Abstract
Background
In adults, distal baseline impedance (BI) is a determinant of esophageal mucosal integrity with values <900 Ω indicating inflammation. Relationships between acid gastroesophageal reflux (GER) and BI in neonates are unclear.
Methods
NICU infants (N = 198, 30.4 ± 0.3 weeks gestation) were evaluated at 43 ± 0.4 weeks postmenstrual age using 24-h pH-impedance. Ten randomly selected 1-min windows during rest from the distal impedance channel (Z6) were averaged. Chi-square, t-tests, and ANOVA were used to compare pH-impedance and symptom characteristics by BI severity (BI < 900 Ω, BI 900–2000 Ω, BI > 2000 Ω). Regression analysis was used to identify potential contributing factors of BI.
Results
In BI < 900 Ω: (1) pH-impedance characteristics were increased (acid reflux event frequency, duration, and severity, all P < 0.05 vs. BI > 2000 Ω), and (2) positive symptom correlations were noted with bolus (73%) and acid events (55%). Significant predictors of BI included chronological age, acid reflux index, and BPD diagnosis (all P < 0.05).
Conclusions
Low BI is associated with prolonged acid exposure, delayed clearance, and greater aerodigestive symptom prevalence, likely associated with inflammation and or increased mucosal permeability. BI > 2000 Ω likely involves little or no inflammation because acid exposure is minimal. Combination of SAP, pH-impedance metrics, and BI along with the clarification of dysmotility mechanisms provides the rationale for personalized anti-reflux therapies as needed.
Similar content being viewed by others
Introduction
Gastroesophageal reflux (GER) is a physiological process in infants, and refers to the passage of gastric contents into the esophagus with or without regurgitation and vomiting.1,2 When troublesome symptoms or complications occur, GER becomes a pathological entity called GER disease (GERD).1,2 GERD can be associated with poor feeding, weight loss, esophagitis, and aerodigestive symptoms. NASPGHAN guidelines and GERD definitions are based on symptoms, although the symptoms are non-specific.1,2 For high-risk NICU infants and non-verbal patients, there is no gold standard for the definition, diagnosis, or management of GERD, based on pathophysiology. This has led to wide variation in pharmacological therapies ranging from 2 to 30% and prolonged hospitalization by 30 days, incurring an additional economic burden by about $70,000 per infant.3,4,5
Common diagnostic tests for GERD may include endoscopy or 24-h pH-impedance testing. However, in the NICU population endoscopic diagnosis of esophagitis is not readily feasible in distinguishing the severity of GERD. Thus, 24-h pH-impedance testing is the preferred test. Ionic/fluid movement caused by the passage of liquid, gas, or mixed boluses in the esophagus can be detected using 24-h pH-impedance, specifying the chemical, physical, spatial, and temporal properties of GER events.2,4,6,7 Electrical impedance measures the resistance to current flow between the adjacent impedance circuits, thus representing changes in conductivity.8,9 Impedance methods have been used to assess non-erosive reflux disease with the measurement of baseline impedance (BI) values in adults.10 Distal BI measurements are an indicator of the structural histopathologic changes in the esophageal mucosal epithelium.10 The degree of BI indicates the level of epithelial damage and has been reported in adults to decrease with increased acid.11 BI values have a close relationship with time of acid exposure, trans-epithelial resistance (functional), and dilation of intercellular spaces, all of which are reflected in various transformations of the stability (structure) of the esophageal mucosa.10,11 Specifically, mucosal inflammation has been associated with lower BI, with values <900 Ω indicating severe inflammation and >2000 Ω indicating normal integrity.12 Correlation between baseline impedance values and several reflux parameters in children has been recognized previously,13 as well as a correlation with macroscopic esophagitis.12
As the safety and efficacy of anti-reflux therapies is still unclear in infants, differences in BI and pH-impedance metrics may clarify pathophysiology and add to our diagnostic armamentarium. Therefore, our objectives were to characterize and differentiate the changes in esophageal mucosal integrity by determining BI and correlating BI with varying acid reflux indices in symptomatic neonatal ICU infants. We compared the characteristics of pH-impedance with BI threshold categories (<900, 900–2000, >2000 Ω). We hypothesized that lower BI is associated with a higher acid reflux index (ARI, a measure of acid-GERD severity due to prolonged exposure time) and therefore associated with higher symptom prevalence.
