Abstract
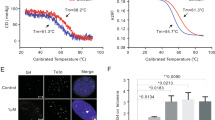
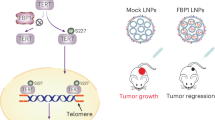
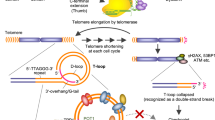
Telomere repeat binding factor 2 (TRF2), a critical element of the shelterin complex, plays a vital role in the maintenance of genome integrity. TRF2 overexpression is found in a wide range of malignant cancers, whereas its down-regulation could cause cell death. Despite its potential role, the selectively small-molecule inhibitors of TRF2 and its therapeutic effects on liver cancer remain largely unknown. Our clinical data combined with bioinformatic analysis demonstrated that TRF2 is overexpressed in liver cancer and that high expression is associated with poor prognosis. Flavokavain B derivative FKB04 potently inhibited TRF2 expression in liver cancer cells while having limited effects on the other five shelterin subunits. Moreover, FKB04 treatment induced telomere shortening and increased the amounts of telomere-free ends, leading to the destruction of T-loop structure. Consequently, FKB04 promoted liver cancer cell senescence without modulating apoptosis levels. In corroboration with these findings, FKB04 inhibited tumor cell growth by promoting telomeric TRF2 deficiency-induced telomere shortening in a mouse xenograft tumor model, with no obvious side effects. These results demonstrate that TRF2 is a potential therapeutic target for liver cancer and suggest that FKB04 may be a selective small-molecule inhibitor of TRF2, showing promise in the treatment of liver cancer.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Wang C, Cao Y, Yang C, Bernards R, Qin W. Exploring liver cancer biology through functional genetic screens. Nat Rev Gastroenterol Hepatol. 2021;18:690–704.
Li X, Ramadori P, Pfister D, Seehawer M, Zender L, Heikenwalder M. The immunological and metabolic landscape in primary and metastatic liver cancer. Nat Rev Cancer. 2021;21:541–57.
Alemi F, Malakoti F, Vaghari-Tabari M, Soleimanpour J, Shabestani N, Sadigh A, et al. DNA damage response signaling pathways as important targets for combination therapy and chemotherapy sensitization in osteosarcoma. J Cell Physiol. 2022;237:2374–86.
Ruman U, Fakurazi S, Masarudin M, Hussein M. Nanocarrier-based therapeutics and theranostics drug delivery systems for next generation of liver cancer nanodrug modalities. Int J Nanomed. 2020;15:1437–56.
Ningarhari M, Caruso S, Hirsch T, Bayard Q, Franconi A, Védie A, et al. Telomere length is key to hepatocellular carcinoma diversity and telomerase addiction is an actionable therapeutic target. J Hepatol. 2021;74:1155–66.
Lin S, Nascimento E, Gajera C, Chen L, Neuhöfer P, Garbuzov A, et al. Distributed hepatocytes expressing telomerase repopulate the liver in homeostasis and injury. Nature. 2018;556:244–8.
Sirma H, Kumar M, Meena JK, Witt B, Weise JM, Lechel A, et al. The promoter of human telomerase reverse transcriptase is activated during liver regeneration and hepatocyte proliferation. Gastroenterology. 2011;141:326–37, 337.e1-3.
Amir M, Khan P, Queen A, Dohare R, Alajmi M, Hussain A, et al. Structural features of nucleoprotein CST/shelterin complex involved in the telomere maintenance and its association with disease mutations. Cells. 2020;9:359.
Davinelli S, Trichopoulou A, Corbi G, De Vivo I, Scapagnini G. The potential nutrigeroprotective role of Mediterranean diet and its functional components on telomere length dynamics. Ageing Res Rev. 2019;49:1–10.
Yu E, Cheung I, Feng Y, Rabie M, Roboz G, Guzman M, et al. Telomere trimming and DNA damage as signatures of high risk neuroblastoma. Neoplasia. 2019;21:689–701.
