Abstract
Background
Twins experience altered growth compared to singletons. The primary aim of this study was to compare growth and body composition (BC) of twin and singleton preterm infants from birth to 3 months according to gestational age (GA). Secondary aims were to evaluate the effect of chorionicity and mode of feeding on twins’ BC.
Methods
Anthropometric measurements and BC were performed at term and 3 months in preterm infants (GA < 37 weeks). Infants were categorized as: extremely, very, moderate and late preterm infants. Chorionicity was assigned as monochorionic, dichorionic or multichorionic. Mode of feeding was recorded as any human milk feeding vs formula feeding.
Results
Five hundred and seventy-six preterm infants were included (223 twins). Late-preterm twins were lighter and shorter at each study point; fat-free mass (FFM) was lower in these infants at each study point, compared to singletons. No differences were found between twins and singleton on the other category. Multichorionic infants had an FFM deficit compared to monochorionic and dichorionic at term, whereas no differences were found at 3 months. FFM at term was negatively associated with being twin and formula-fed.
Conclusions
Twins and singletons born before 34 weeks’ GA showed similar anthropometry and BC. Conversely, twin late-preterm infants showed different growth and BC compared to singletons.
Similar content being viewed by others
Introduction
The global frequency of multiple gestations has increased, representing approximately 3–4% of all pregnancies, probably due to late motherhood and the increased use of assisted reproductive techniques.1,2 Twin infants can experience an altered foetal and postnatal growth compared to singletons.3 However, it is a common practice to assess twin growth with singleton foetal and postnatal growth charts, which could lead to a higher percentage of twins being classified as growth-restricted.4 Despite the existence of foetal growth charts specifically for twins, stratified by chorionicity,5,6 and of a twin-specific growth reference from birth to 2.5 years for full-term infants,7 little is known about twin preterm infant postnatal growth and quality of growth. A study conducted in Australia showed that twin foetuses grew more slowly than singletons from 26 weeks of gestation,8 whereas Belgian twins showed a reduction in weight gain from 32 weeks compared to singletons.9 In addition, a recent multicentre study showed that monochorionic twins showed a markedly different growth than dichorionic foetuses compared to singletons, especially during the third trimester of gestation.6
There is increasing knowledge on the relationship between early quality of growth pattern and later health outcomes.10,11 Specifically, in a study conducted on 110 preterm infants, gestational age (GA) at birth showed a significant negative correlation with fat mass (FM) at term-corrected age (R2 = 0.47).12 Despite this knowledge, few data are available on twins’ body composition.13,14 Considering the paucity of data and that preterm infants show an altered FM and fat-free mass deposition (FFM)15,16 and the clinical implications of the quality of early growth, we performed a study in order to assess growth and body composition of twin preterm infants from birth to 3 months corrected age according to GA. The secondary aim of the study was to evaluate whether the chorionic status can interfere with body composition.
Methods
Study design
This was a 9-year single-centre retrospective study including preterm infants born at Fondazione IRRCS Ca’ Granda Ospedale Maggiore Policlinico and admitted to levels of care I, II and III from January 2008 to December 2016.
Subjects
After institutional review board approval and parent’s informed consent collection, we reviewed the baseline characteristics, anthropometric measurements and body composition data of preterm infants enrolled in previous studies on body composition. Inclusion criteria were GA <37 weeks, Caucasian parentage and clinical stability at term-corrected age. Exclusion criteria for all infants enrolled were the presence of major congenital anomalies, chromosomal abnormalities or cardiac, brain, renal, endocrine or surgical diseases known to affect postnatal growth and severe intrauterine growth restriction (birth weight <3° percentile according to Fenton growth charts17).
Foetal and neonatal characteristics (GA, being singleton or twin, chorionicity and sex) were recorded from the patients’ computerized medical records. Infants enrolled were categorized according to GA at birth as extremely preterm (GA < 28 weeks), very preterm (GA from 28 to 31 6/7 weeks), moderate preterm (GA from 32 to 33 6/7 weeks) and late preterm (GA from 34 to 36 6/7 weeks).18 GA was based on the last menstrual period and first-trimester ultrasonogram. In case of a discrepancy between the last menstrual period and first-trimester ultrasonogram, the early ultrasonogram estimation was considered. Corrected age was calculated using the chronologic age and by adjusting for GA, that is, for the number of additional weeks from term (40 weeks).19
Chorionicity was assigned as monochorionic, dichorionic or multichorionic by standard ultrasonographic criteria (placental number, identification of lambda or T sign, intertwin membrane thickness and determination of foetal gender) at the first ultrasound evaluation.20 Chorionicity was confirmed by postpartum histological assessment of the placenta.