Materials and methods
Subjects
Infants tested were referred to the Innovative Neonatal and Infant Feeding Disorders Program at Nationwide Children’s Hospital, Columbus, OH. Infants were consulted for suspicion of GERD due to persistent troublesome symptoms, such as cardiorespiratory symptoms (coughing, sneezing, choking, apnea, bradycardia, desaturation, stridor, or chronic oxygen needs), feeding difficulties (poor oral intake, aspiration, feeding intolerance, symptoms during feeding) or GERD-type symptoms (arching/irritability and emesis) (Table 1). Subjects were included if they underwent 24-h pH-impedance testing between 2012 and 2015. If the subject had repeat studies, only the first study was included for analysis. Subjects with congenital airway, gastrointestinal (GI), or neurological anomalies were excluded, as well as those who were already receiving acid-suppressive therapy at the time of evaluation. All procedures were performed bedside at Nationwide Children’s Hospital Columbus, OH. Informed parental consent and institutional review board approval were obtained prior to the study. Compliance with the Health Insurance Portability and Accountability Act was followed.
pH-impedance methods
Twenty-four hour multichannel intraluminal impedance methods were performed using a pH-impedance probe (Greenfield MMS-Z1-I, 6.4 French, Medical Measurements Systems, Inc. Dover, New Hampshire) connected to a recording device (Ohmega, Laborie Medical Technologies, Mississauga, ON, Canada) as previously published.4,6,7 The pH-impedance probe-design had six impedance channels (Z1–Z6) spaced 1.5 cm apart and one pH sensor in the most distal channel. Prior to the study, the probe was calibrated with pH 4.0 and pH 7.0 buffer solutions. The probe was placed nasally in the supine-lying infant with the pH sensor in the distal esophagus at T7–T8 vertebral level, which was verified by X-ray.4,6,7,14 Infants were tested in the supine position, except during feeding or care times. Trained nursing assistants blinded to the pH-impedance data observed the infant for the entire 24-h study duration and recorded symptoms, meal times, and cares in real time using event markers on the recording device and written event logs. The nursing assistant’s sole responsibility during their shift was to observe and document symptoms in real time at the infant’s bedside. In this way, the quality of data acquisition was maintained in our lab.
Determination of BI
BI as measured in impedance channel 6 (distal esophagus) has shown a 100% prediction of grade 3 esophagitis (using endoscopy and histology) as well as macroscopic esophagitis.12 To develop a non-invasive marker for the test of mucosal integrity, BI was measured in the distal esophagus which is the area of frequent target for refluxate exposure.
Data analysis
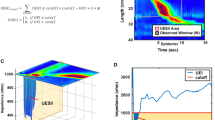
Characteristics of pH-impedance were analyzed using MMS analysis software (v.9.5, Laborie Medical Technologies, Mississauga, ON, Canada), while BI was analyzed using impedance data exported in csv format from MMS analysis software sampled at 10 Hz. The rater (NH) randomly selected ten 1-min windows, every 3 h during esophageal quiescence, i.e., absence of anterograde and retrograde activity from each patient. Periods during meal times and infant cares were excluded from analysis unless otherwise stated. Mean BI was calculated for the distal impedance channel (Z6) as shown in Fig. 1, and subjects were grouped into severe (BI < 900 Ω), moderate (BI 900–2000 Ω), or normal (BI > 2000 Ω) inflammation groups.12 Additionally, as previous work in children has shown a quicker and reliable method to determine BI by including all impedance data (i.e., retrograde and anterograde events) over the 24-h study duration,15 we also calculated the 24-h mean BI (including all impedance events) for correlation with the randomly selected 1-min BI windows samples (excluding impedance events).
Twenty-four hour pH-impedance tracing (top panel) and 1-min baseline impedance segment (lower panel). Ten 1-min windows (yellow bars) at rest (during non-mealtimes, and absence of anterograde or retrograde activity) were randomly selected for calculation of distal baseline impedance (BI). The Z6 (in distal esophagus) channel is close to the site of most acid exposure; hence, BI was calculated at this site
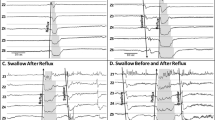
As previously published,4,6,7,16 the following pH-impedance characteristics were analyzed: (1) pH metrics included acid reflux index (ARI) as the % of acid (pH < 4) exposure time, number of acid reflux events (ARE) as acid events with pH < 4.0 for >5 s, number of acid reflux events >5 min, and longest acid reflux duration in seconds. (2) Impedance characteristics included number of ascending acid events (pH < 4), number of weakly acid events (pH 4–7), and total number of events (defined as retrograde reflux with impedance drops > 50% from baseline and reaching at least the Z5 channel). (3) Symptom characteristics including frequency of symptoms (symptoms/h) and positive symptom association probability (SAP) for pH and impedance events defined as SAP >95%.
