Abstract
Background
The major increase in the survival rate among children with cancer is due to improvement in the diagnosis and treatment. Despite this increase, childhood cancer survivors (CCS) are at high risk of developing late complications such as nephrotoxicity due to chemotherapy. So, we aimed to detect early subclinical kidney dysfunction among CCS.
Methods
This cross-sectional study was implemented on 52 survivors of childhood cancer recruited from Pediatric Oncology Unit, Menoufia University. Laboratory evaluations for each participant, including complete blood count, serum urea, creatinine, urinary protein, urinary calcium, uric acid, and serum cystatin C and urinary Neutrophil Gelatinase Associated Lipocalin (UrNGAL) by ELISA were obtained.
Results
Estimated GFR was decreased in 23.1% of cases, with elevated serum cystatin C, UrNGAL and UrNGAL/Cr. There was a significant increase of Uprotein/Cr, UCa/Cr, UACR (p = 0.02), UrNGAL and UrNGAL/Cr (P < 0.001) in patients with tubular dysfunction compared without tubular dysfunction. There was a significant difference between two groups regarding cisplatin (P = 0.03) and high-dose methotrexate chemotherapy (p = 0.04). The AUCs for detecting kidney tubular dysfunction by UrNGAL and UrNGAL/Cr were 0.807 and 0.747.
Conclusion
A significant tubular dysfunction among childhood cancer survivors receiving chemotherapy as cisplatin and high-dose methotrexate.
Impact
-
Detection of kidney dysfunction mainly tubular in childhood cancer survivors after finishing chemotherapy.
-
Urinary NGAL is a good predictor for detection of tubular dysfunction in childhood cancer survivors after finishing chemotherapy.
Similar content being viewed by others
Introduction
The survival rate among children with cancer was significantly improved as a result of anti-cancer treatment. 40% of these cancer patients suffer from growth impairment, neurotoxicity, heart failure, nephrotoxicity, hormonal disturbances and secondary cancer as late complications.1 Risk factors of nephrotoxicity include age, innate drug toxicity, anti-cancer treatment (chemotherapy, surgery, and radiation), and pre-existing kidney damage.2 The most common chemotherapy agents which are responsible for kidney injury are ifosfamide and cisplatin, also other anti-cancer drugs as high-dose methotrexate are associated with compromised glomerular function. Anti-cancer therapy for leukemia and lymphoma including high-dose methotrexate. Methotrexate toxicity acts mainly on the hematopoietic system and the intestinal mucosa.3 Kidney damage in CCS may be due to the precipitation of methotrexate and its less soluble metabolites (7-OH-MTX and DAMPA) in acidic urine.4
Cystatin- C due to its small size, is filtered by the glomerulus, reabsorbed and broken down by kidney tubules and involved in glomerular dysfunction.5
Urine neutrophil gelatinase-associated lipocalin (NGAL) is a protein that was extruded from injured proximal tubular cells into the urine. NGAL is involved in tubular cell injury and repair.6
Therefore, we aimed to estimate serum cystatin-C for detecting kidney glomerular dysfunction and urine NGAL for detecting kidney tubular dysfunction in survivors of pediatric cancer.
Subjects and methods
Design
A cross-sectional study was implemented on fifty-two survivors of pediatric cancer and was evaluated between May 2021 and September 2021. The 25 girls and 27 boys were 4–18 years of age at the time of evaluation; they were recruited from Oncology Unit, Pediatrics Department, Menoufia University. Prior to blood samples collection, a written informed consent was approved from the Ethics Committee of Faculty of Medicine, Menoufia University (ID: 24/5/2021.PEDI) was obtained from the guardians of all participants.
The participants were divided into 2 groups: Group I: included 25 patients with kidney tubular dysfunction. Group II: included 27 patients without kidney tubular dysfunction. Sociodemographic information, cancer type and treatment details, and specific antibacterial and antifungal drugs administered were abstracted from the medical records of each participant who underwent a complete physical examination.