Anthropometric measurements
Body weight, body length and head circumference of infants enrolled were assessed at birth, at term-corrected age and at 3 months’ corrected age according to standard procedures.21 The naked infant’s body weight was measured on an electronic scale accurate to the nearest 0.1 g (PEA POD Infant Body Composition System; COSMED, Italy), and body length was measured to the nearest 1 mm using an infant length board. Head circumference was measured to the nearest 1 mm using a non-stretch measuring tape.
Body composition assessment
Body composition was assessed at term-corrected age and at 3 months’ corrected age. Body composition was assessed using an air-displacement plethysmography system (PEA POD Infant Body Composition System; COSMED, Italy).22,23 The PEA POD assesses FM and FFM by body mass and body volume measurements and by the application of whole-body densitometric principles. Body density was computed from the study subject’s measured mass and volume and then converted to indicate the total absolute (g) and percentage (%) of FM and FFM using sex-specific equations developed by Fomon et al.24 The inter-observer coefficient of variation for the FM percentage estimates was 0.3%. The FM and FFM indexes were calculated at each study point by dividing each infant’s FM of FFM (kg) by the square of his/her length (m).
Nutritional regimen for infants with a birth weight <1500 g
Infants with a birth weight <1500 g qualified for parenteral nutrition. Parenteral nutrition was started on the first day of life. The provided volume increased from 70 to 100 ml/kg on the first day up to 150–180 ml/kg on the seventh day of life, with an energy/protein ratio from 20 kcal/g on the first day up to 26.4 kcal/g on the seventh day of life. Enteral feeding was started within 24 h of postnatal life using breast milk or formula when breast milk was absent. Infants born before 34 weeks’ GA received preterm formula during their hospital stay (energy: 83 kcal/100 ml; carbohydrates: 8.4 g/100 ml; proteins: 2.7 g/100 ml; fat: 4.1 g/100 ml). Infants born after 34 weeks’ GA received standard formula. When the infants tolerated an enteral intake ≥100 ml/kg, individually tailored fortification of breast milk was started.
Nutritional regimen for infants with a birth weight >1500 g
Mothers of full-term infants were encouraged to breastfeed their infants. When human milk was unavailable or insufficient, formula feeding was started. Infants born before 34 weeks’ GA received preterm formula during their hospital stay (energy: 83 kcal/100 ml; carbohydrates: 8.4 g/100 ml; proteins: 2.7 g/100 ml; fat: 4.1 g/100 ml). Infants born after 34 weeks’ GA received standard formula. Infants presenting with any clinical condition that could delay the beginning of enteral nutrition or that could interfere with the ability to feed exclusively by mouth received either parenteral nutrition/intravenous fluids or tube feeding. Provided that infants were in stable clinical condition, mothers were encouraged to breastfeed their infants.
The feeding status for all infants included in the study was categorized as (1) any human milk, including exclusively, predominantly and partially human milk25 and (2) exclusively formula. The feeding status during the hospital stay was collected from the patients’ computerized medical records. The feeding status at term-corrected age (from discharge to term-corrected age) and at 3 months’ corrected age (from term to 3 months’ corrected age) was collected from parents’ daily records or was reported by the parents. The internal nutritional procedure was the same for singletons and twins.
Statistical analysis
Continuous variables are reported as the mean and standard deviation (SD). Categorical variables are reported as absolute numbers or percentages. Anthropometric measurements and body composition of singleton preterm infants and twin preterm infants were compared, according to the categorization of GA, using χ2 test for discrete variables or analysis of variance for continuous variables. In those categories where there was a statistically significant difference between the two groups, we performed a multiple regression analysis to evaluate the association between body composition, being twins and being fed any human milk. Kruskal–Wallis test was conducted to compare anthropometric measurements and body composition according to chorionicity. Statistical significance was set at alpha <0.05. Statistical analyses were conducted using the SPSS (Statistical Package for the Social Sciences) version 12 software (SPSS Inc., Chicago, IL, USA).
