Abstract
Data sources The authors of this rapid review did not disclose which electronic databases were included in their literature search. The inclusion and exclusion criteria for the data sources are not reported in the manuscript.
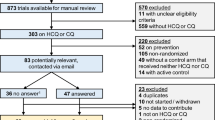
Study selection The authors included six studies on the effectiveness of hydroxychloroquine or chloroquine for the prevention and treatment of COVID-19 in humans. Studies comprised of two randomised controlled trials, two non-randomised trials both of which were non-blinded and open-label and one that was uncontrolled, a prospective cohort study and an interim report. The authors did not report details of any studies that were excluded.
Data extraction and synthesis The data extraction methodology was not reported and it is unclear if the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines were followed. Treatment regimens and the study outcomes were extracted where available and overall findings were presented in a table. There were no comparable outcome measures; therefore, results were deemed unsuitable to combine and no statistical analyses were carried out. A narrative synthesis of each study is presented.
Results The results of the studies in this rapid review are difficult to quantify as each study had different outcome parameters. Due to the heterogeneity of the studies, results were not combined, and no statistical analysis was carried out. Narrative synthesis of each of the included studies identified important and significant limitations, precluding the studies from demonstrating a statistically significant difference in outcomes.
Conclusions This review highlights the urgent need for more high quality evidence on the use of hydroxychloroquine and chloroquine in the prevention and treatment of COVID-19. The results of the studies included should be interpreted with caution due to the weak supporting data and numerous methodological limitations. The authors suggested that the studies be viewed as hypothesis-generating and should not be used in decision making around the recommendations and guidelines in the prevention and treatment of COVID-19. There are currently several ongoing randomised controlled trials looking at the effectiveness and efficacy of these drugs on COVID-19. It is hoped the outcome of these studies can help guide future recommendations and national guidelines.
Similar content being viewed by others
A commentary on
Pastick K A, Okafor E C, Wang F et al.
Review: Hydroxychloroquine and Chloroquine for Treatment of SARS-CoV-2 (COVID-19). Open Forum Infect Dis 2020; 7: DOI: 10.1093/ofid/ofaa130.

GRADE rating
Commentary
In December 2019, a novel coronavirus (SARS-CoV-2) which causes COVID-19 disease emerged from Wuhan in China. It subsequently spread to over 200 countries, with the World Health Organisation (WHO) declaring a pandemic in March 2020.1,2 As of 21 May 2020, there have been 4.8 million confirmed cases of COVID-19, including 321,000 deaths.3 With the numbers rapidly increasing, there is an urgent need to develop effective treatment for symptomatic COVID-19 patients.
There has been a growing interest in the use of hydroxychloroquine and chloroquine in the prevention and treatment of COVID-19. For many years, these drugs have been used for the treatment of malaria, as well as for auto-immune conditions such as systemic lupus erythematosus and rheumatoid arthritis.4 However, findings from in vitro studies have shown both hydroxychloroquine and chloroquine to have an effect on SARS-CoV-2 by impairing replication of the virus.1 Potential side effects of these drugs are well documented and they most commonly cause vomiting, diarrhoea and gastrointestinal upset. However, more severe side effects have been reported and include retinopathy and QTc interval prolongation, which can result in ventricular arrhythmias and sudden cardiac death.1 Clinical trials have been ongoing to determine their effectiveness in treating patients with COVID-19.2,5
The authors of this review did not disclose their search methodology and as such we were not able to repeat or corroborate their database search for appropriate studies. Details of the databases used, search parameters applied or if medical subject headings (MeSH) were utilised are not provided. It was also unclear if studies were included or excluded based on date restrictions, non-English language use or being unpublished/grey literature. Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines do not appear to have been followed. Further, the authors did not detail if registries for randomised controlled trials were searched.
Several barriers and difficulties faced the authors in completing their review. Primarily due to the lack of high-quality evidence available on the use of hydroxychloroquine and chloroquine in the prevention and treatment of COVID-19. The studies that the authors included had significant heterogeneity and numerous limitations. These barriers; combined with poorly defined, non-comparable outcome measures, resulted in difficulty extracting and synthesising relevant data and therefore no useful statistical analyses could be carried out.
The authors employed narrative synthesis to describe the methods and results of each study. The narrative highlighted important and significant limitations with the studies included in this review. All of the studies were underpowered due to their small sample size.2,6,7,8,9,10 Only one of the six studies was appropriately peer-reviewed; however the authors noted that concerns had previously been raised about this study.2 Three of the studies were uncontrolled.8,9,10 Two of the studies were non-randomised, open-label and unblinded.2,9 Significant concerns were also raised about the study designs potentially incorporating inherent bias.2,9 One study did not publish the treatment regimen prescribed to patients and also provided no methodology for the statistical analysis carried out.10 The majority of the studies did not define disease stage and severity of the patients, making it difficult to draw comparisons between studies. Alarmingly, one study included no intention to treat analysis; despite several excluded patients in the treatment arm either requiring ICU admission or dying.2
Currently, there is ongoing research, aimed at determining the effectiveness of hydroxychloroquine and chloroquine in COVID-19 care. Several of the authors in this review have disclosed potential conflicts of interest, as they are principal investigators (PIs) on four ongoing randomised controlled trials investigating the use of hydroxychloroquine in the prevention and treatment of COVID-19 in Catalonia, Canada and the United States. The National Institute for Health Research (NIHR) are also funding a Platform Randomised trial of INterventions against COVID-19 In older peoPLE (PRINCIPLE).11 This UK-based randomised controlled trial will initially look at the use of hydroxychloroquine in modifying the disease course, with the goal of preventing disease progression and reducing hospital admissions. A randomised controlled trial in Brazil investigated the safety and efficacy of chloroquine in patients with established severe COVID-19, however recruitment in the high dose arm was stopped due to potential safety hazards and concerns over an increase in patient mortality.12
Alarmingly, despite the current lack of evidence for the efficacy and effectiveness of hydroxychloroquine and chloroquine in COVID-19 prevention and treatment, several official guidelines have incorporated and suggested their use.13 Perhaps this correlates with the unprecedented pressure caused by the global pandemic, combined with excessive media coverage and public figure endorsements; despite the scarcity of evidence.