Statistical analysis
Analysis was performed using Stata software (v. 14.0, StataCorp, College Station TX). Chi-square tests, t-tests, and ANOVA were used to compare clinical outcomes, and pH-impedance characteristics by BI severity groups (<900, 900–2000, and >2000 Ω). A multivariate linear regression model was developed to identify clinical and ph-impedance predictors of BI. Pearson correlation was used to test the correlation between the sample of ten 1-min windows for BI and the 24-h method. Median (interquartile range [IQR]), mean ± SD, or % are reported, unless stated otherwise. P-values < 0.05 were considered statistically significant.
Results
Clinical and outcome characteristics
A total of 198 neonates (105 males), born at 30.4 ± 0.3 (mean ± SD) weeks gestation were evaluated at 43.0 ± 0.4 weeks postmenstrual age (PMA). Of the 198 subjects analyzed, 11(6%) had BI < 900 Ω, 125 (63%) had BI 900–2000 Ω, and 62(31%) had BI > 2000 Ω. Clinical comparisons between the BI severity groups are shown in Table 1. Note that the infants in the BI > 2000 Ω group had decreased PPI use at discharge, but experienced higher prevalence of BPD (P < 0.01) and increased length of stay (P < 0.01). BI in infants with BPD was 1557 ± 69 Ω vs. 1628 ± 47 Ω in infants without BPD (P < 0.01). BI of infants with tube (nasogastric or orogastric) feeding was 1831 ± 41 Ω compared to 1765 ± 43 Ω for those who were exclusively oral fed at time of study (P = 0.6).
Twenty-four hour pH-impedance metrics
Characteristics of pH-impedance between BI groups are shown in Fig. 2 and Table 2. Note that the ARI, number of acid reflux events, number of AREs > 5 min, longest acid reflux, and number of ascending acid events is increased in the BI < 900 Ω group (P < 0.05). Additionally, although not statistically significant, the BI < 900 Ω group has a high prevalence of positive symptom correlation (SAP > 95%) for both ascending reflux events and acid events.
Effect of esophageal inflammation on esophageal acid exposure. *P < 0.05 vs. BI > 2000 Ω, †P < 0.05 vs. BI 900–2000 Ω. Box plots represent 25th and 75th percentiles; whiskers represent 10th and 90th percentile; median is represented by the black horizontal line and mean by the black dashed line. Acid reflux index (ARI) values are distinct between baseline impedance categories. Note the BI < 900 Ω (severe inflammation) category has increased ARI, whereas >2000 Ω (minimal inflammation) category has decreased ARI
Contributory factors of BI
Regression analysis for clinical and pH-impedance characteristics indicates the postmenstrual age (chronological + gestational age) is a significant predictor of BI (β = 17 ± 8, P = 0.04). However, chronological age and not gestational age was significantly associated with BI, while controlling for risk factors, such as NG-tube use, BPD diagnosis, ARI, and bolus clearance time (Table 3). The correlation coefficient between both the 24-h mean BI and the random 1-min window of quiescence was high (r = 0.88, P < 0.0001).
Discussion
The importance of BI in NICU neonates has not been determined in relation to esophageal pH-impedance measurements. Mucosal inflammation has been associated with lower BI, with values < 900 Ω indicating severity and >2000 Ω indicating normal integrity.12 The hallmark of the current study is the inclusion of data from resting state distal esophageal mucosal impedance, which represents changes in conductivity (lowered resistance) of ions at the mucosal level. This study was undertaken to characterize the mucosal integrity marker (BI < 900 Ω, BI 900–2000 Ω, BI > 2000 Ω) and compare clinical characteristics and pH-impedance metrics to clarify pathophysiology. Specifically, in symptomatic NICU infants evaluated for suspicion of GERD: (1) refluxate characteristics are distinct among BI thresholds, (2) subjects with BI < 900 Ω was observed in only 6% of the population, and likely indicates severe esophageal inflammation due to increased acid event frequency, duration, and severity, and although not significant, a high positive symptom correlation with acid (55%) events. (3) Subjects with BI > 2000 Ω likely involves little or no inflammation due to minimal acid exposure, and although not significant, a low prevalence of symptom correlation during acid (34%) events, (4) 24-h BI may be a potential diagnostic tool for rapid and cost effective assessment of esophageal mucosal integrity,11 in combination with SAP16 and pH-impedance metrics,4,6,7,9 and thus offers careful selection of subjects for personalized anti-reflux therapies.
