Abstract
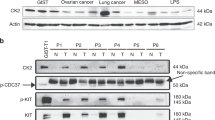
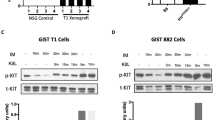
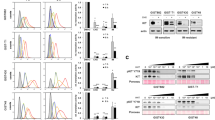
Oncogenic KIT or PDGFRA tyrosine kinase mutations are compelling therapeutic targets in most gastrointestinal stromal tumors (GISTs), and the KIT inhibitor, imatinib, is therefore standard of care for patients with metastatic GIST. However, some GISTs lose expression of KIT oncoproteins, and therefore become KIT-independent and are consequently resistant to KIT-inhibitor drugs. We identified distinctive biologic features in KIT-independent, imatinib-resistant GISTs as a step towards identifying drug targets in these poorly understood tumors. We developed isogenic GIST lines in which the parental forms were KIT oncoprotein-dependent, whereas sublines had loss of KIT oncoprotein expression, accompanied by markedly downregulated expression of the GIST biomarker, protein kinase C-theta (PRKCQ). Biologic mechanisms unique to KIT-independent GISTs were identified by transcriptome sequencing, qRT-PCR, immunoblotting, protein interaction studies, knockdown and expression assays, and dual-luciferase assays. Transcriptome sequencing showed that cyclin D1 expression was extremely low in two of three parental KIT-dependent GIST lines, whereas cyclin D1 expression was high in each of the KIT-independent GIST sublines. Cyclin D1 inhibition in KIT-independent GISTs had anti-proliferative and pro-apoptotic effects, associated with Rb activation and p27 upregulation. PRKCQ, but not KIT, was a negative regulator of cyclin D1 expression, whereas JUN and Hippo pathway effectors YAP and TAZ were positive regulators of cyclin D1 expression. PRKCQ, JUN, and the Hippo pathway coordinately regulate GIST cyclin D1 expression. These findings highlight the roles of PRKCQ, JUN, Hippo, and cyclin D1 as oncogenic mediators in GISTs that have converted, during TKI-therapy, to a KIT-independent state. Inhibitors of these pathways could be effective therapeutically for these now untreatable tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Hum Pathol. 2002;33:459–65.
Corless CL, Fletcher JA, Heinrich MC. Biology of gastrointestinal stromal tumors. J Clin Oncol. 2004;22:3813–25.
Hirota S, Isozaki K, Moriyama Y, Hashimoto K, Nishida T, Ishiguro S, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279:577–80.
Heinrich MC, Corless CL, Duensing A, McGreevey L, Chen CJ, Joseph N, et al. PDGFRA activating mutations in gastrointestinal stromal tumors. Science. 2003;299:708–10.
Tuveson DA, Willis NA, Jacks T, Griffin JD, Singer S, Fletcher CD, et al. STI571 inactivation of the gastrointestinal stromal tumor c-KIT oncoprotein: biological and clinical implications. Oncogene. 2001;20:5054–8.
Demetri GD, von Mehren M, Blanke CD, Van den Abbeele AD, Eisenberg B, Roberts PJ, et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med. 2002;347:472–80.
Heinrich MC, Maki RG, Corless CL, Antonescu CR, Harlow A, Griffith D, et al. Primary and secondary kinase genotypes correlate with the biological and clinical activity of sunitinib in imatinib-resistant gastrointestinal stromal tumor. J Clin Oncol. 2008;26:5352–9.
Blay JY, Bonvalot S, Casali P, Choi H, Debiec-Richter M, Dei Tos AP, et al. Consensus meeting for the management of gastrointestinal stromal tumors. Report of the GIST Consensus Conference of 20–21 March 2004, under the auspices of ESMO. Ann Oncol. 2005;16:566–78.
Demetri GD, Heinrich MC, Fletcher JA, Fletcher CD, Van den Abbeele AD, Corless CL, et al. Molecular target modulation, imaging, and clinical evaluation of gastrointestinal stromal tumor patients treated with sunitinib malate after imatinib failure. Clin Cancer Res. 2009;15:5902–9.
