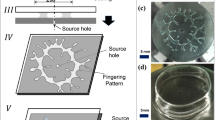
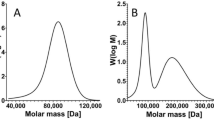
Abstract
“Breath figure formation” during the casting process of polymer solutions under high atmospheric humidity provides honeycomb-patterned polymer films (honeycomb films) with regularly arranged micropores. The development of production technology for large-area honeycomb films is indispensable for their various applications. Manufacturing equipment consisting of three zones (for casting, humidification, and drying of polymer solutions) for successive formation of large-area honeycomb films was newly designed and constructed. By using this equipment, physicochemical experimental parameters, e.g., the surface temperature of polymer solutions, dew point of the humidification zone, humidification time, and interfacial tension between water and the polymer solution, were effectively changed to optimize the density and size of condensed water droplets. Large-area honeycomb films were formed by a roll-to-roll process. Herein, recent developments in biomedical applications of honeycomb films are described.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

















Similar content being viewed by others
References
Campbell M, Sharp DN, Harrison MT, Denning RG, Turberfield AJ. Fabrication of photonic crystals for the visible spectrum by holographic lithography. Nature. 2000;404:53–6.
Hu XB, Li GT, Li MH, Huang J, Li Y, Gao YB, et al. Ultrasensitive specific stimulant assay based on molecularly imprinted photonic hydrogels. Adv Funct Mater. 2008;18:575–83.
Gates B, Yin YD, Xia YN. Fabrication and characterization of porous membranes with highly ordered three-dimensional periodic structures. Chem Mater. 1999;11:2827–36.
Erdogan B, Song LL, Wilson JN, Park JO, Srinivasarao M, Bunz UHF. Permanent bubble arrays from a cross-linked poly (para- phenyleneethynylene): picoliter holes without microfabrication. J Am Chem Soc. 2004;126:3678–9.
Yabu H, Takebayashi M, Tanaka M, Shimomura M. Superhydrophobic and lipophobic properties of self-organized honeycomb and pincushion structures. Langmuir. 2005;21:3235–7.
Fukuhira Y, Ito M, Kaneko H, Sumi Y, Tanaka M, Yamamoto S, et al. Prevention of postoperative adhesions by honeycomb-patterned poly(lactide) film in a rat experimental model. J Biomed Mater PART B Appl Biomater. 2008;86:353–9.
Ishihata H, Tanaka M, Iwata N, Ara M, Shimonishi M, Nagamine M, et al. Proliferation of periodontal ligament cells on biodegradable honeycomb film scaffold with unified micropore organization. J Biomech Sci Eng. 2010;10:252–61.
Schmid H, Michel B. Siloxane polymers for high-resolution, high-accuracy soft lithography. Macromolecules. 2000;33:3042–9.
Aitken J. Breath figures. Proc R Soc Edinb. 1895;20:94–7.
Rayleigh L. Breath figures. Nature. 1911;86:416–7.
Terada T. Physical problems in everyday life. In: Komiya T, editor. Iwanami bunko. Terada Torahiko collected essays Vol. 3 (Iwanamibunko) Tokyo: Iwanami Shoten, Publishers; 1948. p. 15–22.
Widawski G, Rawiso M, François B. Self-organized honeycomb morphology of star-polymer polystyrene films. Nature. 1994;369:387–9.
Maruyama N, Koito T, Nishida J, Sawadaishi T, Cieren X, Ijiro K, et al. Mesoscopic patterns of molecular aggregates on solid substrates. Thin Solid Films. 1998;327-329:854–6.
Stenzel MH. Formation of regular honeycomb-patterned porous film by self-organization. Aust J Chem. 2002;55:239–43.
Bunz UHF. Breath figures as a dynamic templating method for polymers and nanomaterials. Adv Mater. 2006;18:973–89.
Hoa MLK, Lu M, Zhang Y. Preparation of porous materials with ordered hole structure. Adv Colloid Interface Sci. 2006;121:9–23.
Stenzel MH, Barner-Kowollik C, Davis TP. Formation of honeycomb‐structured, porous films via breath figures with different polymer architectures. J Polym Sci, Part A: Polym Chem. 2006;44:2363–75.
Ma HM, Hao JC. Ordered patterns and structures via interfacial self-assembly: superlattices, honeycomb structures and coffee rings. Chem Soc Rev. 2011;40:5457–71.