Results
In the present study, 576 preterm infants were included. Infants’ basal characteristics at birth according to categorization are detailed in Table 1. With regard to anthropometric measures at birth, twin infants categorized as extremely preterm, very preterm and moderate preterm were similar to their singleton counterparts, whereas late-preterm twins showed a lower weight and length compared to singletons.
Effect of GA
Anthropometric measurements and body composition at term and at 3 months’ corrected age according to categorization are shown in Tables 2 and 3. No difference in growth parameters or body composition were found between singletons and twins categorized as extremely preterm, very preterm and moderate preterm either at term or at 3 months’ corrected age. Late-preterm twins were significantly lighter and shorter than singleton late-preterm infants both at term-corrected age and at 3 months’ corrected age. Concerning body composition, late-preterm twins showed an FFM deficit compared to singletons both at term-corrected age and at 3 months’ corrected age. At term and at 3 months’ corrected age, late-preterm twins showed less FM compared to singletons. No differences were found in FM index or FFM index at term or at 3 months’ corrected age.
Effect of nutrition
With regard to the nutrition of preterm infants born before 34 weeks’ GA until discharge, no differences were found between twins and singletons in terms of mean duration of parenteral nutrition (23.2 ± 11.7 and 23.8 ± 12.5 days, respectively) or age of achieving of full enteral feeding at 150 ml/kg (28.7 ± 16.9 days of life for singletons and at 28.1 ± 16.1 days of life for twins). During hospitalization, any human milk was received by 86.6% of twins and 88.3% of singletons.
Conversely, among all infants born after 34 weeks’ GA, 4.1% received parenteral nutrition, 33.7% required nutritional support with intravenous fluids and 2.5% needed a tube feeding. During hospitalization, any human milk was received by 63.1% of twins and 75.2% of singletons.
Considering that late-preterm twin infants showed different growth and body composition compared to their singleton counterparts, a multiple regression analysis was performed (Table 4). It showed that FFM deposition at term-corrected age was negatively associated with being a twin and with being formula-fed. At 3 months’ corrected age, only being a twin was negatively associated with FFM deposition. FM deposition at term-corrected age and at 3 months’ corrected age was not associated with mode of feeding in the regression analysis, whereas being a twin was negatively associated with FM deposition both at term and at 3 months’ corrected age.
Effect of chorionicity
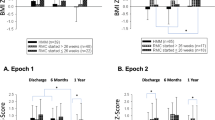
Among all 223 twin infants, 26% were monochorionic, 60.6% were dichorionic and 13.4% were multichorionic infants. Multichorionic infants showed lower GA at birth and were smaller than monochorionic and dichorionic infants (GA: 30.2 ± 1.9 vs 33.5 ± 2.5 and 33.4 ± 2.9 weeks; weight: 1222.8 ± 190 vs 1897.7 ± 575 and 1874.8 ± 632 g; length: 37.7 ± 3.1 vs 42.3 ± 4.6 and 42.1 ± 4.4 cm; head circumference: 27.5 ± 1.9 vs 30.2 ± 2.3 and 30.2 ± 2.9 cm, respectively; p < 0.001). Anthropometric measurements at term and at 3 months’ corrected age were similar among all twins irrespective of chorionicity. With regard to body composition, multichorionic infants had an FFM deficit compared to monochorionic and dichorionic infants at term-corrected age (Fig. 1), whereas no difference was found at 3 months’ corrected age among all twins. No differences were found according to GA categorization.
Discussion
In the present study, a relatively large cohort of preterm infants was investigated to describe growth and body composition of twin preterm infants compared to singletons, taking into account the prematurity category and chorionicity.
