Abstract
Background
Reduced cerebral regional oxygen saturation (crSO2) variability in neonates, as measured by near-infrared spectroscopy, following cardiac surgery with deep hypothermic circulatory arrest (DHCA) is associated with poor neurodevelopmental outcomes. We sought to evaluate the variability of crSO2 in a cohort of neonates following cardiac surgery with brief or no exposure to DHCA.
Methods
Variability of averaged 1-min crSO2 values was calculated for the first 48 h following cardiac surgery in consecutive neonates over a 30-month period. Neonates requiring aortic arch repair underwent antegrade cerebral perfusion with either brief or no exposure to DHCA.
Results
There were 115 neonates included in the study. Reduced crSO2 variability was observed in neonates with aortic arch obstruction (p = 0.02) and non-survivors (p = 0.02). Post hoc analysis demonstrated that the reduction in crSO2 variability was not as marked as in previously studied neonates with aortic arch obstruction who received DHCA alone (p < 0.001).
Conclusions
Neonates with aortic arch obstruction have reduced crSO2 variability following cardiac surgery. The reduction in crSO2 variability observed in aortic arch obstruction is likely influenced by a number of factors, including perioperative perfusion technique. The impact of interventions on crSO2 variability and resultant influence on neurodevelopmental outcomes requires further study.
Impact
-
Neonates with aortic arch obstruction have reduced crSO2 variability following cardiac surgery, which has been associated with poor neurodevelopmental outcomes, and is likely influenced by a number of factors, including perioperative perfusion technique.
-
The contribution of perioperative perfusion technique to crSO2 variability following neonatal cardiac surgery is significant.
-
Monitoring of crSO2 variability may provide insights into the adequacy of cerebral perfusion in neonates following cardiac surgery.
Similar content being viewed by others
Introduction
Near-infrared spectroscopy (NIRS) is routinely used in the noninvasive monitoring of cerebral regional oxygen saturation (crSO2) in neonates following surgery for congenital heart disease.1,2 Monitoring of crSO2 can provide clinicians with insights into cerebral oxygen delivery and consumption in neonates following cardiac surgery.2,3,4,5,6,7,8,9
Recent studies have demonstrated associations between crSO2 values obtained following cardiac surgery in neonates and postoperative outcomes.10,11,12
Measures of postoperative crSO2 in published reports have typically involved the analyses of average crSO2 values and indices of crSO2 desaturation (e.g., period of time crSO2 < 50% during first 24 h following surgery).10,11,12,13,14 More recently, studies have incorporated measures of crSO2 variability into analyses similar to those employed in analyses of heart rate variability.12,13 In the neonatal literature, crSO2 variability is reduced in premature neonates with hypoxic–ischemic encephalopathy and those with exposure to chorioamnionitis.15,16
Intracardiac repair is made possible by cardiopulmonary bypass (CPB), which takes over circulation and pulmonary gas exchange. The deoxygenated blood from the superior and inferior vena cavae is drained into a reservoir via a cannula and then passed through an oxygenator for gas exchange. The oxygenated blood is pumped back into the ascending aorta via another cannula. However, certain situations particularly where aortic arch reconstruction is performed mandate complete cessation of circulation.17 Historically, these situations have been overcome with deep hypothermic circulatory arrest (DHCA) whereby the patient is cooled (15–20 °C) and CPB flow is stopped, allowing for cannula removal and providing a bloodless and motionless surgical field.17 Antegrade cerebral perfusion is a newer technique that directs blood flow to only the brain during these complex stages of surgery, minimizing DHCA time or allowing for its avoidance altogether.17,18
Our group previously found that reduced crSO2 variability in neonates following cardiac surgery with CPB and DHCA was associated with poor neurodevelopmental outcomes at 21 months of age.12 In this study, we sought to evaluate the variability of crSO2 in a cohort of neonates following cardiac surgery with brief or no exposure to DHCA.
Methods
The Institutional Review Board at the University of Virginia School of Medicine approved this study. We included consecutive neonates (<30 days of age) that underwent cardiac surgery with CPB over a 30-month period from October 2017 to March 2020 at the University of Virginia Children’s Hospital. All patients underwent bi-caval venous cannulation and those patients requiring neonatal arch repair underwent antegrade cerebral perfusion (ACP) with either brief or no exposure to DHCA. All patients were monitored with continuous electroencephalography for the first 48 h following surgery.