Methods
Laboratory investigations as complete blood count, serum urea, creatinine, calcium, potassium, sodium, and Cystatin C by ELISA were done. Urinary protein/creatinine ratio, calcium/creatinine ratio and urine uric acid were done through a spot test, and NGAL by ELISA.
Serum urea, creatinine, calcium, potassium, sodium were measured on the Beckman Coulter AU680 analyzer (Indianapolis, IN). Cystatin C was measured using the RayBio® Human Cystatin C ELISA Kit supplied by RayBiotech* (Catalog #: ELH-Cystatin C, Raybiotech, Inc., Norcross, Georgia) according to manufacturer’s instructions. Detection Range: 0.3–20 ng/ml, standard curve points: 20, 10, 5, 2.5, 1.25, 0.625, 0.313 and 0 ng/ml, Intra-Assay CV%: <10% and Inter-Assay CV%: <12%. Urine NGAL was analyzed by an NGAL-ELISA kit (Kit 201-12-1720, Sunred Biological Technology Co., Ltd, Shanghai, China). Urinary calcium, uric acid and urinary calcium/creatinine ratio were measured using the Integra 800 device (Pisa, Italy). The estimated glomerular filtration rate (eGFR) was calculated using the modified Schwartz formula for children.7 A normal value of eGFR was ≥90 mL/min/1.73 m2 and the decreased value of eGFR was <90 mL/min/1.73 m2. Participants were defined to have tubular dysfunction if they had abnormal levels of UProtein/Cr, UCa/Cr, UACR, and UrNGAL.
Statistical analysis
The primary outcome was the prevalence of tubular and glomerular dysfunction. The secondary outcome was the comparison between patients with and without tubular dysfunction by the tubular markers.
Data were analyzed using IBM SPSS statistics version 20 (SPSS Inc., Chicago, IL). Chi-square test was used to examine the relationship between qualitative variables. Fisher’s exact test was used when the expected cell count of more than 25% of cases was less than 5. For quantitative data, comparison between two groups was done using either student t-test or Mann–Whitney test (non-parametric t-test) as appropriate. Pearson’s correlation coefficient or Spearman-rho method (as appropriate) was used to test correlation between numerical variables. Receiver Operator Characteristic Curve (ROC) is a graphic representation of the relationship between sensitivity and specificity at different cut-off points for UrNGAL and UrNGAL/Cr. A p value <0.05 was considered significant.
Results
Demographic and clinical data of studied survivors
Fifty-two cases were recruited from the oncology unit. They were 25 girls and 27 boys, their ages ranged from 4 to 18 years old. 35 cases (67.3%) were diagnosed acute lymphoblastic leukemia, 9 cases (17.3%) were non-Hodgkin lymphoma, 2 cases (3.8%) were Hodgkin lymphoma, 3 cases (5.8%) were Neuroblastoma and 3 cases (5.8%) were Wilms tumor. Duration of chemotherapy ranged from 4 to 36 months according to type of cancer. Age at diagnosis of cancer ranged from 3.5 to 15 years old, age at follow up ranged from 4 to 18 years old. Elapsed time from diagnosis to follow-up ranged from 10 to 60 months. Duration after completing chemotherapy ranged from 4 to 24 months. 25 cases (48.1%) had tubular dysfunction and 27 cases (51.9%) had normal tubular function. There was a significant difference in the frequency of treatment with cisplatin and high-dose methotrexate chemotherapy between those with and without tubular dysfunction (Table 1).
Mild anemia was recorded in some cases. Serum electrolytes (Na, K, and Ca), serum urea and creatinine were normal in all patients. eGFR was decreased in 12 cases, and serum Cystatin C was increased in the same 12 cases who had decreased eGFR. The urine calcium/creatinine ratio (UCa/Cr), urine protein/creatinine ratio (Uprotein/Cr) and urine albumin/creatinine ratio were increased in 25 cases (48.1%). In addition, elevated levels of UrNGAL in 30 cases (57.7%) including 5 cases without tubular dysfunction who had a mild increase in UrNGAL and elevated levels of UrNGAL/Cr in all cases (100%) as shown in (Table 2).