BI values are reflective of changes in ion movement at the mucosal level during resting state.17,18 Because electrical impedance is inversely related to the ionic concentration and highly correlated with trans-epithelial resistance, at baseline in health, the impedance is high thereby indicating normal mucosal integrity. Thus baseline impedance function is attributed to the function at the intercellular spaces and in inflammation. When baseline impedance is low, there is greater ion transfer at the mucosal level and therefore increased permeability. Evidence to this pathophysiology is based on histology and baseline impedance studies in animal and human esophageal experimental models which suggested that the greater the distance between the mucosal intercellular spaces the lower the baseline impedance.17,19,20,21 These data also suggest that at low baseline impedance levels, ARI is high and many of the reflux parameters and positive symptom correlations are increased. The degree and duration of acid exposure has been highly correlated to esophageal inflammation across the sub-mucosal spaces affecting the neural elements in the submucosal and myenteric plexus.17,19,21 Thus, if there is damage to the integrity of esophageal mucosa, the impedance in that recording channel will be affected,12 and resting state BI is a more sensitive marker reflecting chronicity. Because distal esophagus is the most frequent target for acid exposure, we measured the BI at the distal channel (Z6).
True diagnosis and predictability of GERD remains controversial despite the use of multiple tests.22 Baseline impedance thresholds can become a major diagnostic marker for GERD prototypes. Understanding the reasons for symptoms remain a conundrum in non-verbal patients, and GERD is a frequent assumption by parents and providers. Unfortunately, the diagnostic criteria, characterization of severity or evidence-based therapeutic strategies for GERD remain unclear in vulnerable infant populations. Added to this, testing methods are not readily available in most centers, and the use of anti-reflux medications remains high despite the uncertainty of benefits and warnings about the side-effects.3,23 Carefully performed pH-impedance testing with symptom correlation and interpretation can clarify the correlation with troublesome symptoms on a case-by-case basis.7,24,25,26 The indications for GERD diagnosis, for non-pharmacological and pharmacological treatment strategies, for duration of therapies, and for medical and surgical therapies remain to be determined in this vulnerable NICU infant population.
Current pH-impedance metrics alone are inadequate as there is no clear consensus in NICU infants on who, when, how, and for how long to treat for GERD. Heightened esophageal sensitivity upon mechanosensitive or chemosensitive esophageal provocation can cause symptoms if the peristaltic reflexes are impaired and dysmotility is evident.27,28 Similar symptomatology is seen in non-erosive reflux disease in adults that are not responsive to acid-suppressive therapy, or may be the basis for functional esophageal disorders.19,21 Such a characterization in infants is not yet elucidated. Further studies are needed to distinguish the esophageal motility patterns among those with low vs. high baseline impedance. We also note that the low BI (<900 Ω) is associated with low ARI, as shown by the lower ranges in the box plots (Fig. 2), and such infants often do not respond to acid suppressive therapies. As in older patients,11,17,29,30 we speculate that such infants may have one of the several potential factors for such pathophysiology: (a) symptoms may be due to functional esophageal disorder with onset in neonatal period, (b) non-erosive reflux disease, (c) non-acid reflux disease, (d) eosinophilic esophageal disorders, or (e) food allergies. Clarification of such differential diagnosis may require further testing using provocative manometry and/or therapeutic interventions.31,32 We have laid down theoretical framework for potential personalized GERD strategies that may be developed, as follows: (1) A BI < 900 Ω indicates severe inflammation and would support the need for treatment with acid suppressive therapies. It may also provide anticipatory guidance for follow-up and duration of therapy based on response to acid suppressive therapy. (2) For infants with an indeterminate BI (900–2000 Ω), development of diagnostic and treatment strategies are needed. We suggest that an indeterminate BI with positive symptom association to acid could be considered pathologic and require treatment with acid suppressives, but indeterminate BI with negative symptom association may likely not need acid suppressive treatment. However, if there is a positive bolus symptom association, treatments targeting the height and frequency of refluxate may be warranted. (3) If an infant has a BI > 2000 Ω but recurring aerodigestive symptoms, other mechanisms besides GERD should be considered and investigated. Further research studies are needed to test diagnostic and therapeutic strategies based on a combination of diagnostic parameters.
