Abstract
Background
Elevated body mass index (BMI) has been associated with cardiac changes, such as higher epicardial adipose tissue (EAT) thickness. This fat has been identified as a predictive factor of cardiovascular diseases during adulthood. However, few studies have tested the association of multiple cardiovascular risk factors (high weight or blood pressure) with EAT in adolescents and children. Therefore, the main objective of this current research was to determine the impact of BMI, overweight, obesity, and blood pressure on EAT thickness in children.
Methods
A descriptive cross-sectional study focused on elementary and high school students aged 6–16 years was carried out by utilizing diverse measurements and instruments, such as echocardiography.
Results
EAT thickness (N = 228) was linked to sex (more predominant in boys 2.3 ± 0.6; p = 0.044), obesity (2.3 ± 0.6; p < 0.001), and hypertension (2.6 ± 0.6; p = 0.036). The logistic regression indicated that age, sex, and BMI seemed to be more relevant factors in EAT thickness in children (adjusted R square = 0.22; p < 0.001).
Conclusions
This paper examined the associations of sex, age, and cardiovascular risk factors (arthrometric measures and blood pressure) with EAT thickness, indicating that it is necessary to assess whether the findings are associated with future events.
Impact
-
Excessive weight gain and blood pressure in the early stages of life have been associated with adipose tissue. This increase in weight and blood pressure has been attributed to alterations in the epicardial adipose tissue linked to anthropometric markers in adults, but no related study has been implemented in Spanish children.
-
This study revealed how higher epicardial adipose tissue is linked to body mass index, other anthropometric parameters, and blood pressure in Spanish children. These measurements are related to high epicardial adipose tissue thickness, which in early stages does not imply pathology but increases the risk of developing cardiovascular diseases.
Similar content being viewed by others
Introduction
Excessive body fat is a significant risk factor for metabolic,1 hormonal,2 and immunological alterations.3 The prevalence of elevated body mass index (BMI), which can cause serious diseases,3 has increased in recent decades. This shift is reflected by the overweight and obesity rates, resulting in a high prevalence of other health diseases, such as hypertension and diabetes.4,5 In Europe, it is estimated that one out of four individuals is overweight and one out of ten is obese.6
Additionally, high BMI has been associated with increased visceral fat deposits and other cardiac and cardiovascular modifications.7 Additionally, visceral fat deposits have been defined as a marker of cardiovascular risk related to other risk factors, such as metabolic syndrome, diabetes, coronary artery diseases, and arteriosclerosis.8 Moreover, structural changes in the cardiac geometry translate into increased wall thickness and left ventricle mass and index, which are reflected by diverse markers9,10 including the left ventricular end-systolic cavity dimension.11,12 These modifications also seem to be appreciable by analyzing epicardial fat13,14; for instance, epicardial adipose tissue (EAT) thickness has been studied as a possible marker of cardiac modifications and metabolic diseases.14,15 EAT has been highlighted as a potential predictive marker of multiple illnesses, such as hypertension, metabolic syndrome, and cardiovascular disease (CVD).16,17 As a risk factor, EAT thickness has been partially associated with high blood pressure (HBP), subclinical coronary artery diseases, and metabolic disorders.9,18,19,20
The diagnosis of increased EAT thickness is made through echocardiography. Increased BMI, waist circumference (WC), or other anthropometric measurements of adiposity may be useful as indirect predictors of increased EAT.21 Additionally, the combination of BMI and WC, by which visceral fat alteration diagnoses during childhood can be analyzed,18 could be used as a predictor of EAT and, therefore, the risk of CVD.9 Nevertheless, imaging measures have become more important,17,20 despite the cost of imaging measurements, such as MRI. In addition, in recent years, other imaging detection techniques in adults, such as echocardiography, have increased due to their low cost and accessibility.16,17 The use of echocardiography for determining EAT thickness in adults has been improved based on its specific capacity to detect fat modification as a possible indirect marker of CVD and metabolic changes.22
The relevance of EAT as a possible marker for other diseases and its association with diverse parameters, such as BMI, has been mainly established in adults.7,15 More recent articles have indicated that EAT in overweight children tends to be similar to that identified in adults, indicating possible diagnostic value.10,23,24 One study that used echocardiography indicated no significant associations between EAT thickness and diseases in children, specifically metabolic syndrome.25
Despite the importance of determining EAT thickness for its possible predictive value for CVDs and the associations of EAT with indirect measures linked to excess adiposity,16,17,26 little research has combined the indirect (BMI, WC, or BP) and direct (echography) measures in pediatric populations.16 Nonetheless, determining the associations of diverse anthropometric measurements with EAT thickness through echocardiographic screening in children is highly relevant as a noninvasive approach for the early detection and prevention of cardiac problems, especially in primary healthcare and general practice.23,27
Therefore, the main objective of this current research was to determine the impact of BMI, overweight and obesity rates, and BP on EAT thickness in children. The secondary objective was to analyze the associations of indirect measures, such as WC, with EAT thickness.