Values of crSO2 were continuously captured using the INVOS™ cerebral oximeter (Medtronic, Minneapolis, MN) over the first 48 h following surgery and averaged over 1-min intervals. Variability of crSO2 was calculated using the root mean of successive squared differences (RMSSD) {1} of averaged 1-min crSO2 values over the first 48 h following surgery:
where xi is the crSO2 at time i.
In more practical terms, the RMSSD measures the amount of change in crSO2 from minute to minute over specified period of time. We imputed missing crSO2 values by predictive mean matching. To characterize changes in crSO2 variability over time, we also calculated a 60-min moving variability measure for each minute of monitoring using the RMSSD from the previous 60 min.12
Patient and clinical characteristics, including age at the time of surgery, CPB and cross-clamp times, cardiac diagnosis, surgical procedure, and postoperative ventilator and intensive care unit days, were collected. Patients were assigned to one of four previously described diagnostic classes: class 1—two ventricle repair without aortic arch obstruction, class 2—two ventricle repair with aortic arch obstruction, class 3—single ventricle repair without aortic arch obstruction, or class 4—single ventricle repair with aortic arch obstruction.19
In a post hoc analysis, we compared the crSO2 variability of neonates with aortic arch obstruction (classes 2 and 4) in our cohort with those from our previously published study who all underwent DHCA.12 As our previous study was limited to monitoring for the first 24 h following surgery, we used the crSO2 variability at 24 h in the present study for the comparison.
Distribution of continuous variables was assessed using the Wilk–Shapiro test for normality. Continuous variables were compared using Student’s t test, Wilcoxon’s rank-sum testing, or linear regression as appropriate. Categorical variables were compared using χ2 test or Fisher’s exact testing as appropriate. Type I error was set at 0.05. All calculations were performed using STATA/IC 12.1 (STATA Corporation, College Station, TX).
Results
There were 115 neonates included in the study with a median of 8 days of age (interquartile range (IQR) 5–10 days) at the time of surgery. The demographic and clinical characteristics of the included patients are listed in Table 1.
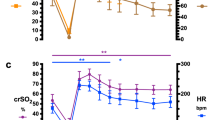
Variability of crSO2 was not associated with patient age, single ventricle repair, durations of CPB or cross-clamp, and postoperative seizures, ventilator, or intensive care unit days. Patients with aortic arch obstruction (p = 0.02) and non-survivors (p = 0.02) demonstrated reduced crSO2 variability during the first 48 h following surgery. The time-series plots of crSO2 values and corresponding crSO2 variability of two representative neonates are shown in Fig. 1.
All patients with aortic arch obstruction underwent ACP with a median duration of 43 min (IQR 29–84 min). Thirty-four (52%) of these patients underwent brief DHCA with a median duration of 7 min (IQR 4–12 min). Neither duration of antegrade cerebral perfusion or DHCA was associated with crSO2 variability. We stratified clinical characteristics and outcomes by aortic arch obstruction in Table 2.
To compare the 67 patients with aortic arch obstruction from this cohort with patients with aortic arch obstruction from our previous DHCA cohort, we calculated the crSO2 variability reflective of the first 24 h following surgery. The median crSO2 variability over the first 24 h was 1.4 (IQR 1.2–1.8), not different from the crSO2 variability for the entire 48-h period of monitoring (p = 0.09). In our previous study, there were 41 patients with aortic arch obstruction with a median duration of DHCA of 46 min (IQR 38–51 min) and crSO2 variability of 1 (IQR 0.8–1.4).12 The patients from our previous DHCA cohort were younger at the time of surgery (4 vs. 6 days, p < 0.001) and more likely to undergo single ventricle repair (p < 0.001). There was no difference in the minimum temperature achieved during cooling between the groups (18 °C in both groups). When comparing the cohorts, patients with aortic arch obstruction who received antegrade cerebral perfusion had 40% greater crSO2 variability at 24 h following surgery (1.4 vs. 1, p < 0.001).