Nguyen L, Zhao J, Cao D, Dang X, Wang L, Lian J, et al. Inhibition of TRF2 accelerates telomere attrition and DNA damage in naïve CD4 T cells during HCV infection. Cell Death Dis. 2018;9:900.
Nguyen L, Nguyen L, Zhao J, Schank M, Dang X, Cao D, et al. TRF2 inhibition rather than telomerase disruption drives CD4T cell dysfunction during chronic viral infection. J Cell Sci. 2022;135:jcs259481.
Kar M, Sultania M, Roy S, Padhi S, Banerjee B. TRF2 overexpression at the surgical resection margin: a potential predictive biomarker in oral squamous cell carcinoma for recurrence. Indian J Surg Oncol. 2021;12:46–51.
Ozden S, Tiber P, Ozgen Z, Ozyurt H, Serakinci N, Orun O. Expression of TRF2 and its prognostic relevance in advanced stage cervical cancer patients. Biol Res. 2014;47:61.
Chen X, Dong Y, Guo T, Yang C, Chen Y, Sun H. N-terminal modified cyclopeptidic mimetics of Apollo as inhibitors of TRF2. Bioorg Med Chem Lett. 2020;30:127401.
Miyata K, Ukawa M, Mohri K, Fujii K, Yamada M, Tanishita S, et al. Biocompatible polymers modified with d-octaarginine as an absorption enhancer for nasal peptide delivery. Bioconjug Chem. 2018;29:1748–55.
Zheng X, Nie X, Fang Y, Zhang Z, Xiao Y, Mao Z, et al. A cisplatin derivative tetra-Pt(bpy) as an oncotherapeutic agent for targeting ALT cancer. J Natl Cancer Inst. 2017;109. https://doi.org/10.1093/jnci/djx061.
Ge Y, Wu S, Xue Y, Tao J, Li F, Chen Y, et al. Preferential extension of short telomeres induced by low extracellular pH. Nucleic Acids Res. 2016;44:8086–96.
Shen Z, Zheng R, Yang H, Xing S, Jin X, Yan H, et al. G-quadruplex stabilizer Tetra-Pt(bpy) disrupts telomere maintenance and impairs FAK-mediated migration of telomerase-positive cells. Int J Biol Macromol. 2022;213:858–70.
Qiu Y, Yan H, Zheng R, Chen X, Wang Y, Yan Q, et al. cyy260 suppresses the proliferation, migration and tumor growth of osteosarcoma by targeting PDGFR-β signaling pathway. Chem Biol Interact. 2022;367:110200.
Dinami R, Petti E, Porru M, Rizzo A, Ganci F, Sacconi A, et al. TRF2 cooperates with CTCF for controlling the oncomiR-193b-3p in colorectal cancer. Cancer Lett. 2022;533:215607.
Kim Y, Jee H, Um J, Kim Y, Bae S, Yun J. Cooperation between p21 and Akt is required for p53-dependent cellular senescence. Aging Cell. 2017;16:1094–103.
Heba A, Toupance S, Arnone D, Peyrin-Biroulet L, Benetos A, Ndiaye N. Telomeres: new players in immune-mediated inflammatory diseases? J Autoimmun. 2021;123:102699.
Tao L, Zhang W, Zhang Y, Zhang M, Zhang Y, Niu X, et al. Caffeine promotes the expression of telomerase reverse transcriptase to regulate cellular senescence and aging. Food Funct. 2021;12:2914–24.
Zhang Z, Zhang T, Ge Y, Tang M, Ma W, Zhang Q, et al. 2D gel electrophoresis reveals dynamics of t-loop formation during the cell cycle and t-loop in maintenance regulated by heterochromatin state. J Biol Chem. 2019;294:6645–56.
Timashev L, Babcock H, Zhuang X, de Lange T. The DDR at telomeres lacking intact shelterin does not require substantial chromatin decompaction. Genes Dev. 2017;31:578–89.