Methods
Design, procedure, and sample
This descriptive cross-sectional study focused on elementary and high school students aged 6–16 years (95% Caucasian) from a small size town in southern Spain (approximately 3000 inhabitants with an average household income per capita of 19,416 00 eur).
Before the inclusion of participants, informative talks were held for parents and students in each educational center. In these meetings, the research objectives and methodology were explained, questions that arose were answered, and a contact email and a telephone number where any subsequent consultation could be made were provided. After reading the participant information statement, consent form, and questionnaires, and signed the consented form by the parents or legal guardians and the students themselves, participants were able to take part in this research.
The inclusion criteria were age, parent or legal guardian acceptance at the time of starting the study, and signed informed consent by the parents or legal guardians, the student, and the principal researcher. The exclusion criteria were children under 6 years and adolescents over 16 years of age who were excluded when the study began or had not signed the informed consent form. The anthropometric measurements and cardiography data of each participant were gathered by two general practitioners in the different centers. Such centers did not have a health consultation facility, but they were close to the primary health center. The anthropometric indices, BP, and physical measurements were obtained in each elementary and high educational center during three or four sessions, generally on a Friday, in which groups between eight and ten children were assessed. Additionally, the echocardiography was taken in the town’s primary health center by appointment.
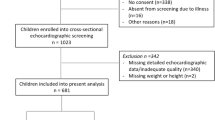
The participants were recruited to participate from March 2018 to February 2019. A nonprobability convenience sample of 254 participants was recruited from March 2018 to February 2019. Most participants agreed to undergo the ultrasound (238; 93.7%) and after gathering all data the final sample totaled 228 subjects (120 boys, ages ranging between 11 and 12 years and 108 girls with ages between 10 and 11 years) (Fig. 1). The girls were significantly younger than the boys with a difference of 8 months (p < 0.05).
Measures and instruments
Anthropometric measurements
Anthropometric variables were measured following the International Standards for Anthropometric Assessment28 using a portable model Seca 213® (Seca, Hammer Steindamm, Hamburgo, Germany). Weight, BMI, percentage of fat mass and lean mass, and total body water composition were obtained with a model Omron BF-511 (Omron Healthcare Europe B.V., Hoofddorp, Netherlands) impedance meter, classified according to Melo Salor.29
BMI was calculated by the BMI equation, and BMI was categorized according to Melo Salor’s tables29 and the World Health Organization.30 The selection of these tables and the structure of BMI to determine the normal weight, overweight, and obesity was based on the modeling of z scores. In this sense, BMI groups29 were grouped using the cut points according to age and sex, for instance, 12-year-old girls have up to 14.4 (underweight), 20.8 (normal weight), 25.0 (overweight), and >31.9 (obesity).
To determine the WC, an inextensible measuring tape was used, and the waist–height ratio (WHtR) was calculated by dividing the WC by the height (cm). The body fat percentage (BF%) and fat-free (FF%) mass were calculated using the Faulkner equation.31
Blood pressure
BP measurements were performed with an automatic sphygmomanometer: the Omron M6 comfort® model. BP was measured following the European Society for Arterial Hypertension recommendations for children and adolescents.32 HBP was defined as at least three successive SBP or DBP measures greater than or equal to the 95th percentile (P95) for a given age, sex, and height percentile.
Regulated echocardiogram
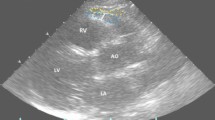
Each subject underwent a two-dimensional (2D) transthoracic echocardiogram. The measurement was carried out via a standard technique with the patients in the left lateral decubitus position, following the method validated by Iacobellis et al.7 An echocardiographer cardiologist performed the technique. The measurement identified the hypoechoic space anterior to the right ventricular wall, with the EAT thickness determined according to the epicardial surface and the parietal pericardium. This assessment was made following recommendations and previous work,33 so EAT was not confused with pericardial fluid. Two cardiologists evaluated the results and assessed EAT thickness appropriately.
All patients underwent a complete ultrasound study with M-mode, 2D transthoracic echocardiography, and Doppler. Images were taken in the long- and short-axis parasternal projection, at the apical point of four cameras, three cameras, and two cameras, as well as in the subxiphoid plane. A Philips iE33 with probe S5-1 (Koninklijke Philips. Electronics N.V., Eindhoven, Netherlands) was used for the measurement.
