Abstract
We hypothesized that reactive O2 species, or their intermediary products, generated during exposure to elevated O2 lead to pathologic endothelin-1 expression in the newborn lung. Endothelin-1 expression and 8-isoprostane content (an in vivo marker of lipid peroxidation) were examined and found to be elevated (p < 0.05) in the lungs of newborn rats with abnormal lung morphology and pulmonary hypertension, as assessed by right ventricular hypertrophy, after a 14-d exposure to 60% O2. The antioxidant and lipid hydroperoxide scavenger, U74389G (10 mg/kg), given by daily i.p. injection prevented O2-dependent right ventricular hypertrophy (p < 0.05 compared with vehicle-treated controls), but had no effect on abnormal lung morphology. Additionally, we observed that 8-isoprostane caused marked endothelin-1 mRNA up-regulation in vitro in primary rat fetal lung cell cultures. We conclude that reactive O2 species, or their bioactive intermediaries, are causative in O2-mediated pulmonary hypertension and endothelin-1 up-regulation. It is likely that the bioactive lipid peroxidation product, 8-isoprostane, plays a key role in pathologic endothelin-1 expression and pulmonary hypertension during oxidant stress.
Similar content being viewed by others
Main
The use of sustained supplemental O2 and mechanical ventilation in premature infants with respiratory failure is associated with the development of bronchopulmonary dysplasia (BPD), a chronic neonatal lung injury (1). Characteristic features of BPD include a dysregulation of lung growth, including fibroproliferative changes (2). This condition remains an important cause of mortality and morbidity during early infancy and in later childhood (3). There are currently no rational or effective therapies available for the prevention or treatment of BPD and its accompanying complications. The putative central role for O2 toxicity in the pathogenesis of BPD has led to considerable interest in the use of antioxidant therapies as preventive and therapeutic agents (4).
Pulmonary hypertension is a common complication in infants with established BPD (5) and early respiratory failure (6), and is an important adverse factor in their outcome (7). An increased expression of ET-1 is observed in adult patients with pulmonary hypertension (8), which may play a causative role. The endothelins are a family of 21-amino acid peptides that are the most potent vasoconstrictor substances yet described (9). ET-1 is the dominant form in the lung and is expressed by multiple cell types, principally vascular endothelium and bronchial epithelium (10, 11). Both ETA and ETB receptors mediate the effects of ET-1. These receptors are both expressed in vascular and bronchial smooth muscle cells (11, 12), whereas vascular endothelial cells express only ETB receptors (13).
Factors known to up-regulate ET-1 include hypoxia (14, 15), proinflammatory cytokines (16), and shear stress (17). We have recently described a newborn rat model with BPD-like histopathology (18) and pulmonary hypertension (19) induced by exposure to 60% O2 for 14 d. Current evidence implicates reactive O2 species, generated in excess of antioxidant defenses, either directly or through the generation of bioactive intermediary products, in the pathogenesis of pulmonary O2 toxicity (20–22). Whether reactive O2 species and reactive O2 species-induced products directly stimulate ET-1 synthesis in the lung is not known.
We hypothesized that hyperoxia leads to lung injury, pulmonary vascular abnormalities, and ET-1 up-regulation in this model through the increased production of reactive O2 species (21), which may exert their effect through the peroxidation of membrane lipids and the consequent release of reactive products such as lipid hydroperoxides and 8-isoprostane (22). To test this hypothesis, we examined the effect of 60% O2 on ET-1 expression in neonatal rat lung tissue and the modifying effect of the synthetic 21-aminosteroid (lazaroid) compound, U74389G. The 21-aminosteroids are derivatives of methylprednisolone that are devoid of glucocorticoid activity (23). This class of compounds is lipophilic and has been reported to scavenge lipid hydroperoxide and superoxide radicals, and to effectively inhibit iron-dependent hydroxyl radical formation and lipid peroxidation (24–27). U74389F, another 21-aminosteroid with a different spectrum of antioxidant properties, has been reported to protect against the alveolar structural changes and pulmonary hypertension observed in neonatal rats exposed to 95% O2 (28).
METHODS
Materials.
