Abstract
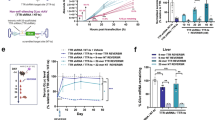
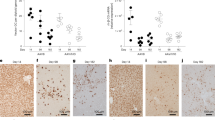
Gene silencing for plasmid-based vectors and the underlying mechanism are critical factors for development of effective gene therapy. The objective of this study is to explore the role of epigenetic regulation for transgene expression. Two reporter genes, mouse interleukin 10 and human secreted alkaline phosphatase under the control of human cytomegalovirus immediate early promoter for expression, were delivered to mouse liver by hrodydynamics-based procedure and reporter gene expression was monitored. Reporter gene expression reached its peak level one day after gene delivery and declined progressively thereafter, reaching the minimal level in about 3 weeks. Intra-peritoneal injection of vorinostat, valproic acid or sodium butyrate, the known histone deacetylase inhibitors, resulted in a dose-dependent reactivation of reporter gene expression. Repeated administration of histone deacetylase inhibitors blocked gene silencing and maintained reporter gene expression. Mechanistic studies reveal that reactivation of reporter genes is corelated with hyperacetylation of histones H3 and H4, and elevated binding of TATA-box binding protein to the promoter region. These results suggest that epigenetic regulation plays a critical role in controlling transgene expression in vivo and demonstrate that enzymes involved in epigenetic regulation such as histone deacetylase could serve as a target to achieve controlled transgene expression for gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Brunner S, Sauer T, Carotta S, Cotten M, Saltik M, Wagner E. Cell cycle dependence of gene transfer by lipoplex, polyplex and recombinant adenovirus. Gene Ther. 2000;7:401–7.
Sarukhan A, Camugli S, Gjata B, von Boehmer H, Danos O, Jooss K. Successful interference with cellular immune responses to immunogenic proteins encoded by recombinant viral vectors. J Virol. 2001;75:269–77.
Lechardeur D, Sohn KJ, Haardt M, Joshi PB, Monck M, Graham RW, et al. Metabolic instability of plasmid DNA in the cytosol: a potential barrier to gene transfer. Gene Ther. 1999;6:482–97.
Zhou HS, Liu DP, Liang CC. Challenges and strategies: the immune responses in gene therapy. Med Res Rev. 2004;24:748–61.
Kass SU, Goddard JP, Adams RL. Inactive chromatin spreads from a focus of methylation. Mol Cell Biol. 1993;13:7372–9.
Yew NS, Zhao H, Przybylska M, Wu IH, Tousignant JD, Scheule RK, et al. CpG-depleted plasmid DNA vectors with enhanced safety and long-term gene expression in vivo. Mol Ther. 2002;5:731–8.
Chen ZY, Yant SR, He CY, Meuse L, Shen S, Kay MA. Linear DNAs concatemerize in vivo and result in sustained transgene expression in mouse liver. Mol Ther. 2001;3:403–10.
Baer A, Schubeler D, Bode J. Transcriptional properties of genomic transgene integration sites marked by electroporation or retroviral infection. Biochemistry. 2000;39:7041–9.
Chen ZY, He CY, Ehrhardt A, Kay MA. Minicircle DNA vectors devoid of bacterial DNA result in persistent and high-level transgene expression in vivo. Mol Ther. 2003;8:495–500.
Argyros O, Wong SP, Niceta M, Waddington SN, Howe SJ, Coutelle C, et al. Persistent episomal transgene expression in liver following delivery of a scaffold/matrix attachment region containing non-viral vector. Gene Ther. 2008;15:1593–605.
Jacobs F, Snoeys J, Feng Y, Van Craeyveld E, Lievens J, Armentano D, et al. Direct comparison of hepatocyte-specific expression cassettes following adenoviral and nonviral hydrodynamic gene transfer. Gene Ther. 2008;15:594–603.
Berger SL. The complex language of chromatin regulation during transcription. Nature. 2007;447:407–12.
Johnstone RW. Histone-deacetylase inhibitors: novel drugs for the treatment of cancer. Nat Rev Drug Discov. 2002;1:287–99.