Escale P, Rubatat L, Billon L, Save M. Recent advances in honeycomb-structured porous polymer films prepared via breath figures. Eur Polym J. 2012;48:1001–25.
Hernandez-Guerrero M, Stenzel MH. Honeycomb structured polymer films via breath figures. Polym Chem. 2012;3:563–77.
Bai H, Du C, Zhang AJ, Li L. Breath figure arrays: unconventional fabrications, functionalizations, and applications. Angew Chem, Int Ed. 2013;52:12240–55.
Muñoz-Bonilla A, Fernández-García M, Rodríguez-Hernández J. Towards hierarchically ordered functional porous polymeric surfaces prepared by the breath figures approach. Prog Polym Sci. 2014;39:510–54.
Wan LS, Zhu LW, Ou Y, Xu ZK. Multiple interfaces in self-assembled breath figures. Chem Commun. 2014;50:4024–39.
Zhang A, Bai H, Li L. Breath figure: a nature-inspired preparation method for ordered porous films. Chem Rev. 2015;115:9801–68.
Yabu H. Fabrication of honeycomb films by the breath figure technique and their applications. Sci Tech Adv Mater. 2018;19:802–22.
Shimomura M, Sawadaishi T. Bottom-up strategy of materials fabrication: a new trend in nanotechnology of soft materials. Curr.Opin. Colloid Interface Sci. 2001;6:11–6.
Iwanaga H, Shiratsuchi K, Yamazaki H. Fujifilm Research & Development. No. 54, 38–42. Fujifilm Value from Innovation;2009.
Elder KR, Katakowski M, Haataja M, Grant M. Modeling elasticity in crystal growth. Phys Rev Lett. 2002;88:245701-1–245701-4.
Elder KR, Grant M. Modeling elastic and plastic deformation in nonequilibrium processing using phasefield crystals. Phys Rev E. 2004;70:051605-1–-051605-18.
Kohashi S, Takaki T, Nishida H. Numerical simulation of self-organized honeycomb-pattern; Ascht 2015 The Asian Symposium on Computational Heat Transfer and Fluid Flow; 2015 June 21–24. Busan, Korea.
Nishida H, Kohashi S, Tanaka M. Construction of seamless immersed boundary phase-field method. Comput Fluids. 2018;164:41–9.
Nishikawa T, Nishida J, Ookura R, Nishimura S, Wada S, Karino T, Shimomura M. Honeycomb-patterned thin films of amphiphilic polymers as cell culture substrates. Mater Sci Eng. 1999;C8-9:495–500.
Nishikawa T, Nonomura M, Arai K, Hayashi J, Sawadaishi T, Nishiura Y, et al. Micropatterns based on deformation of a viscoelastic honeycomb mesh. Langmuir. 2003;19:6193–201.
Yamazaki H, Ito K, Yabu H, Shimomura M. Formation and control of line defects caused by tectonics of water droplet arrays during self-organized honeycomb-patterned polymer film formation. Soft Matter. 2014;10:2741–7.
Ito K, Yamazaki H. Method for production of honeycomb structure film. Japan patent no. 5405374. 2013.
Tanaka M, Shimomura M, Sakaino Y, Ito T, Terashima K.Method for manufacturring blood filtration film, and filtration method. Japan patent no. 2006–194908 2006.
Yamazaki H, Ito K, Shimomura M, Yabu H. Technological prospect and current industrial trend of biomimetics, Ch. 3. Tokyo: CMC Publishing, 2016. p. 41–50.
Tanaka M, Shimomura M, Sakaino Y, Ito T, Ishii N. Covered stent. Japan patent no. 4526589. 2010.
Tanaka M, Shimomura M, Sakaino Y, Ito T, Toyokawa S. Stent for digestive system. Japan patent no. 4512351. 2010.
Fukuhira Y, Ito M, Kaneko H, Sumi Y, Tanaka M, Yamamoto S, et al. Prevention of postoperative adhesions by a novel honeycomb-patterned poly(lactide) film in a rat experimental model. Biomed Mater Res B Appl Biomater. 2008;86B:353–9.
Okuda T, Higashide T, Fukuhira Y, Sumi Y, Shimomura M, Sugiyama K. J Glaucoma. 2009;18:220–6.
Sato K, Tanaka M, Hasebe K, Takebayashi M, Nishikawa K, Kawai T, et al. Preparation of the honeycomb patterned porous film of biodegradable polymer for tissue engineering scaffolds. Int J Nanosci. 2002;1:689–93.