U74389G and CS-4 vehicle (20 mM citric acid monohydrate, 3.2 mM sodium citrate dihydrate, 77 mM NaCl, pH 3.0) were kindly provided by Dr. D. Zimmerman (Pharmacia & Upjohn, Kalamazoo, MI, U.S.A.). Radioisotopes and nylon membranes were purchased from Amersham Canada (Oakville, Ontario), and restriction enzymes were from Amersham Pharmacia Biotech (Baie D'Urfé, Québec). BSA type V, Ficoll 400, polyvinylpyrrolidone, and guanidine thiocyanate were from Sigma Chemical Co. (St. Louis, MO, U.S.A.). Herring sperm DNA was from Roche Molecular Biochemicals (Laval, Québec). Organic solvents were of HPLC grade. A partial prepro-ET-1 cDNA probe (insert size, 543 bp) was synthesized and amplified by reverse transcriptase PCR based on the rat pulmonary endothelial cell cDNA sequence, as described by Terada et al. (29). A plasmid containing a full-length cDNA (insert size, 1.27 kb) of the rat GAPDH (EC 1.2.1.12) was a generous gift from Dr. M. Rabinovitch (University of Toronto, Ontario). Rabbit antiserum to human ET-1 was from Biochem Immunosystems (Montreal, Québec), and goat anti-rabbit IgG-biotin antibody was from GIBCO (Burlington, Ontario). Mouse MAb to α-smooth muscle actin was obtained from Lab Vision (Fremont, CA, U.S.A.), and sheep anti-mouse IgG-biotin antibody was from Santa Cruz Biotechnology (Santa Cruz, CA, U.S.A.). Avidin-biotin-peroxidase and alkaline phosphatase complex immunohistochemistry kits were purchased from Vector Laboratories (Burlingame, CA, U.S.A.). 8-Isoprostane and an 8-isoprostane enzyme immunoassay kit were from Cayman (Ann Arbor, MI, U.S.A.), total protein assay kit was from Bio-Rad (Hercules, CA, U.S.A.), and Sep Pak C18 cartridges were from Waters (Mississauga, Ontario).
Institutional review.
All procedures involving animals were conducted according to criteria established by the Canadian Council for Animal Care. Approval for the study was obtained from the Animal Care Review Committee of the Samuel Lunenfeld Research Institute, Mount Sinai Hospital.
Exposure system.
This has been described in detail previously (18). Briefly, pathogen-free timed pregnant Sprague Dawley rats (250–275 g) were obtained from Charles River (St. Constant, Quebec). Experiments were conducted as paired exposures with one chamber receiving 60% O2 and the other receiving air. On the anticipated day of delivery, each dam was placed in a 60 × 48 × 25-cm plastic chamber with 12 h/12 h light-dark cycles, a temperature maintained at 25 ± 1°C, minimal humidity, and a CO2 concentration of < 0.5%. Food and water were available ad libitum. Equal litter sizes (10–12 pups) were maintained between paired chambers. Dams were exchanged daily between chambers to prevent maternal O2 toxicity. At the termination of each exposure period, animals were killed by ether inhalation.
Interventions.
Pups were maintained in four paired chambers (air and O2) for a 14-d exposure period, each pair receiving either CS-4 vehicle (vehicle control) or U74389G in CS-4 vehicle (2 mg/mL and 10 mg/kg/d). Daily injections (5 μL/g), commenced within 6 h of birth, were via a 30-gauge needle into the right iliac fossa.
Examination of RVH.
RVH is a well-established index of pulmonary hypertension (30) and has been shown to have a direct correlation with vascular smooth muscle hyperplasia in this model (19). At death, the thoracic contents were removed en bloc. The heart was then separated from the lungs, and the right ventricle was dissected free from the left ventricle and septum. Each component was freeze-dried and weighed separately.
Isolation of lung RNA.
The lungs were dissected away from large vessels and airways to be flash frozen in liquid N2 immediately after weighing. Total (nuclear and cytoplasmic) RNA was isolated by lysis of tissue in 4 M guanidine thiocyanate, followed by phenol/chloroform extraction according to the method of Chomsczynski and Sacchi (31).
RNA analyses.
For Northern blot analyses, 20 μg of total lung RNA was fractionated on 1.3% (wt/vol) agarose gels containing 0.66 M formaldehyde and transferred to a nylon membrane. Prepro-ET-1 and GAPDH cDNAs were labeled with deoxycytidine 5′-[α-32P]triphosphate by a random priming labeling system (Amersham, Arlington Heights, IL, U.S.A.), with specific activities of 0.5–2.9 × 109 counts·min−1·μg DNA−1. For GAPDH mRNA, prehybridization (> 4 h) and hybridization (overnight) were performed in 50% (vol/vol) formamide, 5× SSPE, [750 mM NaCl, 50 mM sodium phosphate, 5 mM EDTA, pH 7.4], 5× Denhardt's solution [0.1% (wt/vol) each of BSA, Ficoll, and polyvinylpyrrolidone], 0.5% (wt/vol) SDS, and 100 μg/mL denatured herring sperm DNA at 42°C. For prepro-ET-1 mRNA, prehybridization and hybridization were performed in 5× SSPE, 5× Denhardt's solution, 0.5% (wt/vol) SDS, and 100 μg/mL denatured herring sperm DNA at 65°C. After hybridization, GAPDH blots were washed in 2× SSC [300 mM NaCl, 30 mM sodium citrate, pH 70]; 0.1% SDS for 10 min at room temperature. Prepro-ET-1 blots were washed in 2× SSC; 0.1% SDS for 10 min at room temperature followed by 1× SSC; 0.1% SDS for 10 min at 65°C. All blots were exposed for 24–72 h at −70°C to Kodak Biomax MS film and intensifying screen (Eastman Kodak, Rochester, NY, U.S.A.). The films were electronically scanned and the band densities were quantified using Scion Image software (Version 1.6, National Institutes of Health, Bethesda, MD, U.S.A.). To correct for variations in RNA loading of gels and transfer to membranes, all results were normalized to GAPDH mRNA, which we determined to be unaffected by O2 exposure.