Essat M, Cooper K. Imatinib as adjuvant therapy for gastrointestinal stromal tumors: a systematic review. Int J Cancer. 2011;128:2202–14.
Heinrich MC, Corless CL, Blanke CD, Demetri GD, Joensuu H, Roberts PJ, et al. Molecular correlates of imatinib resistance in gastrointestinal stromal tumors. J Clin Oncol. 2006;24:4764–74.
Liegl B, Kepten I, Le C, Zhu M, Demetri GD, Heinrich MC, et al. Heterogeneity of kinase inhibitor resistance mechanisms in GIST. J Pathol. 2008;216:64–74.
Fletcher JA, Corless CL, Dimitrijevic S, Von Mehren B, Eisenberg B, Joensuu H, et al. Mechanisms of resistance to imatinib mesylate (IM) in advanced gastrointestinal stromal tumor (GIST). Proc Am Soc Clin Oncol. 2003;22:815.
Debiec-Rychter M, Cools J, Dumez H, Sciot R, Stul M, Mentens N, et al. Mechanisms of resistance to imatinib mesylate in gastrointestinal stromal tumors and activity of the PKC412 inhibitor against imatinib-resistant mutants. Gastroenterology. 2005;128:270–9.
Duensing A, Joseph NE, Medeiros F, Smith F, Hornick JL, Heinrich MC, et al. Protein Kinase C theta (PKCtheta) expression and constitutive activation in gastrointestinal stromal tumors (GISTs). Cancer Res. 2004;64:5127–31.
Ou WB, Zhu MJ, Demetri GD, Fletcher CD, Fletcher JA. Protein kinase C-theta regulates KIT expression and proliferation in gastrointestinal stromal tumors. Oncogene. 2008;27:5624–34.
Page K, Li J, Corbit KC, Rumilla KM, Soh JW, Weinstein IB, et al. Regulation of airway smooth muscle cyclin D1 transcription by protein kinase C-delta. Am J Respir Cell Mol Biol. 2002;27:204–13.
Hizli AA, Black AR, Pysz MA, Black JD. Protein kinase C alpha signaling inhibits cyclin D1 translation in intestinal epithelial cells. J Biol Chem. 2006;281:14596–603.
Li H, Weinstein IB. Protein kinase C beta enhances growth and expression of cyclin D1 in human breast cancer cells. Cancer Res. 2006;66:11399–408.
Guan L, Song K, Pysz MA, Curry KJ, Hizli AA, Danielpour D, et al. Protein kinase C-mediated down-regulation of cyclin D1 involves activation of the translational repressor 4E-BP1 via a phosphoinositide 3-kinase/Akt-independent, protein phosphatase 2A-dependent mechanism in intestinal epithelial cells. J Biol Chem. 2007;282:14213–25.
Siegmund K, Thuille N, Posch N, Fresser F, Baier G. Novel protein kinase C theta: coronin 1A complex in T lymphocytes. Cell Commun Signal. 2015;13:22.
Snyder EL, Sandstrom DJ, Law K, Fiore C, Sicinska E, Brito J, et al. c-Jun amplification and overexpression are oncogenic in liposarcoma but not always sufficient to inhibit the adipocytic differentiation programme. J Pathol. 2009;218:292–300.
DeMatteo RP, Ballman KV, Antonescu CR, Maki RG, Pisters PW, Demetri GD, et al. Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: a randomised, double-blind, placebo-controlled trial. Lancet. 2009;373:1097–104.
Serrano C, Wang Y, Marino-Enriquez A, Lee JC, Ravegnini G, Morgan JA, et al. KRAS and KIT gatekeeper mutations confer polyclonal primary imatinib resistance in GI stromal tumors: relevance of concomitant phosphatidylinositol 3-kinase/AKT dysregulation. J Clin Oncol. 2015;33:e93–e96.
Coqueret O. Linking cyclins to transcriptional control. Gene. 2002;299:35–55.