EAT was identified as the echolucent space between the outer wall of the myocardium and the visceral layer of the pericardium. This was measured perpendicularly over the free wall of the right ventricle at the end of systole in three cardiac cycles using a long parasternal or short parasternal view. The measurement was performed on the free wall of the right ventricle for two reasons: (1) this point is anatomically recognized as the one with the most significant epicardial fat and (2) the long parasternal and short parasternal axes allow the most accurate measurements of epicardial fat on the right ventricle, with optimal cursor orientation in each view.
Assessment of fitness level
The assessment of fitness level was conducted through the Eurofit battery tests: Endurance, through the Course Navette test,34 and Muscular Strength through three tests: (a) extended jump test without impulse and with feet together to evaluate the explosive strength of the lower body; (b) hand strength test, and (c) abdominal strength test by performing the maximum number of complete sit-ups over 30 min. A physical condition level was obtained for each child and adolescent from the results of the different tests.
Statistical analysis
After obtaining all the data, SPSS (IBM, Armonk, NY) version 28 was used to analyze each measurement. The absolute and relative frequencies were used for categorical variables, such as normal weight or overweight incidence. Additionally, the median was used for such variables. The mean and standard deviation, along with the 95% confidence interval (CI), were used for the quantitative analysis. To test for normality, the Kolmogorov–Smirnov test with Lilliefors correction and Q-Q graphs were used, showing differences between subgroups (BMI groups) (p < 0.05) and normally distributed data in the whole sample (p > 0.05).
Based on the results from the Kolmogorov–Smirnov test, the comparison of two or three independent means was carried out using the Mann–Whitney U, Student’s t test, and analysis of variance (ANOVA), and the Kruskal–Wallis test according to the variable being analyzed. When indicated, the chi2 test with the Yates correction or Fisher’s test was used. Additionally, the two- and three-way ANOVA test for three factors was applied for EAT values and sex, BMI, and hypertension, transforming the variables according to the cut-off points for diagnosing obesity or hypertension.32 Quantitative bivariate correlations were assessed, and Pearson’s correlation coefficient was used. Finally, logistic regression was used to determine associations between the EAT and obesity and hypertension. The stepwise method was applied in logistic regression according to mean EAT thickness. The elimination of the variables of the model was dependent on their p value, and the receiver operating characteristic (ROC) curve was implemented to determine the impact of BMI and other anthropometric measures, as well as personal factors, such as age or sex, on the EAT thickness.
Results
The prevalence of obesity and overweight in the sample (N = 228) was 49.5%. There were significant differences between the obesity or overweight and normal weight children regarding BMI (22.7 ± 3.6 vs. 18.4 ± 4.2; p < 0.001), WC (71.2 ± 10.1 vs. 62.1 ± 10.2; p < 0.001), WHtR (0.5 ± 0.04 vs. 0.4 ± 0.06; p < 0.001), FF% (30.1 ± 5.6 vs. 20.7 ± 8.3; p < 0.001), SPB (113.2 ± 10.2 vs. 108.4 ± 10.7; p = 0.007), hypertension (13.1 vs. 1.6; p < 0.001), jumps (126.2 ± 27.4 vs. 135.8 ± 36.5; p < 0.001), and sit-ups (19.4 ± 4.9 vs. 20.0 ± 6.0; p < 0.001).
Sex differences were assessed for anthropometric indices, BP, and physical activity, showing significant differences between girls and boys (Table 1). Table 1 shows that there were no significant differences in the rate of overweight regarding sex (28.7%, 95% CI 20.7–37.9 vs. 25.8%, 95% CI 16.5–33.4; p = 0.57). The BMI showed a mean of 20.3 with a standard deviation of 4.5 (95% CI 19.7–20.9), and the participants were close to the 85th percentile. The WHtR was 0.45 ± 0.06 with a 95% CI of 0.45–0.46, which was significantly higher in boys than in girls (0.46 ± 0.07 vs. 0.44 ± 0.05; p < 0.05). The BF% was 24.8 ± 8.6 with a 95% CI from 23.7 to 25.9, and the mean FF% was 31.8 ± 4.3, with a 95% CI 31.2–32.3 (33.3 ± 4.7 in boys vs. 30.1 ± 3 in girls; p < 0.001), with no significant differences between sexes for BF%. Finally, these parameters showed significant differences between girls and boys (Table 1).