Discussion
We observed reduced crSO2 variability in neonates with aortic arch obstruction in the first 48 h following cardiac surgery with antegrade cerebral perfusion and brief or no exposure to DHCA. The reduction in crSO2 variability was not as marked as in previously studied neonates who received DHCA alone. The reduction in crSO2 variability observed in aortic arch obstruction is likely influenced by a number of factors.
Brain-sparing refers to the adaptation of autoregulatory mechanisms to increase cerebral perfusion in fetuses with congenital heart disease.20 Fetal ultrasound studies have demonstrated that fetuses with hypoplastic left heart syndrome may have inadequate autoregulatory compensatory mechanisms, influencing neurodevelopmental abnormalities observed in this group.20,21,22 Fetuses with aortic arch obstruction have decreased middle cerebral artery pulsatility indices, indicating restricted cerebral blood flow in this group.23 During the transition to extrauterine life there is a decline in pulmonary vascular resistance and increase in system vascular resistance. In the neonate with ductal-dependent cardiac lesions, the maintenance of adequate cerebral blood flow in the setting of decreased pulmonary vascular resistance and an open ductus arteriosus may be difficult.24 There is evidence to suggest a persistence of impaired cerebral autoregulation in neonates with congenital heart disease during the first 24–48 h of life prior to cardiac surgery.24,25 The degree to which fetal and transitional cerebral autoregulation and perfusion impacts crSO2 variability in the early neonatal period requires further study.
The impact of the type of perfusion technique employed during neonatal cardiac surgery on neurodevelopmental outcomes remains controversial.26 The findings of the Boston Circulatory Arrest Trial suggest that a duration of DHCA of 41 min or less does not adversely impact neurodevelopmental outcomes as measured at 8 years of age.27 Two studies have demonstrated no difference in neurodevelopmental outcomes at 1 year of age in infants who underwent aortic arch reconstruction between DHCA alone and DHCA with brief or no antegrade cerebral perfusion.28,29 More recently, in a cohort of neonates receiving antegrade cerebral perfusion undergoing aortic arch reconstruction with specialized intraoperative neuromonitoring, Andropoulos et al.30 demonstrated 12-month cognitive outcomes at reference population norms.
In our previous work, we found an association between decreased crSO2 variability and worse neurodevelopmental outcomes at 21 months of age.12 We observed a strong association between duration of DHCA and degree of crSO2 variability reduction (i.e., longer DHCA duration associated with a greater reduction of crSO2 variability).12 Our observation in the present study of reduced crSO2 variability in neonates with aortic arch obstruction, although with significantly less reduction than in our previous study, supports the notion that multiple factors (e.g., intrauterine, transitional, perioperative) influence our findings.
There are several limitations to our study, including the single-center nature. We are limited in our ability to make conclusions regarding the impact of therapeutic interventions on crSO2 variability, although our institutional practice is largely uniform as it relates to the selection and use of sedative and vasoactive infusions in the postoperative period. Conclusions drawn from our comparison of data from the present study with our previous study require careful consideration given a number of limitations. While both cohorts were limited to patients with aortic arch obstruction, the patients in the previous cohort were younger (4 vs. 6 days) and more likely to undergo single ventricle repair. In addition, our prior study employed a different NIRS monitor (NIRO-200; Hamamatsu Photonics KK, Hamamatsu, Japan).12 Studies have demonstrated differences in the absolute values of regional oxygen saturation indices between different NIRS monitors.5,31 However, the calculation of variability is, by definition, agnostic to the measured index so the comparison of values between devices should not be significantly impacted.
There are some important next steps to consider. First, at present, we are not aware of a commercially available NIRS device that provides readily accessible crSO2 variability data. As such, monitoring of crSO2 variability requires the extraction of crSO2 values from the NIRS device for the extra-device calculation and assessment of variability. The integration of variability measures with NIRS devices could potentially enhance the value of these measures for real-time clinical assessment and decision support. Second, we have recently initiated work on a follow-up project, capturing both preoperative and postoperative values in neonates undergoing cardiac surgery, in an effort to better understand the perioperative influence on crO2 variability. Finally, assessing the relationship between crSO2 variability and neurodevelopmental outcomes in patients with minimal or no exposure to DHCA will be an important next step.
