the cisterna magna. Mol Ther. 2020;28:411–21.
Uchihara T, Ohashi K, Kitagawa M, Kurata M, Nakamura A, Hirokawa K, et al. Sialidosis type I carrying V217M/G243R mutations in lysosomal sialidase: an autopsy study demonstrating terminal sialic acid in lysosomal lamellar inclusions and cerebellar dysplasia. Acta Neuropathol. 2010;119:135–45.
Hordeaux J, Buza EL, Dyer C, Goode T, Mitchell TW, Richman L, et al. Adeno-associated virus-induced dorsal root ganglion pathology. Hum Gene Ther. 2020;31:808–18.
Lundstrom K. Viral vectors in gene therapy. Diseases. 2018;6:42.
Dunbar CE, High KA, Joung JK, Kohn DB, Ozawa K, Sadelain M. Gene therapy comes of age. Science. 2018;359:eaan4672.
Das S, Huang S, Lo AW. Acceleration of rare disease therapeutic development: a case study of AGIL-AADC. Drug Discov Today. 2019;24:678–84.
Tai CH, Lee NC, Chien YH, Byrne BJ, Muramatsu SI, Tseng SH, et al. Long-term efficacy and safety of eladocagene exuparvovec in patients with AADC deficiency. Mol Ther. 2022;30:509–18.
Howlader MA, Demina EP, Samarani S, Guo T, Caillon A, Ahmad A, et al. The Janus-like role of neuraminidase isoenzymes in inflammation. FASEB J. 2022;36:e22285.
Rama Rao KV, Kielian T. Astrocytes and lysosomal storage diseases. Neuroscience. 2016;323:195–206.
van de Vlekkert D, Demmers J, Nguyen XX, Campos Y, Machado E, Annunziata I, et al. Excessive exosome release is the pathogenic pathway linking a lysosomal deficiency to generalized fibrosis. Sci Adv. 2019;5:eaav3270.
Sumida M, Hane M, Yabe U, Shimoda Y, Pearce OM, Kiso M, et al. Rapid trimming of cell surface polysialic acid (PolySia) by exovesicular sialidase triggers release of preexisting surface neurotrophin. J Biol Chem. 2015;290:13202–14.
Hinek A, Bodnaruk TD, Bunda S, Wang Y, Liu K. Neuraminidase-1, a subunit of the cell surface elastin receptor, desialylates and functionally inactivates adjacent receptors interacting with the mitogenic growth factors PDGF-BB and IGF-2. Am J Pathol. 2008;173:1042–56.
Orkand RK, Nicholls JG, Kuffler SW. Effect of nerve impulses on the membrane potential of glial cells in the central nervous system of amphibia. J Neurophysiol. 1966;29:788–806.
Feng Y, Liu J, Zhang WS. Etomidate-induced myoclonus correlates with the dysfunction of astrocytes and glutamate transporters in the neocortex of Sprague-Dawley rats. Eur Rev Med Pharmacol Sci. 2022;26:6221–35.
Thompson JA, Miralles RM, Wengert ER, Wagley PK, Yu W, Wenker IC, et al. Astrocyte reactivity in a mouse model of SCN8A epileptic encephalopathy. Epilepsia Open. 2022;7:280–92.
Du M, Li J, Ying W, Yu Y. A dynamics model of neuron-astrocyte network accounting for febrile seizures. Cogn Neurodyn. 2022;16:411–23.
Pshezhetsky AV, Ashmarina M. Keeping it trim: roles of neuraminidases in CNS function. Glycoconj J. 2018;35:375–86.
Zincarelli C, Soltys S, Rengo G, Rabinowitz JE. Analysis of AAV serotypes 1-9 mediated gene expression and tropism in mice after systemic injection. Mol Ther. 2008;16:1073–80.
Wang L, Wang H, Bell P, McMenamin D, Wilson JM. Hepatic gene transfer in neonatal mice by adeno-associated virus serotype 8 vector. Hum Gene Ther. 2012;23:533–9.
Acknowledgements
This study is funded by the Sialidosis Research Fund of National Taiwan University Hospital (MS501) and by the Center for Frontier Medicine, National Taiwan University Hospital (111G-003). We thank the technical services provided by the “Transgenic Mouse Model Core Facility of the National Core Facility for Biopharmaceuticals, National Science and Technology Council, Taiwan” and the “Gene Knockout Mouse Core Laboratory of National Taiwan University Center of Genomic and Precision Medicine”. The authors also thank Dr. Yi-Ting Tsai, who interpreted the pathological features of the mice, and other scientists from the Laboratory Animal Center and the First Core Laboratory of National Taiwan University College of Medicine for their help in animal care and image data processing.
Author information
Authors and Affiliations
Contributions
All authors contributed to the concept and design of the manuscript; participated in drafting the manuscript, including the contribution of critical intellectual input; and approved the final version for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The experimental procedures were approved by the National Taiwan University College of Medicine and the College of Public Health Institutional Animal Care and Use.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hwu, WL., Chang, K., Liu, YH. et al. Gene therapy corrects the neurological deficits of mice with sialidosis. Gene Ther (2024). https://doi.org/10.1038/s41434-024-00443-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41434-024-00443-3