Comparison between cases with tubular dysfunction and those without tubular dysfunction
There was a significant difference in Uprotein/Cr (P < 0.001), UCa/Cr (P < 0.001), UrNGAL (P < 0.001) and UrNGAL/Cr (P < 0.001) in patients with tubular dysfunction compared to those without tubular dysfunction (Table 3).
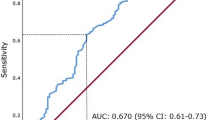
There was a significant positive correlation between UrNGAL, and UrNGAL/Cr regarding serum urea (r = 0.307, P = 0.001, and r = 0.504, P < 0.001 respectively). Also, there was a significant positive correlation between serum cystatin C, UrNGAL, and UrNGAL/Cr regarding serum creatinine (r = 0.521, P < 0.001, r = 0.423, P = 0.003 and r = 0.494, P < 0.001 respectively). There was a significant positive correlation between UrNGAL, UrNGAL/Cr regarding Uprotein/Cr (r = 0.431, P = 0.001, and r = 0.408, P = 0.003 respectively). There was a significant positive correlation between UrNGAL and UACR (r = 0.472, P < 0.001). On the other hand, there was a significant negative correlation between serum Cystatin C regarding eGFR (r = −0.518, P < 0.001) as illustrated in (Table 4). Figure 1 and Table 5 showed the comparison of the diagnostic values between UrNGAL and UrNGAL/Cr for detection of kidney tubular dysfunction among studied cases using the ROC curve. The AUC of distinguishing patients with kidney tubular dysfunction from those without tubular dysfunction by UrNGAL was 0.807 at the cutoff point of 1964.7 ng/g/creatinine where the sensitivity was 84% with the specificity being 70.4%, the accuracy was 76.9%, the PPV was 72.4% and the NPV was 82.6%. The AUC of distinguishing patients with kidney tubular dysfunction from those without tubular dysfunction by UrNGAL/Cr was 0.747 at the cutoff value of 2182 at which level the sensitivity was 84% at a specificity of 40.7%, the accuracy was 61.5%, the PPV was 56.8% and the NPV was 73.3%.
Discussion
The reported prevalence of kidney dysfunction varied from 0 to 84%. This wide range may be related to variations in the definition of abnormal kidney function and cohort factors, including the type of malignancy, chemotherapeutic agents, radiation therapy, and supportive drug use.8,9,10
The present study revealed tubular dysfunction among childhood cancer survivors. We evaluated patients by eGFR, proteinuria, albuminuria and urinary markers for tubular dysfunction. We found low eGFR in 12 cases (23.1%), proteinuria, microalbuminuria and elevated UCa/Cr, UrNGAL and UrNGAL/Cr as well as elevated serum Cystatin C. In addition, there was a significant difference between patients with tubular dysfunction and those without tubular dysfunction regarding cisplatin and methotrexate chemotherapy. Erdem et al.11 showed that low GFR was detected in 32% of all survivors mainly who received nephrotoxic drugs including aminoglycosides, vancomycin or amphotericin B. Certain studies revealed that cisplatin, high-dose methotrexate, and nephrotoxic drugs taken during febrile neutropenia were associated with low eGFR.12,13,14
A Cumulative dose of high-dose MTX, in the range of 1000–33,000 mg/m2 with a combination of calcium leucovorin, is associated with acute kidney injury (AKI) in 0–12.4% with an overall incidence of 1.8%.15
The current study revealed 36.5% of cases had proteinuria and 28.8% of cases had microalbuminuria. Erdem et al.11 showed that microalbuminuria was found in 10.1% of all survivors. Also; Oberlin et al.16 illustrated that proteinuria in 24-h urine collection was detected in 11.3% of survivors and Kninjenburg et al.17 detected albuminuria in 14.5% and decrease eGFR in 62 survivors (4.2%) of all 1442 survivors who were treated with chemotherapeutic.