Hu X, Lv X, Wang R, Long H, Feng J, Wang B, et al. Optimization of N-phenylpropenoyl-l-amino acids as potent and selective inducible nitric oxide synthase inhibitors for Parkinson’s disease. J Med Chem. 2021;64:7760–77.
Zizza P, Dinami R, Porru M, Cingolani C, Salvati E, Rizzo A, et al. TRF2 positively regulates SULF2 expression increasing VEGF-A release and activity in tumor microenvironment. Nucleic Acids Res. 2019;47:3365–82.
de Lange T. Shelterin-mediated telomere protection. Annu Rev Genet. 2018;52:223–47.
Diehl M, Idowu M, Kimmelshue K, York T, Jackson-Cook C, Turner K, et al. Elevated TRF2 in advanced breast cancers with short telomeres. Breast Cancer Res Treat. 2011;127:623–30.
El Maï M, Wagner K, Michiels J, Ambrosetti D, Borderie A, Destree S, et al. The telomeric protein TRF2 regulates angiogenesis by binding and activating the PDGFRβ promoter. Cell Rep. 2014;9:1047–60.
Biroccio A, Rizzo A, Elli R, Koering C, Belleville A, Benassi B, et al. TRF2 inhibition triggers apoptosis and reduces tumourigenicity of human melanoma cells. Eur J Cancer. 2006;42:1881–8.
Wang Z, Wu X. Abnormal function of telomere protein TRF2 induces cell mutation and the effects of environmental tumor‑promoting factors (Review). Oncol Rep. 2021;46:184.
Denham J, Sellami M. Exercise training increases telomerase reverse transcriptase gene expression and telomerase activity: A systematic review and meta-analysis. Ageing Res Rev. 2021;70:101411.
Guo T, Dong Y, Chen Y, Liu L, Sun H. Development and optimization of a cascade of screening assays for inhibitors of TRF2. Anal Biochem. 2020;602:113796.
El Maï M, Janho Dit Hreich S, Gaggioli C, Roisin A, Wagner N, Ye J, et al. A novel screen for expression regulators of the telomeric protein TRF2 identified small molecules that impair TRF2 dependent immunosuppression and tumor growth. Cancers. 2021;13:2998.
Choubey D, Panchanathan R. IFI16, an amplifier of DNA-damage response: role in cellular senescence and aging-associated inflammatory diseases. Ageing Res Rev. 2016;28:27–36.
Doksani Y, Wu J, de Lange T, Zhuang X. Super-resolution fluorescence imaging of telomeres reveals TRF2-dependent T-loop formation. Cell. 2013;155:345–56.
Van Ly D, Low R, Frölich S, Bartolec T, Kafer G, Pickett H, et al. Telomere loop dynamics in chromosome end protection. Mol Cell. 2018;71:510–25.e6.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82273788, 81973168, 82273995), the Natural Science Foundation of Zhejiang Province (LZ22H300002), Wenzhou Science and Technology Plan Project (Y2023180, Y20220200, Y20220195), and Wenzhou Key Laboratory of Research and Transformation of Chinese Medicine. We also thank the Scientific Research Center of Wenzhou Medical University for the consultation and instrument availability that supported this work.
Author information
Authors and Affiliations
Contributions
ZGL and XHZ designed the study and supervised the experimental work; YDQ, QY, Yi Wang, YFY, Yan Wang, MYW, PPW, and DLW performed experiments; SYZ, HY, and JR analyzed data; ZGL, XHZ, and YDQ wrote the manuscript; YJZ, LHH, NC, and KW polished the language of the manuscript. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qiu, Yd., Yan, Q., Wang, Y. et al. Discovery of a selective TRF2 inhibitor FKB04 induced telomere shortening and senescence in liver cancer cells. Acta Pharmacol Sin (2024). https://doi.org/10.1038/s41401-024-01243-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41401-024-01243-6