Kuo MH, Allis CD. Roles of histone acetyltransferases and deacetylases in gene regulation. BioEssays : News Rev Mol, Cell Dev Biol. 1998;20:615–26.
Yang SH, Vickers E, Brehm A, Kouzarides T, Sharrocks AD. Temporal recruitment of the mSin3A-histone deacetylase corepressor complex to the ETS domain transcription factor Elk-1. Mol Cell Biol. 2001;21:2802–14.
Khochbin S, Verdel A, Lemercier C, Seigneurin-Berny D. Functional significance of histone deacetylase diversity. Curr Opin Genet Dev. 2001;11:162–6.
Fischle W, Kiermer V, Dequiedt F, Verdin E. The emerging role of class II histone deacetylases. Biochem Cell Biol = Biochim Et Biol Cell. 2001;79:337–48.
Reeves R, Gorman CM, Howard B. Minichromosome assembly of non-integrated plasmid DNA transfected into mammalian cells. Nucleic Acids Res. 1985;13:3599–615.
Cremisi C, Pignatti PF, Croissant O, Yaniv M. Chromatin-like structures in polyoma virus and simian virus 10 lytic cycle. J Virol. 1975;17:204–11.
Polisky B, McCarthy B. Location of histones on simian virus 40 DNA. Proc Natl Acad Sci USA. 1975;72:2895–9.
Davie JR. Inhibition of histone deacetylase activity by butyrate. J Nutr. 2003;133:2485S–93S.
Marks PA, Xu WS. Histone deacetylase inhibitors: Potential in cancer therapy. J Cell Biochem. 2009;107:600–8.
Finnin MS, Donigian JR, Cohen A, Richon VM, Rifkind RA, Marks PA, et al. Structures of a histone deacetylase homologue bound to the TSA and SAHA inhibitors. Nature. 1999;401:188–93.
MacDonald VE, Howe LJ. Histone acetylation: where to go and how to get there. Epigenetics. 2009;4:139–43.
Shahbazian MD, Grunstein M. Functions of site-specific histone acetylation and deacetylation. Annu Rev Biochem. 2007;76:75–100.
Hochheimer A, Tjian R. Diversified transcription initiation complexes expand promoter selectivity and tissue-specific gene expression. Genes Dev. 2003;17:1309–20.
Lee TI, Young RA. Transcription of eukaryotic protein-coding genes. Annu Rev Genet. 2000;34:77–137.
Davidson I. The genetics of TBP and TBP-related factors. Trends Biochem Sci. 2003;28:391–8.
Wong SP, Argyros O, Harbottle RP. Sustained expression from DNA vectors. Adv Genet. 2015;89:113–52.
Friehs K. Plasmid copy number and plasmid stability. Adv Biochem Eng Biotechnol. 2004;86:47–82.
Al-Dosari M, Zhang G, Knapp JE, Liu D. Evaluation of viral and mammalian promoters for driving transgene expression in mouse liver. Biochem Biophys Res Commun. 2006;339:673–8.
Schüttrumpf J, Milanov P, Abriss D, Roth S, Tonn T, Seifried E. Transgene loss and changes in the promoter methylation status as determinants for expression duration in nonviral gene transfer for factor IX. Hum Gene Ther. 2011;22:101–6.
Argyros O, Wong SP, Fedonidis C, Tolmachov O, Waddington SN, Howe SJ, et al. Development of S/MAR minicircles for enhanced and persistent transgene expression in the mouse liver. J Mol Med. 2011;89:515–29.
Wang YH, Griffith JD. The [(G/C)3NN]n motif: a common DNA repeat that excludes nucleosomes. Proc Natl Acad Sci USA. 1996;93:8863–7.
Johansen J, Tornøe J, Møller A, Johansen TE. Increasedin vitroandin vivotransgene expression levels mediated throughcis-acting elements. J Gene Med. 2003;5:1080–9.
Miao CH, Ye X, Thompson AR. High-level factor VIII gene expression in vivo achieved by nonviral liver-specific gene therapy vectors. Hum Gene Ther. 2003;14:1297–305.
