Abstract
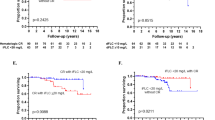
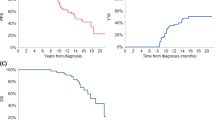
Use of melphalan in multiple myeloma was observed to have a deleterious effect on stem cell collection in older studies. There is limited data on the impact of melphalan in light chain (AL) amyloidosis, especially in the plerixafor era. We retrospectively evaluated stem cell mobilization in 610 patients with AL amyloidosis, of which 79 had prior exposure to melphalan, 167 to other chemotherapeutics, while 364 had no chemotherapy exposure. Collection of CD34+ stem cells × 106/kg was lower in the melphalan group. Median total yields in the melphalan, non-melphalan, and no chemotherapy groups were 5.5, 7.7, and 7.8, respectively; p < 0.001. Day-1 yields were 2.7, 3.5, and 4.0 (p = 0.0003), respectively, and median yields per collection were 2.0, 3.3, and 4.0 (p < 0.001), respectively. Similar results were observed in the sub-group analysis after plerixafor was integrated in our collection algorithm (2009). Patients in the melphalan group had higher failure rate of 9% vs. 2% each in the other two groups (p = 0.006). Impact of melphalan was dose-dependent, with cumulative melphalan exposure of >150 mg (median: three cycles) resulting in lower yields. Therefore, duration of melphalan exposure prior to stem cell collection should be limited, ideally, not exceeding more than two cycles of treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Dispenzieri A, Buadi F, Kumar SK, Reeder CB, Sher T, Lacy MQ, et al. Treatment of immunoglobulin light chain amyloidosis: mayo stratification of myeloma and risk-adapted therapy (mSMART) consensus statement. Mayo Clin Proc. 2015;90:1054–81.
Gertz MA, Lacy MQ, Dispenzieri A, Kumar SK, Dingli D, Leung N, et al. Refinement in patient selection to reduce treatment-related mortality from autologous stem cell transplantation in amyloidosis. Bone Marrow Transplant. 2013;48:557–61.
Mikhael JR, Schuster SR, Jimenez-Zepeda VH, Bello N, Spong J, Reeder CB, et al. Cyclophosphamide-bortezomib-dexamethasone (CyBorD) produces rapid and complete hematologic response in patients with AL amyloidosis. Blood. 2012;119:4391–4.
Boccadoro M, Palumbo A, Bringhen S, Merletti F, Ciccone G, Richiardi L, et al. Oral melphalan at diagnosis hampers adequate collection of peripheral blood progenitor cells in multiple myeloma. Haematologica. 2002;87:846–50.
Desikan KR, Tricot G, Munshi NC, Anaissie E, Spoon D, Fassas A, et al. Preceding chemotherapy, tumour load and age influence engraftment in multiple myeloma patients mobilized with granulocyte colony-stimulating factor alone. Br J Haematol. 2001;112:242–7.
Jerjis S, Croockewit S, Preijers F, Schaap N, De Witte T. Impact of chemotherapy on the mobilisation, harvest and economic costs of autologous peripheral stem cell transplantation in patients with multiple myeloma. Leuk Lymphoma. 2000;37:551–60.
Kazmi MA, Ahsan G, Schey SA. The effects of prior induction therapy with melphalan on subsequent peripheral blood progenitor cell transplantation for myeloma. Clin Lab Haematol. 2001;23:125–9.
Knudsen LM, Rasmussen T, Jensen L, Johnsen HE. Reduced bone marrow stem cell pool and progenitor mobilisation in multiple myeloma after melphalan treatment. Med Oncol. 1999;16:245–54.
Wuchter P, Ran D, Bruckner T, Schmitt T, Witzens-Harig M, Neben K, et al. Poor mobilization of hematopoietic stem cells-definitions, incidence, risk factors, and impact on outcome of autologous transplantation. Biol Blood Marrow Transplant. 2010;16:490–9.
de la Rubia J, Blade J, Lahuerta JJ, Ribera JM, Martinez R, Alegre A, et al. Effect of chemotherapy with alkylating agents on the yield of CD34+ cells in patients with multiple myeloma. Results of the Spanish Myeloma Group (GEM) Study. Haematologica. 2006;91:621–7.