The BP results showed no differences between boys and girls, except for systolic BP (SBP) (Table 1). Boys had slightly higher levels of diastolic BP (DBP) than girls (68.5 ± 6.5 vs. 68.1 ± 6.6; p = 0.59), hypertension being slightly more prevalent in girls (5.5 vs. 7.3%; p = 0.83). SBP was significantly higher in boys than in girls (112.3 ± 11.1 vs. 108.5 ± 10.1; p < 0.05), with a difference of 3.8 ± 1. Finally, the physical activity test showed a mean value of 4.4 ± 2.4 min with a 95% CI of 4.1–4.7, with no significant difference between boys and girls (4.6 ± 2.3 vs. 4.2 ± 2.1; p = 0.13). In addition, there was a considerable difference regarding the mean number of jumps (140.8 ± 35.6 vs. 121.5 ± 26.1; p < 0.001) and sit-ups (20.7 ± 6.2 vs. 118.6 ± 4.4; p < 0.01) between boys and girls.
The sample was formed by 73 children (32.0%) whose age was between 6 and 9 years, 71 (31.1%) who were between 9 and 12 years, and 84 (36.8%) who were >12 years. The differences between the age groups (children, prepuberal, and adolescents) in sex, anthropometric indices, BP, and physical activity were analyzed, showing no differences between the groups, except for WC (59.8 ± 9.6 vs. 66.1 ± 9.6 vs. 71.6 ± 10.4; p = 0.01), presence of hypertension (1.3 vs. 0.8% vs. 4.2%; p = 0.046), the number of jumps (103.2 ± 16.2 vs. 130.6 ± 20.8 vs. 156.8 ± 32.3; p = 0.001), and sit-ups (16.2 ± 8.8 vs. 19.9 ± 4.3 vs. 21.7 ± 6.5; p = 0.03) (Table 2).
EAT thickness was analyzed according to the ultrasound measurement planes (long and short sternal) as well as overall (mean of the total measurements) and according to sex (Fig. 2), obesity, and arterial hypertension. EAT thickness was higher among boys for the long and global planes than among girls (Table 3). EAT was higher in obese children than in normal weight children (2.2 ± 0.7 vs. 1.8 ± 0.5; p < 0.001 for the long parasternal axis (LPA); 2.4 ± 0.7 vs. 1.8 ± 0.6; p < 0.001 for the short parasternal axis (SPA); 2.3 ± 0.6 vs. 1.8 ± 0.5; p < 0.001 for the global), and the difference was more significant in boys (2.3 ± 0.8 vs. 1.8 ± 0.5; p < 0.001 for the LPA; 2.4 ± 0.8 vs. 1.9 ± 0.6; p < 0.001 for the SPA; 2.4 ± 0.7 vs. 1.8 ± 0.5; p = 0.006 for the global) than in girls (2 ± 0.6 vs. 1.7 ± 0.4; p < 0.05 for the LPA; 2.2 ± 0.7 vs. 1.7 ± 0.5; p < 0.001 for the SPA; 2.1 ± 0.4 vs. 1.7 ± 0.4; p < 0.001 for the global). Additionally, the EAT thickness was higher in hypertensive children (2.2 ± 0.7 vs. 1.8 ± 0.5; p = 0.031 for LPA and 2.2 ± 0.7 vs. 1.8 ± 0.5; p = 0.031 for the global). Moreover, the two-way ANOVA for the three factors (sex, hypertension, and obesity) (Table 3) indicated that sex, obesity, and hypertension were related to EAT thickness. The global EAT thickness was significantly different according to obesity and hypertension (2.7 ± 0.6 vs. 1.7 ± 0.5; p = 0.05), with no differences between girls and boys in regard to hypertension and obesity. The results among the boys indicated that EAT thickness was mediated by the presence of obesity (2.4 ± 0.7 vs. 1.8 ± 0.5; p = 0.006 for the global), which was the same for the girls (2.1 ± 0.4 vs. 1.7 ± 0.4; p < 0.001 for the global). Additionally, the girls with obesity and HBP had more diverse EAT thickness values than girls who did not have both indicators (2.6 ± 0.6 vs. 1.6 ± 0.4; p = 0.036).
A deeper analysis was carried out using logistic regression based on the significant association between EAT thickness and BMI (Fig. 3a) and EAT and BP (Fig. 3b). Figure 3a shows the area under the curve (AUC) of the ROC curve, showing significant values for LPA (0.76 with a 95% CI from 0.682 to 0.833), SPA (0.76; 95% CI of 0.682–0.833), and global (0.746; 95% of 0.67–0.813) (p < 0.001). The analysis focused on hypertension and EAT thickness measured in the SPA, which obtained the highest discriminant capacity (0.723, 95% CI of 0.59–0.852; p < 0.001), followed by the global parasternal axis (0.677 with a 95% CI of 0.531–0.824; p < 0.05) (Fig. 3b).
Representation of the association and significant values based on the logistic regression between obesity rate (a) and hypertension (b) related to the long, short and global epicardial adipose tissue thickness (represented in the figures as different colours lines) including the sensibility (axis Y) and specificity (axis X).