The current study showed elevated levels of UrNGAL, UrNGAL/Cr and serum Cystatin. Mehdiabadi et al.18 reported an abnormal increase in UrNGAL in 8.9% of patients with acute lymphoblastic leukemia with a mean level of 63 ± 113 ng/mL. In addition, eGFR was less than 60 mL/min/1.73 m2 in 13.3% of these patients indicating kidney dysfunction. Sterling et al.19 showed increased UrNGAL and IL-18 after cisplatin indicating AKI. Latoch et al.20 reported elevated UrNGAL/Cr ratio in cancer survivors receiving cisplatin and ifosfamide, cyclophosphamide and methotrexate. Also, they reported a significantly increased urinary NGAL, NGAL/Cr ratio and KIM-1/Cr ratio in solid tumor survivors indicating kidney tubular dysfunction many years after chemotherapy. Li et al.21 estimated the utility of serum NGAL in early detection of AKI induced by a high-dose of methotrexate.
Barnfield et al.22 reported a significant increase in serum Cystatin C and illustrated the diagnostic value of serum Cystatin C-assessed GFR that was significantly superior to that of serum creatinine-assessed GFR in the detection of kidney dysfunction in cancer children. Lankisch et al.23 revealed that serum Cystatin C was an alternative to serum creatinine for monitoring kidney functions and GFR in pediatric cancer patients with solid tumors and hematological malignancies as it is less dependent on influential factors like weight, height, and muscle mass. Serum Cystatin C had an increased diagnostic accuracy for decreased GFR when compared to serum creatinine.23
Limitation of the study
Small sample size and a small number of cases receiving ifosfamide and cisplatin.
Conclusion
There was a significant tubular dysfunction among childhood cancer survivors receiving chemotherapy as cisplatin and high-dose methotrexate. So, early detection of subclinical kidney dysfunction is very important for early intervention to prevent long-term complications such as chronic kidney disease.
References
Geenen, M. M. et al. Medical assessment of adverse health outcomes in long-term survivors of childhood cancer. JAMA-J. AM MED ASSOC 297, 2705–2715 (2007).
Gatta, G. et al. Survival of European children and young, adults with cancer diagnosed 1995–2002. Eur. J. Cancer 45, 992–1005 (2009). https://doi.org/10.1016/j.ejca.2008.11.042.
Ruggiero, A., Ferrara, P., Attinà, G., Rizzo, D. & Riccardi, R. Renal toxicity and chemotherapy in children with cancer. Br. J. Clin. Pharmacol. 83, 2605–2614 (2017). https://doi.org/10.1111/bcp.13388.
Lankelma, J. & van der Klein, E. The role of 7-hydroxymethotrexate during methotrexate anticancer chemotherapy. Cancer Lett. 9, 133–142 (1980). https://doi.org/10.1016/0304-3835(80)90117-2.
Tanaka, A., Suemaru, K. & Araki, H. A new approach for evaluating renal function and its practical application. J. Pharmacol. Sci. 105, 1–5 (2007). https://doi.org/10.1254/jphs.cp0070058.
Mishra, J. et al. Neutrophil gelatinase-associated lipocalin: a novel early urinary biomarker for cisplatin nephrotoxicity. Am. J. Nephrol. 24, 307–315 (2004). https://doi.org/10.1159/000078452.
Schwartz, G. J. et al. New equations to estimate GFR in children with CKD. J. Am. Soc. Nephrol. 20, 629–637 (2009). https://doi.org/10.1681/ASN.2008030287.
Knijnenburg, S. L. et al. Early and late renal adverse effects after potentially nephrotoxic treatment for childhood cancer. Cochrane Database Syst. Rev. CD008944. (2013). https://doi.org/10.1002/14651858.CD008944.pub2
Blufpand, H. N., Hes, N., Bökenkamp, A., van de Wetering, M. D. & Kaspers, G. J. Diversity in renal function monitoring and dose modifications during treatment for childhood cancer: a call for standardization. Pediatr. Blood Cancer 61, 337–344 (2014). https://doi.org/10.1002/pbc.24572.
National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am. J. Kidney Dis. 39, S1–266 (2002).