This review emphasises the importance of more robust, adequately powered, future randomised controlled trials into the use of hydroxychloroquine and chloroquine for COVID-19; prior to their recommendation in national guidelines. Studies would ideally categorise disease severity and symptomatology prior to participants being randomised into treatment and control groups. Future studies should also focus on meaningful and comparable clinical outcome measures around patient hospital admission, ICU admission and patient mortality. This would allow data from different studies to be more readily compared, synthesised and extrapolated to larger populations. With the current dearth of evidence, patients subjected to hydroxychloroquine and chloroquine may not only be receiving no clinical benefit from the drug but could be coming to harm.14
References
Yao X, Ye F, Zhang M et al. In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin Infect Dis 2020; DOI: 10.1093/cid/ciaa237.
Gautret P, Lagier J-C, Parola P et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents 2020; DOI: 10.1016/j.ijantimicag.2020.105949
World Health Organisation. WHO Coronavirus Disease (COVID-19) Dashboard. https://covid19.who.int (accessed May 2020).
Gbinigie K, Frie K. Should chloroquine and hydroxychloroquine be used to treat COVID-19? A rapid review. BJGP Open 2020; DOI: 10.3399/bjgpopen20x101069.
Chorin E, Dai M, Shulman E et al. The QT Interval in Patients with SARS-CoV-2 Infection Treated with Hydroxychloroquine/Azithromycin. medRxiv. 2020; DOI: 10.1101/2020.04.02.20047050.
Chen J, Liu D, Liu L et al. A pilot study of hydroxychloroquine in treatment of patients with moderate coronavirus disease-19 (COVID-19). J Zhejiang Univ (Med Sci) 2020; 49: 215-219.
Chen Z, Hu J, Zhang Z et al. Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial. medRxiv 2020; DOI: 10.1101/2020.03.22.20040758.
Molina J, Delaugerre C, Goff J et al. No Evidence of Rapid Antiviral Clearance or Clinical Benefit with the Combination of Hydroxychloroquine and Azithromycin in Patients with Severe COVID-19 Infection. Med Mal Infect 2020; DOI: 10.1016/j.medmal.2020.03.006.
Gautret P, Lagier J C, Parola P et al. Clinical and microbiological effect of a combination of hydroxychloroquine and azithromycin in 80 COVID-19 patients with at least a six-day follow up: A pilot observational study. Travel Med Infect Dis 2020; 34: 101663. DOI: 10.1016/j.tmaid.2020.101663.
Gao J, Tian Z, Yang X. Breakthrough: Chloroquine phosphate has shown apparent efficacy in treatment of COVID19 associated pneumonia in clinical studies. Biosci Trends 2020; 14: 72-73.
NHS Health Research Platform. Randomised trial of INterventions against COVID-19 In older peoPLe [PRINCIPLE]. NIHR. Available at https://www.phctrials.ox.ac.uk/principle-trial (accessed May 2020).
Borba M G S, Val F de A, Sampaio V S et al. Chloroquine diphosphate in two different dosages as adjunctive therapy of hospitalized patients with severe respiratory syndrome in the context of coronavirus (SARS-CoV-2) infection: Preliminary safety results of a randomized, double-blinded, phase IIb cl. medRxiv 2020; 4: 20056424. DOI: 10.1101/2020.04.07.20056424.
Lenzer J. Covid-19: US gives emergency approval to hydroxychloroquine despite lack of evidence. BMJ 2020; DOI: 10.1136/bmj.m1335.
Guastalegname M, Vallone A. Could Chloroquine /Hydroxychloroquine Be Harmful in Coronavirus Disease 2019 (COVID-19) Treatment? Clin Infect Dis 2020; DOI: 10.1093/cid/ciaa321.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wright, C., Ross, C. & Mc Goldrick, N. Are hydroxychloroquine and chloroquine effective in the treatment of SARS-COV-2 (COVID-19)?. Evid Based Dent 21, 64–65 (2020). https://doi.org/10.1038/s41432-020-0098-2
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41432-020-0098-2
This article is cited by
-
An observational, retrospective, comprehensive pharmacovigilance analysis of hydroxychloroquine-associated cardiovascular adverse events in patients with and without COVID-19
International Journal of Clinical Pharmacy (2022)