Neonates with aortic arch obstruction have reduced crSO2 variability following cardiac surgery. The reduction in crSO2 variability observed in aortic arch obstruction is likely influenced by a number of factors, including perioperative perfusion technique. The impact of interventions on crSO2 variability and resultant influence on neurodevelopmental outcomes requires further study.
References
Ghanayem, N. S. & Hoffman, G. M. Near infrared spectroscopy as a hemodynamic monitor in critical illness. Pediatr. Crit. Care Med. 17, S201–S206 (2016).
Hoffman, G. M., Ghanayem, N. S. & Tweddell, J. S. Noninvasive assessment of cardiac output. Semin. Thorac. Cardiovasc. Surg. Pediatr. Card. Surg. 8, 12–21 (2005).
Li, J. et al. Assessment of the relationship between cerebral and splanchnic oxygen saturations measured by near-infrared spectroscopy and direct measurements of systemic haemodynamic variables and oxygen transport after the Norwood procedure. Heart 92, 1678–1685 (2006).
McQuillen, P. S. et al. Regional and central venous oxygen saturation monitoring following pediatric cardiac surgery: concordance and association with clinical variables. Pediatr. Crit. Care Med. 8, 154–160 (2007).
Weiss, M. et al. Near-infrared spectroscopic cerebral oxygenation reading in neonates and infants is associated with central venous oxygen saturation. Paediatr. Anaesth. 15, 102–109 (2005).
Chakravarti, S. B. et al. Multisite near-infrared spectroscopy predicts elevated blood lactate level in children after cardiac surgery. J. Cardiothorac. Vasc. Anesth. 23, 663–667 (2009).
Nagdyman, N. et al. Relation of cerebral tissue oxygenation index to central venous oxygen saturation in children. Intens. Care Med. 30, 468–471 (2004).
Nagdyman, N. et al. Comparison of different near-infrared spectroscopic cerebral oxygenation indices with central venous and jugular venous oxygenation saturation in children. Paediatr. Anaesth. 18, 160–166 (2008).
Tortoriello, T. A. et al. A noninvasive estimation of mixed venous oxygen saturation using near-infrared spectroscopy by cerebral oximetry in pediatric cardiac surgery patients. Paediatr. Anaesth. 15, 495–503 (2005).
Hoffman, G. M. et al. Postoperative cerebral and somatic near-infrared spectroscopy saturations and outcome in hypoplastic left heart syndrome. Ann. Thorac. Surg. 103, 1527–1535 (2017).
Aly, S. A. et al. Cerebral tissue oxygenation index and lactate at 24 h postoperative predict survival and neurodevelopmental outcome after neonatal cardiac surgery. Congenit. Heart Dis. 12, 188–195 (2017).
Spaeder, M. C. et al. Perioperative near-infrared spectroscopy monitoring in neonates with congenital heart disease: relationship of cerebral tissue oxygenation index variability with neurodevelopmental outcome. Pediatr. Crit. Care Med. 18, 213–218 (2017).
Flechet, M. et al. Near-infrared cerebral oximetry to predict outcome after pediatric cardiac surgery: a prospective observational study. Pediatr. Crit. Care Med. 19, 433–441 (2018).
Simons, J. et al. Predictive value of near-infrared spectroscopy on neurodevelopmental outcome after surgery for congenital heart disease in infancy. J. Thorac. Cardiovasc. Surg. 143, 118–125 (2012).
Arriaga-Redondo, M. Lack of variability in cerebral oximetry tendency in infants with severe hypoxic-ischemic encephalopathy under hypothermia. Ther. Hypothermia Temp. Manag. 9, 243–250 (2019).
Yanowitz, T. D. et al. Variability in cerebral oxygen delivery is reduced in premature neonates exposed to chorioamnionitis. Pediatr. Res. 59, 299–304 (2006).
Ginther, R. M. & Forbess, J. M. in Fuhrman & Zimmerman’s Pediatric Critical Care (eds Fuhrman, B. P. & Zimmerman, J. J.) 430–446 (Elsevier, Philadelphia, 2017).
Fraser, C. D. & Andropoulos, D. B. Principles of antegrade cerebral perfusion during arch reconstruction in newborns/infants. Semin. Thorac. Cardiovasc. Surg. Pediatr. Card. Surg. 11, 61–68 (2008).
