Abstract
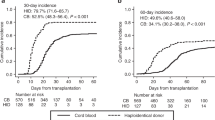
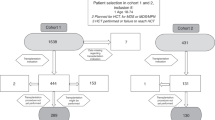
Allogeneic hematopoietic cell transplantation (HCT) is associated with significant morbidity and mortality especially in the first year after HCT. In this study, we examine the long-term outcomes of patients who survived at least one year post HCT without evidence of relapse. We analyzed the records for 389 consecutive patients receiving an allogeneic transplant from 2005 to 2016 from a MRD, MUD, or haploidentical donor, who were alive and disease free at one year post-transplant. Patient characteristics and outcome parameters were extracted from our institutional database where they had been prospectively entered. A total of 389 patients met the selection criteria with donor graft including MRD 37%, MUD 39%, and Haploidenitcal relative 24%. The median follow-up of survivors from time of HCT was 48.2 months. The median overall survival and disease-free survival at 5 years after the first anniversary post HCT was 78 and 74%, respectively. The most common causes of late mortality were disease relapse, chronic GVHD and infections. The major risk factors for late mortality included chronic GVHD requiring immunosuppression, being transplanted between 2005 and 2009 compared to later years and male sex. Patients with high risk disease risk index (DRI) had worse OS compared to low risk DRI. The risk factors for late relapse included male sex and high/very high disease risk index. The projected long-term survival of 1-year survivors following allogeneic HCT is excellent. However, some patients remain at high risk of late relapse and late mortality. Early referral to transplant, adopting post-transplant consolidation strategies for high risk patients, and implementing newer GVHD prevention methods are potential interventions to help minimize the risk of late relapse and death.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Majhail NS, Tao L, Bredeson C, Davies S, Dehn J, Gajewski JL, et al. Prevalence of hematopoietic cell transplant survivors in the United States. Biol Blood Marrow Transplant. 2013;19:1498–501.
Hahn T, McCarthy PL Jr., Hassebroek A, Bredeson C, Gajewski JL, Hale GA, et al. Significant improvement in survival after allogeneic hematopoietic cell transplantation during a period of significantly increased use, older recipient age, and use of unrelated donors. J Clin Oncol. 2013;31:2437–49.
Michelis FV, Gupta V, Zhang MJ, Wang HL, Aljurf M, Bacher U, et al. Cytogenetic risk determines outcomes after allogeneic transplantation in older patients with acute myeloid leukemia in their second complete remission: a Center for International Blood and Marrow Transplant Research cohort analysis. Cancer. 2017; 123:2035–42.
Shimoni A, Hardan I, Shem-Tov N, Yeshurun M, Yerushalmi R, Avigdor A, et al. Allogeneic hematopoietic stem-cell transplantation in AML and MDS using myeloablative versus reduced-intensity conditioning: the role of dose intensity. Leukemia. 2006;20:322–8.
Estey E, de Lima M, Tibes R, Pierce S, Kantarjian H, Champlin R, et al. Prospective feasibility analysis of reduced-intensity conditioning (RIC) regimens for hematopoietic stem cell transplantation (HSCT) in elderly patients with acute myeloid leukemia (AML) and high-risk myelodysplastic syndrome (MDS). Blood. 2007;109:1395–400.
Kasper C, Sayer HG, Mugge LO, Schilling K, Scholl S, Issa MC, et al. Combined standard graft-versus-host disease (GvHD) prophylaxis with mycophenolate mofetil (MMF) in allogeneic peripheral blood stem cell transplantation from unrelated donors. Bone Marrow Transplant. 2004;33:65–9.
Barker JN, Byam CE, Kernan NA, Lee SS, Hawke RM, Doshi KA, et al. Availability of cord blood extends allogeneic hematopoietic stem cell transplant access to racial and ethnic minorities. Biol Blood Marrow Transplant. 2010;16:1541–8.
Wingard JR, Majhail NS, Brazauskas R, Wang Z, Sobocinski KA, Jacobsohn D, et al. Long-term survival and late deaths after allogeneic hematopoietic cell transplantation. J Clin Oncol. 2011;29:2230–9.
Socie G, Stone JV, Wingard JR, Weisdorf D, Henslee-Downey PJ, Bredeson C, et al. Long-term survival and late deaths after allogeneic bone marrow transplantation. Late Effects Working Committee of the International Bone Marrow Transplant Registry. N Engl J Med. 1999;341:14–21.
Solomon SR, Sizemore CA, Zhang X, Brown S, Holland HK, Morris LE, et al. Impact of donor type on outcome after allogeneic hematopoietic cell transplantation for acute leukemia. Biol Blood Marrow Transplant. 2016;22:1816–22.
Schroeder T, Rautenberg C, Haas R, Kobbe G. Hypomethylating agents after allogeneic blood stem cell transplantation. Stem Cell Investig. 2016;3:84.
Antar A, Kharfan-Dabaja MA, Mahfouz R, Bazarbachi A. Sorafenib maintenance appears safe and improves clinical outcomes in FLT3-ITD acute myeloid leukemia after allogeneic hematopoietic cell transplantation. Clin Lymphoma Myeloma Leuk. 2015;15:298–302.
Giralt S, Ballen K, Rizzo D, Bacigalupo A, Horowitz M, Pasquini M, et al. Reduced-intensity conditioning regimen workshop: defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol Blood Marrow Transplant. 2009;15:367–9.