Effect of GA
Our results show that moderate, very and extremely preterm twin infants were similar to their singleton counterparts both at birth and during the first months of life, whereas twin late-preterm infants were smaller than their singleton counterparts at birth, and the differences remained significant at 3 months’ corrected age. These results are consistent with Hennequin et al., who demonstrated that Belgian twins showed a reduction in weight gain from 32 weeks compared to singletons.9 Paviotti et al. and Demarini et al., in two different studies conducted on infants born from 35 to 40 weeks’ GA, observed that body composition was similar in twins and singletons at birth and during the first month of life.13,14 Conversely, our study demonstrated that the body composition of twin late-preterm infants showed a significant deficit of FFM and FM compared to singletons at each study point. It has to be taken into account that twin late-preterm infants were shorter than singletons at birth, and the difference was still detected at 3 months of corrected age; this led them to have FFM and FM indexes both at term and at 3 months’ corrected age that were similar to singleton late-preterm infants. Indeed, a higher FM index has been positively associated with the presence of metabolic syndrome in adulthood.26 Therefore, we can argue that twins, despite a different FM and FFM deposition compared to singletons, may not have an increased risk to develop metabolic syndrome compared to singletons. Actually, in a previous study conducted in our centre, we demonstrated that late-preterm infants, who showed a higher FM accretion than full-term infants at term-corrected age, had FM and FFM indexes similar to full-term infants at 3 months’ corrected age, and this was not associated with perturbation of the glucose homoeostasis.27
Effect of nutrition
Our results show that feeding any human milk played an important role in promoting FFM deposition at term-corrected age. Similar results were found in a meta-analysis investigating the effect of breastfeeding in a large cohort of preterm infants.28 Specifically, breastfed infants had a significantly lower FM in comparison to formula-fed infants (mean difference 0.24; 95% CI 0.17, 0.31 kg) at term-corrected age. This result was confirmed by previous results obtained by our group in late-preterm infants.29 In line with these results, we demonstrated that twin infants who received less human milk were more at risk to experience an FFM deficit compared to singletons.
Effect of chorionicity
Chorionicity can influence the growth of twin foetuses.6,30 Postnatally, full-term monochorionic infants experienced a slower pattern of growth compared to dichorionic infants during the first 6 months of life.7 The present study demonstrated that multichorionic infants were born at a lower GA and, consequently, were smaller than monochorionic and dichorionic infants at birth, even if they subsequently achieved anthropometric measurements similar to monochorionic and dichorionic infants at term-corrected age and at 3 months’ corrected age. However, we must consider that the number of multichorionic infants included in this study is relatively small (29 infants), due to the rarity of multifoetal pregnancy (triplets or more).
To our knowledge, there is a paucity of data regarding body composition of twins according to chorionicity. Our results show an FFM deficit at term-corrected age in multichorionic compared to monochorionic and dichorionic infants. Focussing on late-preterm infants, our results suggest that, in this category of infants, being monochorionic or dichorionic did not have a strong influence on postnatal growth.
A limitation of this analysis is the lack of data on zygosity, which may be of interest since dizygotic twins are often taller and have higher body mass index, especially during childhood.31
Conclusions
Below 34 weeks of GA, twins and singletons showed similar birth anthropometry and similar measurements and body composition at term-corrected age and at 3 months of corrected age. In contrast, late-preterm twins and singletons had different weight and length at birth, term and 3 months of corrected age, and significant differences also appeared in the body composition analyses. Despite a relatively short follow-up, this study included a large cohort of infants, and its results confirm that late-preterm infants are a peculiar category of infants and that being a twin represents an additional risk factor for altered growth.
Even if the neonatological management of premature infants is similar for twin and singleton premature infants, additional difficulties in establishing and maintaining breastfeeding may be encountered by twins. Considering that human milk feeding can modulate the FFM deposition, health-care professionals should give special effort to promoting and supporting breastfeeding in these infants.
References
Burgess, J. L. et al. Risk of late-preterm stillbirth and neonatal morbidity for monochorionic and dichorionic twins. Am. J. Obstet. Gynecol. 210, 578 e1–9. (2014).
Hayes, E. J. et al. Very-low-birthweight neonates: do outcomes differ in multiple compared with singleton gestations? Am. J. Perinatol. 24, 373–376 (2007).
Loos, R. J. F. et al. Determinants of birthweight and intrauterine growth in liveborn twins. Paediatr. Perinat. Epidemiol. 19, 15–22 (2005).
Zhang, J. et al. An adjustable fetal weight standard for twins: a statistical modeling study. BMC Med. 13, 159 (2015).
Mendez-Figueroa, H. et al. Growth among twins: use of singleton versus twin-specific growth nomograms. Am. J. Perinatol. 35, 184–191 (2018).
Ghi, T. et al. Development of customized fetal growth charts in twins. Am. J. Obstet. Gynecol. 216, 514.e1–514.e17 (2017).
van Dommelen, P. et al. Growth references for height, weight and body mass index of twins aged 0–2.5 years. Acta Paediatr. 97, 1099–1104 (2008).