De Clercq E. The AMD3100 story: the path to the discovery of a stem cell mobilizer (Mozobil). Biochem Pharmacol. 2009;77:1655–64.
DiPersio JF, Micallef IN, Stiff PJ, Bolwell BJ, Maziarz RT, Jacobsen E, et al. Phase III prospective randomized double-blind placebo-controlled trial of plerixafor plus granulocyte colony-stimulating factor compared with placebo plus granulocyte colony-stimulating factor for autologous stem-cell mobilization and transplantation for patients with non-Hodgkin’s lymphoma. J Clin Oncol. 2009;27:4767–73.
DiPersio JF, Stadtmauer EA, Nademanee A, Micallef IN, Stiff PJ, Kaufman JL, et al. Plerixafor and G-CSF versus placebo and G-CSF to mobilize hematopoietic stem cells for autologous stem cell transplantation in patients with multiple myeloma. Blood. 2009;113:5720–6.
Dhakal B, Strouse C, D’Souza A, Arce-Lara C, Esselman J, Eastwood D, et al. Plerixafor and abbreviated-course granulocyte colony-stimulating factor for mobilizing hematopoietic progenitor cells in light chain amyloidosis. Biol Blood Marrow Transplant. 2014;20:1926–31.
Kaul E, Shah G, Chaulagain C, Comenzo RL. Plerixafor and G-CSF for autologous stem cell mobilization in AL amyloidosis. Bone Marrow Transplant. 2014;49:1233.
Micallef IN, Sinha S, Gastineau DA, Wolf R, Inwards DJ, Gertz MA, et al. Cost-effectiveness analysis of a risk-adapted algorithm of plerixafor use for autologous peripheral blood stem cell mobilization. Biol Blood Marrow Transplant. 2013;19:87–93.
JMP®, Version 12. SAS Institute Inc., Cary, NC, 1989–2007.
Kumar S, Dispenzieri A, Lacy MQ, Hayman SR, Buadi FK, Gastineau DA, et al. Impact of lenalidomide therapy on stem cell mobilization and engraftment post-peripheral blood stem cell transplantation in patients with newly diagnosed myeloma. Leukemia. 2007;21:2035–42.
Muchtar E, Gertz MA, Kumar SK, Lacy MQ, Dingli D, Buadi FK, et al. Improved outcomes for newly diagnosed AL amyloidosis between 2000 and 2014: cracking the glass ceiling of early death. Blood. 2017;129:2111–9.
Palladini G, Milani P, Foli A, Obici L, Lavatelli F, Nuvolone M, et al. Oral melphalan and dexamethasone grants extended survival with minimal toxicity in AL amyloidosis: long-term results of a risk-adapted approach. Haematologica. 2014;99:743–50.
Gertz MA, Lacy MQ, Dispenzieri A, Buadi FK, Dingli D, Hayman SR, et al. Stem cell transplantation compared with melphalan plus dexamethasone in the treatment of immunoglobulin light-chain amyloidosis. Cancer. 2016;122:2197–205
Clinicaltrials.gov. A randomized open-label Multicenter Phase III Trial of Melphalan and Dexamethasone (MDex) Versus Bortezomib, Melphalan and Dexamethasone (BMDex) for untreated patients with systemic light-chain (AL) amyloidosis. 2017. https://clinicaltrials.gov/ct2/show/NCT01277016.
Galsky MD, Pal SK, Chowdhury S, Harshman LC, Crabb SJ, Wong YN, et al. Comparative effectiveness of gemcitabine plus cisplatin versus methotrexate, vinblastine, doxorubicin, plus cisplatin as neoadjuvant therapy for muscle-invasive bladder cancer. Cancer. 2015;121:2586–93.
Ibrahim JG, Chu H, Chen M-H. Missing data in clinical studies: issues and methods. J Clin Oncol. 2012;30:3297–303.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Sidana, S., Tandon, N., Gertz, M.A. et al. Impact of prior melphalan exposure on stem cell collection in light chain amyloidosis. Bone Marrow Transplant 53, 326–333 (2018). https://doi.org/10.1038/s41409-017-0020-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-017-0020-5