In addition, correlation analysis was performed using the coefficient correlation between the quantitative variables and EAT thickness. The results showed that there were associations between global EAT thickness and age (r = 0.13; p < 0.05), height (r = 0.21; p < 0.01), weight (r = 0.39; p < 0.001), BMI (r = 0.43; p < 0.001), SBP (r = 0.23; p < 0.001), DBP (r = 0.16; p < 0.01), WC (r = 0.42; p < 0.001), WHtR (r = 0.34; p < 0.001), and BF% (r = 0.35; p < 0.001). Based on the significance of the results, linear regression was implemented for the previous anthropometric measures and the global thickness. The analysis showed that age and anthropometric indices were significantly associated with the global thickness (Table 4). The analysis indicated high significance for the variables (R square = 0.23; Adjusted R square = 0.22; p < 0.001). Table 4 shows strong associations with global EAT thickness, except for FF%, which had a lower significance value. Therefore, these findings aroused interest in developing a predictive model based on multiple linear regression. BMI was the anthropometric index that obtained the best predictive capacity (goodness of fit, r2). It was found that the combination of more than one anthropometric indicator (compatible to avoid collinearity) did not improve the goodness of fit of the prediction equation. Therefore, the linear equation for the theoretical determination of global EAT thickness is shown in Table 4, where BMI is calculated by dividing the weight (kg) by the height (m2), sex equals one for boys and zero for girls, and age is included in years (Model 1: Determination of global EAT thickness: Global EAT thickness (mm) = 0.82 + 0.055 (BMI) + 0.17 (sex) − 0.014 (age)).
Discussion
This paper examined the association between sex, age, cardiovascular risk factors (anthropometric measures and BP), and EAT thickness.
The current study indicated a high prevalence of overweight and obese children (approximately 25% each), which was associated with sex, BP, physical activity, and EAT thickness. Another factor related to EAT thickness and BMI was hypertension, which has also been increasing in this population. These outcomes have shown that weight alteration is complex and effects affect EAT thickness at similar levels as in adults.10,23
In addition, the ANOVA tests and ROC curves highlighted the effect of BMI and BP on EAT thickness (short, long, and global) among children, although the significant factor in girls and both sexes was BMI. In this sense, the EAT measures (short, long, and global) indicated to be related to different associations with sex, BP, and obesity prediction, being the most relevant the global and long EAT thickness for boy and global and short EAT thickness for girls. Moreover, based on multivariant analysis, the model showed that age, sex, and BMI could be among the most significant factors related to the EAT thickness and was represented in the model in one out of four children.
The obesity and overweight rates we detected in our study were highly unexpected since previous studies have indicated lower frequencies.35,36 The prevalence of obesity in children (5–17 years old) was found to be between 10.2 and 12.6%;35,36 moreover, previous Spanish studies conducted in the 2000s showed that the combination of obese and overweight children was 26.3%,37 with an obesity rate of approximately 15%.37,38
Despite the previous findings from the early 2000s, the current research is more in sync with a recent Spanish study that the prevalence of overweight was 29.4% (9-18 years old) and obesity was 7.0 (9-18 years old), being higher in boys compared to girls (35.6 vs. 22.8 and 8.5 vs. 5.3).39 The obesity rate among Spaniards between 9 to 18 years old in the ENPE’s research39 was higher by five points than the enKid’s study (2006).40 All these results highlight how the tendency of overweight and obesity among Spaniards is highly increasing achieving concerning values.
Besides, the BP results and other anthropometric data, such as WC or BMI, were consistent with other authors regarding the prevalence of hypertension and the relevance of BMI as a relevant indicator of cardiovascular risk factors in children.39,41,42 The physical condition of the sample matched that described in previous studies, showing a value under the health standards for adequate growth,41,42 which seemed related to EAT thickness.
High levels of BMI and BP provoke structural changes in cardiac geometry, mainly causing increased wall thickness,43 which seems to be reflected by EAT thickness. In this sense, previous studies have linked BMI, SBP, and WC, as cardiovascular risk factors, to thicker EAT and, therefore, a higher risk of CVD.10,23,24,44 The previous results and the current findings were in agreement in regard to the relevance of cardiovascular risk factors and cardiac modifications in children. Moreover, the associations between EAT thickness and sex, BP, and obesity prediction match a previous study focused on adults (30 to 64 years) that indicated how long parasternal axes were associated with males.45 These differences between EAT thickness might be caused by age-related changes in sex hormones, although no research in children has indicated such differences.44
In general, most studies focused on the associations between variables but did not differentiate between sexes and assessed only one arthrometric variable (BMI or WHtR) in relation to EAT thickness.44,46 Most studies have focused on the associations between a few arthrometric variables and EAT thickness47,48 and its potential for detecting other diseases, such as metabolic syndrome. Nevertheless, complex models with multiple variables are scarce,23,25,46 indicating the need for more research in this field. In this sense, the model created could have significant value for healthcare professionals in primary care. This model could provide an effective tool for preventing cardiac modifications in 1 out of 4 children, helping to avoid excess adiposity and allowing for early detection of such changes among children or preadolescents.