Antoniou M, Harland L, Mustoe T, Williams S, Holdstock J, Yague E, et al. Transgenes encompassing dual-promoter CpG islands from the human TBP and HNRPA2B1 loci are resistant to heterochromatin-mediated silencing. Genomics. 2003;82:269–79.
Hagedorn C, Antoniou MN, Lipps HJ. Genomic cis-acting sequences improve expression and establishment of a nonviral vector. Mol Ther Nucleic Acids. 2013;2:e118. https://doi.org/10.1038/mtna.2013.47.
Gracey Maniar LE, Maniar JM, Chen ZY, Lu J, Fire AZ, Kay MA. Minicircle DNA vectors achieve sustained expression reflected by active chromatin and transcriptional level. Mol Ther. 2013;21:131–8.
Wang J, Zhuang J, Iyer S, Lin X, Whitfield TW, Greven MC, et al. Sequence features and chromatin structure around the genomic regions bound by 119 human transcription factors. Genome Res. 2012;22:1798–812.
Rincon-Arano H, Halow J, Delrow, Jeffrey J, Parkhurst, Susan M, et al. UpSET recruits HDAC complexes and restricts chromatin accessibility and acetylation at promoter regions. Cell. 2012;151:1214–28.
Badding MA, Dean DA. Highly acetylated tubulin permits enhanced interactions with and trafficking of plasmids along microtubules. Gene Ther. 2013;20:616–24.
Elmer JJ, Christensen MD, Barua S, Lehrman J, Haynes KA, Rege K. The histone deacetylase inhibitor Entinostat enhances polymer-mediated transgene expression in cancer cell lines. Biotechnol Bioeng. 2016;113:1345–56.
Gorman CM, Howard BH, Reeves R. Expression of recombinant plasmids in mammalian cells is enhanced by sodium butyrate. Nucleic Acids Res. 1983;11:7631–48.
Chen L, Fischle W, Verdin E, Greene WC. Duration of nuclear NF-kappaB action regulated by reversible acetylation. Science. 2001;293:1653–7.
Horion J, Gloire G, El Mjiyad N, Quivy V, Vermeulen L, Vanden Berghe W, et al. Histone deacetylase inhibitor trichostatin A sustains sodium pervanadate-induced NF-kappaB activation by delaying IkappaBalpha mRNA resynthesis: comparison with tumor necrosis factor alpha. J Biol Chem. 2007;282:15383–93.
Glozak MA, Sengupta N, Zhang X, Seto E. Acetylation and deacetylation of non-histone proteins. Gene. 2005;363:15–23.
Ishii K, Arib G, Lin C, Van Houwe G, Laemmli UK. Chromatin boundaries in budding yeast: the nuclear pore connection. Cell. 2002;109:551–62.
Sun FL, Elgin SC. Putting boundaries on silence. Cell. 1999;99:459–62.
Ghirlando R, Giles K, Gowher H, Xiao T, Xu Z, Yao H, et al. Chromatin domains, insulators, and the regulation of gene expression. Biochim Biophys Acta. 2012;1819:644–51.
Nishihara M, Kanda GN, Suzuki T, Yamakado S, Harashima H, Kamiya H. Enhanced transgene expression by plasmid-specific recruitment of histone acetyltransferase. J Biosci Bioeng. 2017;123:277–80.
Sugiyama KI, Furusawa H, Gruz P, Honma M (2017). Functional role of DNA methylation at the FLO1 promoter in budding yeast. FEMS Microbiol Lett. 2017;fnx221. https://doi.org/10.1093/femsle/fnx221.
Liu F, Song Y, Liu D. Hydrodynamics-based transfection in animals by systemic administration of plasmid DNA. Gene Ther. 1999;6:1258–66.
Acknowledgements
This study is supported by grants from NIH (RO1EB007357 and RO1HL098295). We are grateful to Dr. Leping Li at the National Institute of Environmental Health Sciences for providing the sequence information regarding the transcription factor binding sites in CMV promotor.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary information
Rights and permissions
About this article
Cite this article
Zhang, C., Zhang, G. & Liu, D. Histone deacetylase inhibitors reactivate silenced transgene in vivo. Gene Ther 26, 75–85 (2019). https://doi.org/10.1038/s41434-018-0053-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41434-018-0053-4