Jirawatnotai S, Hu Y, Michowski W, Elias JE, Becks L, Bienvenu F, et al. A function for cyclin D1 in DNA repair uncovered by protein interactome analyses in human cancers. Nature. 2011;474:230–4.
Casimiro MC, Di Sante G, Di Rocco A, Loro E, Pupo C, Pestell TG, et al. Cyclin D1 restrains oncogene-induced autophagy by regulating the AMPK-LKB1 signaling axis. Cancer Res. 2017;77:3391–405.
Sherr CJ. Cancer cell cycles. Science. 1996;274:1672–7.
Diehl JA. Cycling to cancer with cyclin D1. Cancer Biol Ther. 2002;1:226–31.
Li Z, Wang C, Prendergast GC, Pestell RG. Cyclin D1 functions in cell migration. Cell Cycle. 2006;5:2440–2.
Landis MW, Pawlyk BS, Li T, Sicinski P, Hinds PW. Cyclin D1-dependent kinase activity in murine development and mammary tumorigenesis. Cancer Cell. 2006;9:13–22.
Lee YM, Sicinski P. Targeting cyclins and cyclin-dependent kinases in cancer: lessons from mice, hopes for therapeutic applications in human. Cell Cycle. 2006;5:2110–4.
Shan J, Zhao W, Gu W. Suppression of cancer cell growth by promoting cyclin D1 degradation. Mol Cell. 2009;36:469–76.
Barbash O, Diehl JA. SCF(Fbx4/alphaB-crystallin) E3 ligase: when one is not enough. Cell Cycle. 2008;7:2983–6.
Barbash O, Zamfirova P, Lin DI, Chen X, Yang K, Nakagawa H, et al. Mutations in Fbx4 inhibit dimerization of the SCF(Fbx4) ligase and contribute to cyclin D1 overexpression in human cancer. Cancer Cell. 2008;14:68–78.
Yang P, Chen W, Li X, Eilers G, He Q, Liu L, et al. Downregulation of cyclin D1 sensitizes cancer cells to MDM2 antagonist Nutlin-3. Oncotarget. 2016;7:32652–63.
Pruneri G, Mazzarol G, Fabris S, Del Curto B, Bertolini F, Neri A, et al. Cyclin D3 immunoreactivity in gastrointestinal stromal tumors is independent of cyclin D3 gene amplification and is associated with nuclear p27 accumulation. Mod Pathol. 2003;16:886–92.
Draper N, Bui M, Boulware DC, Lloyd M, Chiappori AA, Pledger WJ, et al. Increased cyclin D3 expression significantly correlates with p27 nuclear positivity in gastrointestinal stromal tumors. Hum Pathol. 2008;39:1784–91.
Tashiro E, Tsuchiya A, Imoto M. Functions of cyclin D1 as an oncogene and regulation of cyclin D1 expression. Cancer Sci. 2007;98:629–35.
Fernando R, Foster JS, Bible A, Ström A, Pestell RG, Rao M, et al. Breast cancer cell proliferation is inhibited by BAD: regulation of cyclin D1. J Biol Chem. 2007;282:28864–73.
Vikhanskaya F, Toh WH, Dulloo I, Wu Q, Boominathan L, Ng HH, et al. p73 supports cellular growth through c-Jun-dependent AP-1 transactivation. Nat Cell Biol. 2007;9:698–705.
Mizuno T, Murakami H, Fujii M, Ishiguro F, Tanaka I, Kondo Y, et al. YAP induces malignant mesothelioma cell proliferation by upregulating transcription of cell cyclepromoting genes. Oncogene. 2012;31:5117–22.
Blay P, Astudillo A, Buesa JM, Campo E, Abad M, García-García J, et al. Protein kinase C theta is highly expressed in gastrointestinal stromal tumors but not in other mesenchymal neoplasias. Clin Cancer Res. 2004;10:4089–95.
Motegi A, Sakurai S, Nakayama H, Sano T, Oyama T, Nakajima T. PKC theta, a novel immunohistochemical marker for gastrointestinal stromal tumors (GIST), especially useful for identifying KIT-negative tumors. Pathol Int. 2005;55:106–12.