Luger SM, Ringden O, Zhang MJ, Perez WS, Bishop MR, Bornhauser M, et al. Similar outcomes using myeloablative vs reduced-intensity allogeneic transplant preparative regimens for AML or MDS. Bone Marrow Transplant. 2012;47:203–11.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9.
Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME, et al. Validation and refinement of the disease risk index for allogeneic stem cell transplantation. Blood. 2014;123:3664–71.
Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transplant. 2005;11:945–56.
Duncan CN, Majhail NS, Brazauskas R, Wang Z, Cahn JY, Frangoul HA, et al. Long-term survival and late effects among one-year survivors of second allogeneic hematopoietic cell transplantation for relapsed acute leukemia and myelodysplastic syndromes. Biol Blood Marrow Transplant. 2015;21:151–8.
Brunner AM, Li S, Fathi AT, Wadleigh M, Ho VT, Collier K, et al. Haematopoietic cell transplantation with and without sorafenib maintenance for patients with FLT3-ITD acute myeloid leukaemia in first complete remission. Br J Haematol. 2016;175:496–504.
Brunner AM, Fathi AT, Chen YB. Life after transplant: are we becoming high maintenance in AML? Bone Marrow Transplant. 2016;51:1423–30.
Bashey A, Zhang X, Jackson K, Brown S, Ridgeway M, Solh M, et al. Comparison of outcomes of hematopoietic cell transplants from T-replete haploidentical donors using post-transplantation cyclophosphamide with 10 of 10 HLA-A, -B, -C, -DRB1, and -DQB1 allele-matched unrelated donors and HLA-identical sibling donors: a multivariable analysis including disease risk index. Biol Blood Marrow Transplant. 2016;22:125–33.
Ciurea SO, Zhang MJ, Bacigalupo AA, Bashey A, Appelbaum FR, Aljitawi OS, et al. Haploidentical transplant with posttransplant cyclophosphamide vs matched unrelated donor transplant for acute myeloid leukemia. Blood. 2015;126:1033–40.
Solh M, Zhang X, Connor K, Brown S, Solomon SR, Morris LE, et al. Factors predicting graft-versus-host disease-free, relapse-free survival after allogeneic hematopoietic cell transplantation: multivariable analysis from a single center. Biol Blood Marrow Transplant. 2016;22:1403–9.
Savani BN, Griffith ML, Jagasia S, Lee SJ. How I treat late effects in adults after allogeneic stem cell transplantation. Blood. 2011;117:3002–9.
Battiwalla M, Hashmi S, Majhail N, Pavletic S, Savani BN, Shelburne N. National institutes of health hematopoietic cell transplantation late effects initiative: developing recommendations to improve survivorship and long-term outcomes. Biol Blood Marrow Transplant. 2017;23:6–9.
Gea-Banacloche J, Komanduri KV, Carpenter P, Paczesny S, Sarantopoulos S, Young JA, et al. National institutes of health hematopoietic cell transplantation late effects initiative: the immune dysregulation and pathobiology working group report. Biol Blood Marrow Transplant. 2017;23:870–81.
Hashmi SK, Bredeson C, Duarte RF, Farnia S, Ferrey S, Fitzhugh C, et al. National institutes of health blood and marrow transplant late effects initiative: the healthcare delivery working group report. Biol Blood Marrow Transplant. 2017;23:717–25.
Bevans M, El-Jawahri A, Tierney DK, Wiener L, Wood WA, Hoodin F, et al. National institutes of health hematopoietic cell transplantation late effects initiative: the patient-centered outcomes working group report. Biol Blood Marrow Transplant. 2017;23:538–51.
Morton LM, Saber W, Baker KS, Barrett AJ, Bhatia S, Engels EA, et al. National institutes of health hematopoietic cell transplantation late effects initiative: the subsequent neoplasms working group report. Biol Blood Marrow Transplant. 2017;23:1208–14.
Shaw BE, Hahn T, Martin PJ, Mitchell SA, Petersdorf EW, Armstrong GT, et al. National institutes of health hematopoietic cell transplantation late effects initiative: the research methodology and study design working group report. Biol Blood Marrow Transplant. 2017;23:10–23.
Armenian SH, Chemaitilly W, Chen M, Chow EJ, Duncan CN, Jones LW, et al. National Institutes of health hematopoietic cell transplantation late effects initiative: the cardiovascular disease and associated risk factors working group report. Biol Blood Marrow Transplant. 2017;23:201–10.
Solomon SR, Sizemore C, Zhang X, Ridgeway M, Solh M, Morris LE, et al. Current Graft-versus-host disease-free, relapse-free survival: a dynamic endpoint to better define efficacy after allogenic transplant. Biol Blood Marrow Transplant. 2017.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was presented at ASH Annual meeting held in December 2016 at San Diego, CA.
Rights and permissions
About this article
Cite this article
Solh, M.M., Bashey, A., Solomon, S.R. et al. Long term survival among patients who are disease free at 1-year post allogeneic hematopoietic cell transplantation: a single center analysis of 389 consecutive patients. Bone Marrow Transplant 53, 576–583 (2018). https://doi.org/10.1038/s41409-017-0076-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-017-0076-2
This article is cited by
-
Late cardiac events after allogeneic stem cell transplant: incidence, risk factors, and impact on overall survival
Cardio-Oncology (2023)
-
Chimerism analysis for clinicians: a review of the literature and worldwide practices
Bone Marrow Transplantation (2022)
-
Phenotypical diversity of airway morphology in chronic lung graft vs. host disease after stem cell transplantation
Modern Pathology (2019)