Erdem, A. Y., Emir, S., Çakar, N., Demir, H. A. & Özyörük, D. Examination of nephrotoxicity in survivors of childhood cancer and comparison of methods for estimating glomerular filtration rate. İzmir Dr. Behçet. Uz Çocuk Hast. Derg. 11, 139–146 (2021). https://doi.org/10.5222/buchd.2021.90377.
Dekkers, I. A. et al. Long-term nephrotoxicity in adult survivors of childhood cancer. Clin. J. Am. Soc. Nephrol. 8, 922–929 (2013). https://doi.org/10.2215/CJN.09980912.
Kooijmans, E. C. et al. Early and late adverse renal effects after potentially nephrotoxic treatment for childhood cancer. Cochrane Database Syst. Rev. 3, CD008944 (2019). https://doi.org/10.1002/14651858.CD008944.pub3.
Barnett, L. M. A. & Cummings, B. S. Nephrotoxicity and renal pathophysiology: a contemporary perspective. Toxicol. Sci. 164, 379–390 (2018). https://doi.org/10.1093/toxsci/kfy159.
Widemann, B. C. et al. High-dose methotrexate-induced nephrotoxicity in patients with osteosarcoma. Cancer 100, 2222–2232 (2004). https://doi.org/10.1002/cncr.20255.
Oberlin, O. et al. Long-term evaluation of ifosfamide-related nephrotoxicity in children. J. Clin. Oncol. 27, 5350–5355 (2009). https://doi.org/10.1200/JCO.2008.17.5257.
Knijnenburg, S. L. et al. Renal dysfunction and elevated blood pressure in long-term childhood cancer survivors. Clin. J. Am. Soc. Nephrol. 7, 1416–1427 (2012). https://doi.org/10.2215/CJN.09620911.
Mehdiabadi, G. B. Otukesh, H., Shamsabadi, R. H., Arjomand M. E. Long-term follow-up of renal disorders in children with ALL by evaluating urine NGAL. J. Ped. Nephrol. 7, (2019). https://doi.org/10.22037/jpn.v7i2.24506.
Sterling, M. et al. Urine biomarkers of acute kidney injury in noncritical ill, hospitalized children treated with chemotherapy. Pediatr. Blood Cancer. 64, (2017). https://doi.org/10.1002/pbc.26538.
Latoch, E. et al. Urine NGAL and KIM-1—tubular injury biomarkers in long-term survivors of childhood solid tumors: A Cross-Sectional Study. J. Clin. Med. 10, 399 (2021). https://doi.org/10.3390/jcm10030399.
Li, H., Xu, Q., Wang, Y., Chen, K. & Li, J. Serum neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for predicting high dose methotrexate associated acute kidney injury in children with acute lymphoblastic leukemia. Cancer Chemother., Pharm. 85, 95–103 (2020). https://doi.org/10.1007/s00280-019-03980-6.
Barnfield, M. C. et al. Cystatin C in assessment of glomerular filtration rate in children and young adults suffering from cancer. Nucl. Med. Commun. 34, 609–614 (2013). https://doi.org/10.1097/MNM.0b013e328360d929.
Lankisch, P. et al. Serum cystatin C is a suitable marker for routine monitoring of renal function in pediatric cancer patients, especially of very young age. Pediatr. Blood Cancer 46, 767–772 (2006). https://doi.org/10.1002/pbc.20581.
Acknowledgements
The authors thank the parents of children for participation in the study.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
A.M., H.A., S.E., F.Z., H.A., M.E.: took part in database design, data collection, and writing, reporting of search and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The Institutional Review Board (IRB) of the Menoufia Faculty of Medicine approved the study (ID: 24/ 5/2021.PEDI). Research work was performed in accordance with the Declaration of Helsinki.
Informed consent
Informed consent was obtained from all individual participants included in the study or their legally authorized representatives ROC curve of UrNGAL and, UrNGAL/Cr for detection of kidney tubular dysfunction.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mahmoud, A.A.S., Elsalam, H.B.A., El-Deeb, S.M. et al. Evaluation of kidney dysfunction in childhood cancer survivors. Pediatr Res 92, 1689–1694 (2022). https://doi.org/10.1038/s41390-022-02015-w
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-022-02015-w