Clancy, R. R. et al. Preoperative risk-of-death prediction model in heart surgery with deep hypothermic circulatory arrest in the neonate. J. Thorac. Cardiovasc. Surg. 119, 347–357 (2000).
Donofrio, M. T. et al. Autoregulation of cerebral blood flow in fetuses with congenital heart disease: the brain sparing effect. Pediatr. Cardiol. 24, 436–443 (2003).
Berg, C. et al. Doppler indices of the middle cerebral artery in fetuses with cardiac defects theoretically associated with impaired cerebral oxygen delivery in utero: is there a brain-sparing effect? Ultrasound Obstet. Gynecol. 34, 666–672 (2009).
Arduini, M. et al. Cerebral blood flow autoregulation and congenital heart disease: possible causes of abnormal prenatal neurologic development. J. Matern. Fetal Neonatal Med. 24, 1208–1211 (2011).
Yamamoto, Y. et al. Severe left heart obstruction with retrograde arch flow influences fetal cerebral and placental blood flow. Ultrasound Obstet. Gynecol. 42, 294–299 (2013).
Peyvandi, S. & Donofrio, M. T. Circulatory changes and cerebral blood flow and oxygenation during transition in newborns with congenital heart disease. Semin. Pediatr. Neurol. 28, 38–47 (2018).
Votava-Smith, J. K. et al. Impaired cerebral autoregulation in preoperative newborn infants with congenital heart disease. J. Thorac. Cardiovasc. Surg. 154, 1038–1044 (2017).
Ohye, R. G. et al. The quest to optimize neurodevelopmental outcomes in neonatal arch reconstruction: the perfusion techniques we use and why we believe in them. J. Thorac. Cardiovasc. Surg. 137, 803–806 (2009).
Wypij, D. et al. The effect of duration of deep hypothermic circulatory arrest in infant heart surgery on late neurodevelopment: the Boston Circulatory Arrest Trial. J. Thorac. Cardiovasc. Surg. 126, 1397–1403 (2003).
Visconti, K. J. et al. Regional low-flow perfusion versus circulatory arrest in neonates: one-year neurodevelopmental outcome. Ann. Thorac. Surg. 82, 2207–2211 (2006).
Goldberg, C. S. et al. A randomized clinical trial of regional cerebral perfusion versus deep hypothermic circulatory arrest: outcomes for infants with functional single ventricle. J. Thorac. Cardiovasc. Surg. 133, 880–887 (2007).
Andropoulos, D. B. et al. Neurodevelopmental outcomes after regional cerebral perfusion with neuromonitoring for neonatal aortic arch reconstruction. Ann. Thorac. Surg. 95, 648–654 (2013).
Gagnon, R. E. et al. Comparison of two spatially resolved NIRS oxygenation indices. J. Clin. Monit. Comput. 17, 6 (2002).
Author information
Authors and Affiliations
Contributions
M.C.S. made substantial contributions to the conception and design, acquisition, analysis and interpretation of data; drafted the article for important intellectual content; and approved the final version to be published. V.J.S. made substantial contributions to the acquisition of data; and approved the final version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Statement of ethics
The authors received a waiver of consent by the Institutional Review Board at the University of Virginia School of Medicine for this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Spaeder, M.C., Surma, V.J. Cerebral regional oxygen saturation variability in neonates following cardiac surgery. Pediatr Res 90, 815–818 (2021). https://doi.org/10.1038/s41390-020-01171-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-020-01171-1
This article is cited by
-
The prediction of estimated cerebral perfusion pressure with trans-systolic time in preterm and term infants
European Journal of Pediatrics (2024)
-
Changes in Cerebral Regional Oxygen Saturation Variability in Neonates Undergoing Cardiac Surgery: A Prospective Cohort Study
Pediatric Cardiology (2023)
-
Implementation of a Regional Oxygen Saturation Thought Algorithm and Association with Clinical Outcomes in Pediatric Patients Following Cardiac Surgery
Pediatric Cardiology (2023)
-
Regional Cerebral Oxygen Saturation and Estimated Oxygen Extraction Ratio as Predictive Markers of Major Adverse Events in Infants with Congenital Heart Disease
Pediatric Cardiology (2023)