Even though this research has highlighted some interesting findings, there are some limitations. First, the sample was homogeneous and focused on children from southern Spain who were mainly Caucasian; therefore, the findings should not be applied to other ethnic groups or adolescents. Additionally, the data indicated that BP and BMI were relevant to EAT thickness, and BMI in the model had a predictive factor lower than desirable. Moreover, the model is theoretical, which implies the need for further validation before it can be applied in healthcare or other fields.
Despite these limitations, the implications of this paper are exciting since the study explored associations with more variables (i.e., FF%, BMI, or EAT thickness) in a broader sample of children and conducted a more in-depth analysis resulting in a predictive model. Moreover, the findings may help healthcare workers understand the implications of overweight and obesity and the possible use of noninvasive screening instruments such as BMI, sex, and age to determine EAT thickness. Additionally, the differences regarding the EAT measures (short, long, and global) and sex, BP, and obesity prediction could be highly relevant for physicians since the EAT thickness (short for girls or long for boys and global for both sexes) should be measured, instead of using the long axis for both sexes which is more is most likely to be used. Finally, it would be necessary to assess whether these findings are significantly associated with future events and whether an intervention in this population can reverse the high prevalence of these modifiable cardiovascular risk factors and prevent future events.
Conclusions
In summary, the findings indicated a high rate of overweight and obesity, with high values of various measurements. Additionally, there were associations between cardiovascular risk factors, mainly high weight and BP, and greater EAT thickness. Finally, a model that explained one out of four children was obtained, which could be useful in the future, although it requires further research.
Data availability
The datasets analyzed during the current study are not publicly available due to the inclusion of personal details, identification numbers from the health care system, and further data for future research. Still, they are available from the corresponding author on reasonable request
References
Atzendorf, J., Apfelbacher, C., de Matos, E. G., Kraus, L. & Piontek, D. Patterns of multiple lifestyle risk factors and their link to mental health in the German adult population: a cross-sectional study. BMJ Open 8, e022184 (2018).
Hoare, E. et al. Lifestyle behavioural risk factors and emotional functioning among schoolchildren: The Healthy Growth Study. Eur. Psychiatry 61, 79–84 (2019).
Marck, C. H. et al. Health outcomes and adherence to a healthy lifestyle after a multimodal intervention in people with multiple sclerosis: three year follow-up. PLoS ONE 13, e0197759 (2018).
von Katzler, R. et al. Lifestyle behaviour and prevalence of cardiovascular risk factors - a pilot study comparing Kiribati and European seafarers. BMC Public Health 19, 855 (2019).
Sharma, S., Shivpuri, A. & Shivpuri, A. Childhood obesity: review of a growing problem. Int. J. Clin. Pediatr. Dent. 5, 237–241 (2012).
Brambilla, P., Pozzobon, G. & Pietrobelli, A. Physical activity as the main therapeutic tool for metabolic syndrome in childhood. Int. J. Obes. 35, 16–28 (2011).
Iacobellis, G. et al. Epicardial fat from echocardiography: a new method for visceral adipose tissue prediction. Obes. Res. 11, 304–310 (2003).
Talman, A. H. et al. Epicardial adipose tissue: far more than a fat depot. Cardiovasc. Diagn. Ther. 4, 416–429 (2014).
Rodicio, M. M. et al. Early cardiac abnormalities in obese children and their relationship with adiposity. Nutrition 46, 83–89 (2018).
López-Bermejo, A., Prats-Puig, A., Osiniri, I., Martínez-Calcerrada, J.-M. & Bassols, J. Perirenal and epicardial fat and their association with carotid intima-media thickness in children. Ann. Pediatr. Endocrinol. Metab. 24, 220–225 (2019).
Chumlea, W. M. C., Schubert, C. M., Towne, B., Siervogel, R. M. & Sun, S. S. Left ventricular mass, abdominal circumference and age: The Fels Longitudinal Study. J. Nutr. Health Aging 13, 821 (2019).
Krishnan, R., Becker, R. J., Beighley, L. M. & López-Candales, A. Impact of body mass index on markers of left ventricular thickness and mass calculation: results of a pilot analysis. Echocardiography 22, 203–210 (2005).
Villasante Fricke, A. C. & Iacobellis, G. Epicardial adipose tissue: clinical biomarker of cardio-metabolic risk. Int. J. Mol. Sci. 20, 5989 (2019).