Future directions and limitations from the current study are as follows: (1) the population studied was symptomatic infants referred for GERD. As it is not ethically appropriate to recruit asymptomatic neonates for pH-impedance testing, data do not exist for control comparisons. However, further testing could be performed to evaluate infants with normal ARI and negative symptom correlation as control comparisons (as these results would indicate no GER pathophysiology). (2) This was a single center study of infants in the inpatient setting; multicenter studies are needed for both, inpatient and outpatient settings. (3) SAP values may be prone to inaccuracy due to human error of charting. However, our lab has tried to minimize these inaccuracies by dedicated staff and training at the bedside to document symptoms in real time. Further studies are needed to develop automated detection of symptoms. (4) The focus of our current study was largely on the distal esophageal acid exposure indices and their correlational effects on the esophageal mucosal integrity marker. Further detailed study is needed to evaluate BI values in relation to anatomical regions of the pharynx, proximal and distal esophagus, and their effects on the motility metrics of the skeletal and smooth muscle portions of the esophagus. (5) Additionally, proximal baseline impedance has been utilized with BCT to determine esophageal dysmotility.33 We noted that when the distal BI was low, the BCT was larger, suggesting that inflammation modifies motility. Our prior work shows that bowel inflammation modifies the muscarinic signaling mechanisms in the smooth muscle.34 Further studies are needed to prospectively test this hypothesis. (6) Although there was high correlation with randomly selected 1-min windows and 24-h BI values, there may be instances where the 24 h BI is not accurate due to multiple patient factors, hence, clinical discretion is needed. (7) Age has been shown to have an effect on BI and esophagitis,35,36 while non-acid/weakly acid reflux has been shown to be related to symptoms, specifically in the infant population and respiratory manifestations.37,38,39,40,41,42,43,44,45 However, as endoscopy was not performed in the current study, further work is needed to gather prospective data regarding subject factors and response to tailored therapies targeting non-acid, weakly acid, and acid GER.
In conclusion, as clinical suspicion of GERD is often inaccurate, distal baseline impedance is likely a valuable addition to pH-impedance testing to objectively discern GERD in NICU infants. The inverse relationship of esophageal acid exposure index to esophageal baseline impedance suggests that mucosal disruption may result from acid reflux exposure in these high-risk vulnerable infants. Infants with suspected GERD should be thoroughly investigated before beginning anti-reflux medications or life-style changing medical or surgical therapies. Understanding the pathophysiological basis for symptoms or signs in relation to objectively diagnosed GERD will pave way for well-targeted therapies and successful outcomes. A combination of SAP, pH-impedance metrics, and BI along with the clarification of dysmotility mechanisms provides rationale for careful selection of subjects for personalized anti-reflux therapies, either medical or surgical.
References
Rosen, R. et al. Pediatric Gastroesophageal Reflux Clinical Practice Guidelines: Joint Recommendations of the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition and the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition. J. Pediatr. Gastroenterol. Nutr. 66, 516–554 (2018).
Vandenplas, Y. et al. Pediatric gastroesophageal reflux clinical practice guidelines: joint recommendations of the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition (NASPGHAN) and the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN). J. Pediatr. Gastroenterol. Nutr. 49, 498–547 (2009).
Jadcherla, S. R. et al. Practice variance, prevalence, and economic burden of premature infants diagnosed with GERD. Hosp. Pediatr. 3, 335–341 (2013).
Jadcherla, S. R. et al. Significance of gastroesophageal refluxate in relation to physical, chemical, and spatiotemporal characteristics in symptomatic intensive care unit neonates. Pediatr. Res. 70, 192–198 (2011).
Slaughter, J. L. et al. Neonatal histamine-2 receptor antagonist and proton pump inhibitor treatment at United States Children’s Hospitals. J. Pediatr. 174, 63–70.e63 (2016).