Liu, Y. C. & Blair, E. M. Predicted birthweight for singletons and twins. Twin Res. 5, 529–537 (2002).
Hennequin, Y. et al. Twins: interpretation of height-weight curves at birth. Rev. Med. Brux. 20, 81–85 (1999).
Roggero, P. et al. Measuring the body composition of preterm and term neonates: from research to clinical applications. J. Pediatr. Gastroenterol. Nutr. 45, 159–162 (2007).
Wells, J. C., Chomtho, S. & Fewtrell, M. S. Programming of body composition by early growth and nutrition. Proc. Nutr. Soc. 66, 423–434 (2007).
Roggero, P. et al. Is term newborn body composition being achieved postnatally in preterm infants? Early Hum. Dev. 85, 349–352 (2009).
Paviotti, G. et al. Longitudinal growth and body composition of twins versus singletons in the first month of life. Sci. World J. 2013, 108189 (2013).
Demarini, S., Koo, W. W. K. & Hockman, E. M. Bone, lean and fat mass of newborn twins versus singletons. Acta Paediatr. 95, 594–599 (2006).
Johnson, M. J. et al. Preterm birth and body composition at term equivalent age: a systematic review and meta-analysis. Paediatrics 130, e640–e649 (2012).
Giannì, M. L. et al. Body composition in late preterm infants according to percentile at birth. Pediatr. Res. 79, 710–715 (2016).
Fenton, T. R. & Kim, J. H. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pediatr. 13, 59 (2013).
Blencowe, H. et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: a systematic analysis and implications. Lancet 379, 2162–2172 (2012).
Engle, W. A. American Academy of Pediatrics Committee on Fetus and Newborn. Age terminology during the perinatal period. Pediatrics 114, 1362–1364 (2004).
Hehir, M. P. et al. Prenatal prediction of significant intertwin birthweight discordance using standard second and third trimester sonographic parameters. Acta Obstet. Gynecol. Scand. 96, 472–478 (2017).
Agostoni, C. et al. Growth patterns of breast fed and formula fed infants in the first 12 months of life: an Italian study. Arch. Dis. Child. 81, 395–399 (1999).
Ma, G. et al. Validation of a new pediatric air displacement plethysmograph for assessing body composition in infants. Am. J. Clin. Nutr. 79, 653–660 (2004).
Ellis, K. J. et al. Body composition assessment in infancy: air-displacement plethysmography compared with a reference 4-compartment model. Am. J. Clin. Nutr. 85, 90–95 (2007).
Fomon, S. J. et al. Body composition of reference children from birth to age 10 years. Am. J. Clin. Nutr. 35, 1169–1175 (1982).
Section on Breastfeeding. Breastfeeding and the use of human milk. Pediatrics 129, e827–e841 (2012).
Liu, P. et al. The utility of fat mass index vs. body mass index and percentage of body fat in the screening of metabolic syndrome. BMC Public Health 13, 629 (2013).
Liotto, N. et al. Is fat mass accretion of late preterm infants associated with insulin resistance? Neonatology 111, 353–359 (2017).
Huang, P. et al. Effects of breast-feeding compared with formula-feeding on preterm infant body composition: a systematic review and meta-analysis. Br. J. Nutr. 116, 132–141 (2016).
Giannì, M. L. et al. Postnatal catch-up fat after late preterm birth. Pediatr. Res. 72, 637–640 (2012).
Grantz, K. et al. Dichorionic twin trajectories: the NICHD Fetal Growth Studies. Am. J. Obstet. Gynecol. 215, 221 (2016).
Jelenkovic, A. et al. Zygosity differences in height and body mass index of twins from infancy to old age: a study of the CODATwins Project. Twin Res. Hum. Genet. 18, 557–570 (2015).
Author information
Authors and Affiliations
Contributions
N.L. and P.R. conceptualized and designed the study, drafted the manuscript and reviewed and revised the manuscript. F.G., M.L.G. and F.M. critically reviewed the manuscript for important intellectual content. D.M., B.B., C.M., A.O., P.P. and O.A. collected the data, were responsible for database management, analysed the data and contributed to the discussion of the results. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liotto, N., Roggero, P., Giuliani, F. et al. How does gestational age affect growth and body composition of preterm twins?. Pediatr Res 87, 57–61 (2020). https://doi.org/10.1038/s41390-019-0530-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-019-0530-5