Christensen, R. H., von Scholten, B. J., Lehrskov, L. L., Rossing, P. & Jørgensen, P. G. Epicardial adipose tissue: an emerging biomarker of cardiovascular complications in type 2 diabetes? Ther. Adv. Endocrinol. Metab. 11, 2042018820928824 (2020).
Bertaso, A. G., Bertol, D., Duncan, B. B. & Foppa, M. Epicardial fat: definition, measurements and systematic review of main outcomes. Arq. Bras. Cardiol. 101, e18–e28 (2013).
Toemen, L. et al. Pericardial adipose tissue, cardiac structures, and cardiovascular risk factors in school-age children. Eur. Heart J. Cardiovasc. Imaging 22, 307–313 (2021).
Patro Golab, B., Voerman, E., van der Lugt, A., Santos, S. & Jaddoe, V. W. V. Subcutaneous fat mass in infancy and abdominal, pericardial and liver fat assessed by magnetic resonance imaging at the age of 10 years. Int. J. Obes. 43, 392–401 (2019).
Cabrera-Rego, J. O. et al. Epicardial fat thickness correlates with carotid intima-media thickness, arterial stiffness, and cardiac geometry in children and adolescents. Pediatr. Cardiol. 35, 450–456 (2014).
Kim, S. J. et al. Correlation between epicardial fat thickness by echocardiography and other parameters in obese adolescents. Korean Circ. J. 42, 471–478 (2012).
Cavalcante, J. L. et al. Association of epicardial fat, hypertension, subclinical coronary artery disease, and metabolic syndrome with left ventricular diastolic dysfunction. Am. J. Cardiol. 110, 1793–1798 (2012).
Sinha, S. K. et al. Epicardial adipose tissue thickness and its association with the presence and severity of coronary artery disease in clinical setting: a cross-sectional observational study. J. Clin. Med. Res. 8, 410–419 (2016).
Markopoulou, P., Papanikolaou, E., Analytis, A., Zoumakis, E. & Siahanidou, T. Preterm birth as a risk factor for metabolic syndrome and cardiovascular disease in adult life: a systematic review and meta-analysis. J. Pediatr. 210, 69.e5–80.e5 (2019).
Shin, J.-H. Is the measurement of epicardial fat in obese adolescents valuable? Korean Circ. J. 42, 447 (2012).
Mazur, A., Ostański, M., Telega, G. & Malecka-Tendera, E. Is epicardial fat tissue a marker of metabolic syndrome in obese children? Atherosclerosis 211, 596–600 (2010).
Ahmed, A. M., Ragab, S. H., Ismail, N. A., Awad, M. A. & Kandil, M. E. Echocardiographic assessment of epicardial adipose tissue in obese children and its relation to clinical parameters of metabolic syndrome. J. Saudi Heart Assoc. 25, 108 (2013).
Srikanthan, K., Feyh, A., Visweshwar, H., Shapiro, J. I. & Sodhi, K. Systematic review of metabolic syndrome biomarkers: a panel for early detection, management, and risk stratification in the West Virginian population. Int. J. Med. Sci. 13, 25–38 (2016).
Reyes, Y. et al. Espesor del tejido adiposo epicárdico en niños y adolescentes con factores de riesgo cardiometabólico. Endocrinol. Nutr. 63, 70–78 (2016).
Norton, K. I. in Kinanthropometry and Exercise Physiology (eds Norton, K. & Eston, R.) 68–137 (Routledge, 2018).
Melo Salor, J. Tabla de valores de IMC para sobrepeso y obesidad infantojuvenil de 5 a 19 años según estándares de la OMS. Source: WHO Growth Reference 5-19 years, 2007, https://www.sspa.juntadeandalucia.es/servicioandaluzdesalud/sites/default/files/sincfiles/wsas-media-mediafile_sasdocumento/2019/TablaRapidaIMC_OMS_PIOBIN.pdf (2007).
Zvonar, M., Štefan, L. & Kasović, M. Percentile curves for body-mass index, waist circumference, waist-to-height ratio and waist-to-height ratio(Exp) in Croatian adolescents. Int. J. Environ. Res. Public Health 16, 1920 (2019).
Lozano-Berges, G. et al. Accurate prediction equation to assess body fat in male and female adolescent football players. Int. J. Sport Nutr. Exerc. Metab. 29, 297–302 (2019).
Lurbe, E. et al. 2016 European Society of Hypertension guidelines for the management of high blood pressure in children and adolescents. J. Hypertens. 34, 1887–1920 (2016).
Smulyan, H. The computerized ECG: friend and foe. Am. J. Med. 132, 153–160 (2019).
García, G. C. & Secchi, J. D. Test course navette de 20 metros con etapas de un minuto. Una idea original que perdura hace 30 años. Apunts Med. l’Esport 49, 93–103 (2014).