Jadcherla, S. R. et al. Spatiotemporal characteristics of acid refluxate and relationship to symptoms in premature and term infants with chronic lung disease. Am. J. Gastroenterol. 103, 720–728 (2008).
Sivalingam, M. et al. Effects of esophageal acidification on troublesome symptoms: an approach to characterize true acid GERD in dysphagic neonates. Dysphagia 32, 509–519 (2017).
Bredenoord, A. J. et al. Technology review: esophageal impedance monitoring. Am. J. Gastroenterol. 102, 187–194 (2007).
Loots, C. M.et al. Esophageal impedance baselines in infants before and after placebo and proton pump inhibitor therapy. Neurogastroenterol. Motil. 24, 758–762, e351–e352 (2012).
Zhong, C. et al. Esophageal intraluminal baseline impedance is associated with severity of acid reflux and epithelial structural abnormalities in patients with gastroesophageal reflux disease. J. Gastroenterol. 48, 601–610 (2013).
Borrelli, O. et al. Relationship between baseline impedance levels and esophageal mucosal integrity in children with erosive and non-erosive reflux disease. Neurogastroenterol. Motil. 24, 828–e394 (2012).
Cohen Sabban, J. et al. Low-impedance baseline values predict severe esophagitis. J. Pediatr. Gastroenterol. Nutr. 65, 278–280 (2017).
van der Pol, R. J. et al. Outcomes of endoscopy and novel pH-impedance parameters in children: is there a correlation? J. Pediatr. Gastroenterol. Nutr. 56, 196–200 (2013).
Gupta, A. & Jadcherla, S. R. The relationship between somatic growth and in vivo esophageal segmental and sphincteric growth in human neonates. J. Pediatr. Gastroenterol. Nutr. 43, 35–41 (2006).
Ummarino, D. et al. Esophageal impedance baseline according to different time intervals. Eur. J. Med. Res. 17, 18 (2012).
Bredenoord, A. J., Weusten, B. L. & Smout, A. J. Symptom association analysis in ambulatory gastro-oesophageal reflux monitoring. Gut 54, 1810–1817 (2005).
Farre, R. et al. Evaluation of oesophageal mucosa integrity by the intraluminal impedance technique. Gut 60, 885–892 (2011).
Silny, J. Intraluminal multiple electric impedance procedure for measurement of gastrointestinal motility. Wiley Online Libr. 3, 151–162 (1991).
Barlow, J. D., Gregersen, H. & Thompson, D. G. Identification of the biomechanical factors associated with the perception of distension in the human esophagus. Am. J. Physiol. Gastrointest. Liver Physiol. 282, G683–G689 (2002).
Kessing, B. F. et al. Esophageal acid exposure decreases intraluminal baseline impedance levels. Am. J. Gastroenterol. 106, 2093–2097 (2011).
Barlow, W. J. & Orlando, R. C. The pathogenesis of heartburn in nonerosive reflux disease: a unifying hypothesis. Gastroenterology 128, 771–778 (2005).
Salvatore, S. et al. Behind the (impedance) baseline in children. Dis. Esophagus 27, 726–731 (2014).
Slaughter, J. L., Stenger, M. R. & Reagan, P. B. Variation in the use of diuretic therapy for infants with bronchopulmonary dysplasia. Pediatrics 131, 716–723 (2013).
Qureshi, A. et al. The role of sleep in the modulation of gastroesophageal reflux and symptoms in NICU neonates. Pediatr. Neurol. 53, 226–232 (2015).
Sankaran, J. et al. Effect of severity of esophageal acidification on sleep vs wake periods in infants presenting with brief resolved unexplained events. J. Pediatr. 179, 42–48 e41 (2016).
Bhat, R. Y. et al. Acid gastroesophageal reflux in convalescent preterm infants: effect of posture and relationship to apnea. Pediatr. Res. 62, 620–623 (2007).
Jadcherla, S. R. et al. Mechanisms of cough provocation and cough resolution in neonates with bronchopulmonary dysplasia. Pediatr. Res. 78, 462–469 (2015).
Hasenstab, K. A. & Jadcherla, S. R. Respiratory events in infants presenting with apparent life threatening events: is there an explanation from esophageal motility? J. Pediatr. 165, 250–255 e251 (2014).
Ravi, K., et al. Baseline impedance measured during high-resolution esophageal impedance manometry reliably discriminates GERD patients. Neurogastroenterol. Motil. 29, 1–8 e12974 (2017).