Lin RZ, Chang HY. Recent advances in three-dimensional multicellularspheroid culture for biomedical research. Biotechnol J. 2008;3:1172–84.
Hirschhaeuser F, Menne H, Dittfeld C, West J, Mueller-Klieser W, Kunz-Schughart LA. Multicellular tumor spheroids. an underestimated tool is catching up again. J Biotechnol. 2010;148:3–15.
Kinney MA, Sargent CY, McDevitt TC. The multiparametric effects of hydrodynamic environments on stem cell culture. Tissue Eng Part B. 2011;17:249–62.
Koide N, Sakaguchi K, Koide Y, Asano K, Kawaguchi M, Matsushima H, et al. Formation of multicellularspheroids composed of adult rat hepatocytes in dishes with positively charged surfaces and under other nonadherente nvironments. Exp Cell Res. 1990;186:227–35.
Tong J-Z, Lagausie P-D, Furlan V, Cresteil T, Bernard O, Alvarez F. Long-term culture of adult rat hepatocyte spheroids. Exp Cell Res. 1992;200:326–32.
Abu-Absi SF, Friend JR, Hansen LK, Hu WS. Structural polarity and functional bile canaliculi in rat hepatocyte spheroids. Exp Cell Res. 2002;274:56–67.
Tsuruma A, Tanaka M, Fukushima N, Shimomura M. Morphological changes of neurons in Self-organized honeycomb-patterned films. e-J Surf Sci Nanotechnol. 2005;3:159–64.
Morita Y, Yamamoto S, Yabu H, Ito E, Honmou O, Ijiro K, et al. Response of mesenchymal stem cells from rat adult bone marrow to honeycomb-patterned porous polymer films. J Surf Sci Soc Jpn. 2010;31:392–9.
Mohr JC, de Pablo JJ, Palecek SP. 3-D microwell culture of human embryonic stem cells. Biomaterials. 2006;7:6032–42.
Sakai Y, Nakazawa K. Technique for the control of spheroid diameter using microfabricated chips. Acta Biomater. 2007;3:1033–40.
Sakai Y, Yoshida S, Yoshiura Y, Mori R, Tamura T, Yahiro K, et al. Effect of microwell chipstructure on cell microsphere production of various animal cells. J Biosci Bioeng. 2010;110:223–9.
Nakazawa K, Yoshiura Y, Koga H, Sakai Y. Characterization of mouse embryoid bodies cultured on microwell chips with different well sizes. J Biosci Bioeng. 2013;116:628–33.
Yamazaki H, Yabu H, Shimomura M, Nakazawa K, et al. Micropatterned culture of HepG2 spheroids using microwell chip with honeycomb-patterned polymer film. J Biosci Bioeng. 2014;118:455–60.
Ito K, Kakinuma C, Hikimoto D, Mima S, Nevile CM, Sundback CA. Blood vessel model. U.S. patent no. 20200110075.2020.
Miyoshi H, Naito T, Suehiro T, Hikimoto D, Nishino M, Oba T, et al. Development of blood vessel model by organ-on-chip technology and its application for evaluation of drug induced vascular injury. Poster presented at the 32rd Annual Meeting of the Japanese Society for Alternatives to Animal Experiments. Nov 2019; Tsukuba, Japan.
Acknowledgements
Parts of these studies were supported by 2004–2005 R&D projects of regional regeneration consortiums commissioned by the Ministry of Economy, Trade and Industry and 2007–2011 R&D projects commissioned by the New Energy and Industrial Technology Development Organization (NEDO) for practical application of nanotechnology and advanced materials. We are deeply grateful to these related departments and stakeholders as well as to Professor Kohji Nakazawa of the University of Kitakyushu, Associate Professor Hiroshi Yabu of Tohoku University, Professor Hidetoshi Nishida, Professor Tomohiro Takagi, and Professor Masashi Yamakawa of Kyoto Institute of Technology, Professor Masaru Tanaka of Kyushu University, Professor Yasutaka Matsuo of Hokkaido University, Associate Professor Yuji Hirai of Chitose Institute of Science and Technology and all those involved for their invaluable support in promoting each research area.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yamazaki, H., Kohashi, S., Ito, K. et al. Production technology and applications of honeycomb films. Polym J 54, 107–120 (2022). https://doi.org/10.1038/s41428-021-00549-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41428-021-00549-0