Farajian, P. et al. Very high childhood obesity prevalence and low adherence rates to the Mediterranean diet in Greek children: the GRECO study. Atherosclerosis 217, 525–530 (2011).
Pérez-Ríos, M. et al. Excess weight and abdominal obesity in Galician children and adolescents. Ann. Pediatr. 89, 302–308 (2018).
Serra-Majem, L. et al. Obesidad infantil y juvenil en España. Resultados del Estudio enKid (1998-2000). Med. Clín. 121, 725–732 (2003).
Schröder, H. et al. Prevalence of abdominal obesity in Spanish children and adolescents. Do we need waist circumference measurements in pediatric practice? PLoS ONE 9, e87549 (2014).
Aranceta-Bartrina, J., Gianzo-Citores, M. & Perez-Rodrigo, C. Prevalence of overweight, obesity and abdominal obesity in the spanish population aged 3 to 24 years. The Enpe Study. Rev. Espanola Cardiol. 73, 290–299 (2020).
Serra-Majem, L., Bartrina, J. A., Perez-Rodrigo, C., Ribas-Barba, L. & Delgado-Rubio, A. Prevalence and deteminants of obesity in Spanish children and young people. Br. J. Nutr. 96, S67–S72 (2006).
Garcia, L. M. T., da Silva, K. S., Del Duca, G. F., da Costa, F. F. & Nahas, M. V. Sedentary behaviors, leisure-time physical inactivity, and chronic diseases in Brazilian workers: a cross sectional study. J. Phys. Act. Health 11, 1622–1634 (2014).
Rose, T. et al. A systematic review of digital interventions for improving the diet and physical activity behaviors of adolescents. J. Adolesc. Health 61, 669–677 (2017).
Valerio, G. et al. Childhood obesity classification systems and cardiometabolic risk factors: a comparison of the Italian, World Health Organization and International Obesity Task Force references. Ital. J. Pediatr. 43, 19 (2017).
van Hoek, E., Koopman, L. P., Feskens, E. J. M. & Janse, A. J. Assessment of epicardial adipose tissue in young obese children. Child Adolesc. Obes. 2, 96–107 (2019).
Cho, D. H. et al. Association between epicardial adipose tissue, high-sensitivity C-reactive protein and myocardial dysfunction in middle-aged men with suspected metabolic syndrome. Cardiovasc. Diabetol. 17, 9 (2018).
Güneş, H., Güneş, H. & Temiz, F. The relationship between epicardial adipose tissue and insulin resistance in obese children. Arq. Bras. Cardiol. 114, 675–682 (2020).
Radgoudarzi, M., Pazouki, A., Fahmfam, Z. & Soheilipour, F. Evaluation of changes in cardiac dimensions and functional parameters and their association with anthropometric parameters and laboratory indices in obese adolescents. Med. Sci. 24, 1520–1527 (2020).
Bedir, A., Mehmet, B. & Cevriye, A. Relationship of epicardial adipose tissue thickness with early indicators of atherosclerosis and cardiac functional changes in obese adolescents with metabolic syndrome. J. Clin. Res. Pediatr. Endocrinol. 5, 156–163 (2013).
Acknowledgements
We would like to thank the parents and children who decided to participate in this research.
Funding
This research did not receive any funding.
Author information
Authors and Affiliations
Contributions
Conceptualization, I.M.B.S., F.J.F.P., and M.D.M.R.; methodology, I.M.B.S., F.J.F.P., and M.D.M.R.; validation, I.M.B.S., FJ.F.P., and P.A.-M.; formal analysis, P.A.-M., M.R.O., I.M.B.S., and M.D.M.R.; investigation, I.M.B.S., C.H.A.-D., J.F.C.; resources, I.M.B.S., C.H.A.-D., J.F.C., FJ.F.P., and M.D.M.R.; writing—original draft preparation, P.A.-M., FJ.F.P., and I.M.B.S.; writing—review and editing, P.A.-M., M.R.O., FJ.F.P., and I.M.B.S.; supervision, FJ.F.P. and M.D.M.R. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This research follows the Helsinki Code and Biomedicine Principles and obtained the Approval from Andalusian Ethical Committee, also known as PEIBA, specifically at the Regional Committee at the Reina Sofia’s Hospital number 2353. Informed consent was obtained by the parents or legal guardians, the student himself, and the principal researcher.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Blancas Sánchez, I.M., Aristizábal-Duque, C.H., Fernández Cabeza, J. et al. Role of obesity and blood pressure in epicardial adipose tissue thickness in children. Pediatr Res 92, 1681–1688 (2022). https://doi.org/10.1038/s41390-022-02022-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-022-02022-x