Zhu MJ, Ou WB, Fletcher CD, Cohen PS, Demetri GD, Fletcher JA. KIT oncoprotein interactions in gastrointestinal stromal tumors: therapeutic relevance. Oncogene. 2007;26:6386–95.
Shaulian E, Karin M. AP-1 in cell proliferation and survival. Oncogene. 2001;20:2390–400.
Mariani O, Brennetot C, Coindre JM, Gruel N, Ganem C, Delattre O, et al. JUN oncogene amplification and overexpression block adipocytic differentiation in highly aggressive sarcomas. Cancer Cell. 2007;11:361–74.
Shen Q, Uray IP, Li Y, Krisko TI, Strecker TE, Kim HT, et al. The AP-1 transcription factor regulates breast cancer cell growth via cyclins and E2F factors. Oncogene. 2008;27:366–77.
Bauer S, Duensing A, Demetri GD, Fletcher JA. KIT oncogenic signaling mechanisms in imatinib-resistant gastrointestinal stromal tumor: PI3-kinase/AKT is a crucial survival pathway. Oncogene. 2007;26:7560–8.
Rubin BP, Singer S, Tsao C, Duensing A, Lux ML, Ruiz R, et al. KIT activation is a ubiquitous feature of gastrointestinal stromal tumors. Cancer Res. 2001;61:8118–21.
Huang SF, Xiao S, Renshaw AA, Loughlin KR, Hudson TJ, Fletcher JA. Fluorescence in situ hybridization evaluation of chromosome deletion patterns in prostate cancer. Am J Pathol. 1996;149:1565–73.
Thomazy VA, Luthra R, Uthman MO, Davies PJ, Medeiros LJ. Determination of cyclin D1 and CD20 mRNA levels by real-time quantitative RT-PCR from archival tissue sections of mantle cell lymphoma and other non-Hodgkin’s lymphomas. J Mol Diagn. 2002;4:201–8.
Husson H, Carideo EG, Neuberg D, Schultze J, Munoz O, Marks PW, et al. Gene expression profiling of follicular lymphoma and normal germinal center B cells using cDNA arrays. Blood. 2002;99:282–9.
Jean S, Bideau C, Bellon L, Halimi G, De Méo M, Orsière T, et al. The expression of genes induced in melanocytes by exposure to 365-nm UVA: study by cDNA arrays and real-time quantitative RT-PCR. Biochim Biophys Acta. 2001;1522:89–96.
Acknowledgements
This work was supported by NIH grants 1P50CA127003 and 1P50CA168512, and by the GIST Cancer Research Fund, SARC and David Foundation. This research was also supported by National Natural Science Foundation of China (81728012, 81602061), the Natural Science Foundation of Zhejiang Province (LY18H160065), Zhejiang medical and health science and technology plan project (2018KY651), Zhejiang Xinmiao Talents Program (2017R406050), National Undergraduate Training Program for Innovation and Entrepreneurship, Science Foundation of Zhejiang Sci-Tech University (14042107-Y), Graduate research and innovation projects of Zhejiang Sci-Tech University, China. We thank Adalis Maisonet, Terry Haley, and Edward Fox for technical and biostatistical support in the whole transcriptome sequencing.
Author information
Authors and Affiliations
Contributions
Study concept and design: WO, JAF; Data acquisition: WO, NN, HW, WZ, MZ, AK, DW, GE, GDD, HQ, BL, AM-E, JAF; Analysis and interpretation of data: WO, NN, MZ, AK, GE, AM-E, JAF; Drafting of the manuscript: WO, AM-E, JAF; Technical or material support: WO, GDD, JAF; Study supervision: WO, JAF.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ou, WB., Ni, N., Zuo, R. et al. Cyclin D1 is a mediator of gastrointestinal stromal tumor KIT-independence. Oncogene 38, 6615–6629 (2019). https://doi.org/10.1038/s41388-019-0894-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-019-0894-3