Heard, R. et al. Characterization of patients with low baseline impedance on multichannel intraluminal impedance-pH reflux testing. J. Clin. Gastroenterol. 46, e55–e57 (2012).
Jadcherla, S. R. et al. Impact of personalized feeding program in 100 NICU infants: pathophysiology-based approach for better outcomes. J. Pediatr. Gastroenterol. Nutr. 54, 62–70 (2012).
Jadcherla, S. R. et al. Feeding methods at discharge predict long-term feeding and neurodevelopmental outcomes in preterm infants referred for gastrostomy evaluation. J. Pediatr. 181, 125–130 e121 (2017).
Tambucci, R. et al. Clinical relevance of esophageal baseline impedance measurement: just an innocent bystander. J. Pediatr. Gastroenterol. Nutr. 60, 776–782 (2015).
Jadcherla, S. R. Inflammation inhibits muscarinic signaling in in vivo canine colonic circular smooth muscle cells. Pediatr. Res. 52, 756–762 (2002).
Salvatore, S. et al. Esophageal impedance baseline is age dependent. J. Pediatr. Gastroenterol. Nutr. 57, 506–513 (2013).
Salvatore, S. et al. Low mean impedance in 24-hour tracings and esophagitis in children: a strong connection. Dis. Esophagus 29, 10–14 (2016).
Mousa, H. et al. Testing the association between gastroesophageal reflux and apnea in infants. J. Pediatr. Gastroenterol. Nutr. 41, 169–177 (2005).
Salvatore, S. et al. Esophageal impedance in children: symptom-based results. J. Pediatr. 157, 949–954.e1–2 (2010).
Rosen, R. & Nurko, S. The importance of multichannel intraluminal impedance in the evaluation of children with persistent respiratory symptoms. Am. J. Gastroenterol. 99, 2452–2458 (2004).
Rosen, R. et al. The utility of endoscopy and multichannel intraluminal impedance testing in children with cough and wheezing. Pediatr. Pulmonol. 49, 1090–1096 (2014).
Ghezzi, M. et al. Acid and weakly acid gastroesophageal refluxes and type of respiratory symptoms in children. Respir. Med. 105, 972–978 (2011).
Borrelli, O. et al. Role of gastroesophageal reflux in children with unexplained chronic cough. J. Pediatr. Gastroenterol. Nutr. 53, 287–292 (2011).
Francavilla, R. et al. Comparison of esophageal pH and multichannel intraluminal impedance testing in pediatric patients with suspected gastroesophageal reflux. J. Pediatr. Gastroenterol. Nutr. 50, 154–160 (2010).
Corvaglia, L. et al. The frequency of apneas in very preterm infants is increased after non-acid gastro-esophageal reflux. Neurogastroenterol. Motil. 23, 303–307, e152 (2011).
Mattioli, G. et al. Esophageal impedance/pH monitoring in pediatric patients: preliminary experience with 50 cases. Dig. Dis. Sci. 51, 2341–2347 (2006).
ACKNOWLEDGMENTS
We are grateful to Rebecca Moore, BSN RN, for helping with IRB coordination, demographic data extraction, and verification. The study was supported in part by RO1 NIH DK 068158.
Author information
Authors and Affiliations
Contributions
S.R.J. developed the concepts, study design, obtained grant funding and ethical approvals from IRB. S.N. and K.A.H. were also associated with analysis design. N.H. and K.A.H. acquired data and performed analysis. S.N. performed statistical analysis. N.H. created figures. S.R.J., N.H., K.A.H., and S.N. were all involved with data verification, interpretation of results, writing, editing, and approval of final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jadcherla, S.R., Hanandeh, N., Hasenstab, K.A. et al. Differentiation of esophageal pH-impedance characteristics classified by the mucosal integrity marker in human neonates. Pediatr Res 85, 355–360 (2019). https://doi.org/10.1038/s41390-018-0237-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-018-0237-z
This article is cited by
-
Characteristics of esophageal refluxate and symptoms in infants compared between pre-treatment and on treatment with proton pump inhibitors
Journal of Perinatology (2024)
-
Differentiating esophageal sensitivity phenotypes using pH–impedance in intensive care unit infants referred for gastroesophageal reflux symptoms
Pediatric Research (2021)