Immunohistochemistry and autoradiography.
Animals were anesthetized with i.p. ketamine (80 mg/kg) and xylazine (20 mg/kg). The pulmonary circulation was flushed with PBS containing 1 U/mL heparin to clear the lungs of blood and fixed with 4% (wt/vol) paraformaldehyde by perfusion under a constant airway pressure of 10 cm H2O via a tracheal catheter. For autoradiography, animals received 1 μCi/g i.p. [3H]thymidine 2 h before lung fixation. Before immunohistochemical staining, lung sections were examined to confirm a normal lung structure from air-exposed control pups and the presence of patchy areas of interstitial thickening and emphysema in 60% O2-exposed pups (18). When lung sections were examined for the presence of ET-1, dilutions of the primary and secondary antisera were 1:650 and 1:520, respectively. When lung sections were examined for the presence of α-smooth muscle actin, dilutions of the primary and secondary antisera were 1:1000 and 1:300, respectively. Antibody specificity was verified by omitting the primary antiserum. After completion of immunohistochemical studies, using an avidin-biotin-peroxidase complex method (32), slides were lightly counterstained with Carazzi hematoxylin, dehydrated, cleared in xylene, and mounted. Slides were coated with Kodak NBT-3 emulsion for autoradiography and developed after 4 wk at 4°C for examination by light microscopy to identify dense areas of silver granule concentration over cell nuclei undergoing active DNA synthesis.
Lung volume-pressure loops.
Animals were anesthetized with sodium pentobarbitone (5–10 mg/kg) and paralyzed with pancuronium bromide (0.3 mg/kg). After tracheotomy, the lungs were degassed and volume-pressure loops in the closed chest recorded as previously described (18).
Total (free and esterified) lung 8-isoprostane measurement.
To prevent auto-oxidation, lung tissue was immediately flash frozen in liquid N2 at harvest and stored at −80°C until analysis. Upon thawing, the tissue sample was homogenized in 1× PBS with 0.005% (vol/vol) butylated hydroxytoluene, and 5000 cpm of [3H]PGF2α was added to quantify recovery during purification. Proteins were precipitated with ethanol and removed by centrifugation. The supernatant was incubated with an equal volume of 15% (wt/vol) potassium hydroxide at 40°C for 1 h for alkaline hydrolysis before solid-phase extraction using Sep Pak C18 cartridges. After purification, samples were analyzed in duplicate for 8-isoprostane content using a commercially available ELISA kit as previously described (33). Recovery from the purification step was analyzed by liquid scintillation counting of the extract and factored into the calculation along with correction for tissue protein content.
Effect of 8-isoprostane on prepro-ET-1 mRNA expression.
Primary mixed lung cell cultures were prepared from d 22 rat fetuses as previously described (34). Briefly, after removal of the heart, major vessels, and airways, the combined lungs were minced with scissors. The tissue was then subjected to sequential enzymatic dissociation, initially using 1 mg/mL trypsin and 0.01 mg/mL DNase. Cells were grown to confluence in Eagles' minimum essential medium with 10% (vol/vol) fetal bovine serum at a gas phase of 21% O2, 5% CO2, and 74% N2. 8-Isoprostane (10, 100, and 1000 nM) was added, and cultures were incubated in 21% O2 at 37°C in minimum essential medium without serum or antibiotics for 48 h. Cells were gently scraped from the culture flasks for RNA isolation and analysis as previously described.
Data presentation.
Unless otherwise stated, all values are for mean ± SEM of four litters. Statistical significance (p < 0.05) was determined by one-way ANOVA followed by assessment of differences using Duncan's multiple range test (35). Where error bars are not evident in figures, they fall within the plot point.
RESULTS
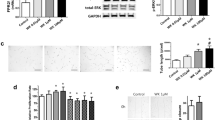
The effects of U74389G on survival and wet lung and body weights are shown in Table 1. Animals exposed to 60% O2 for 14 d had an approximately 10% mortality, which was greater, although not significantly so (p > 0.05), than that for air-exposed pups. Treatment with U74389G slightly reduced survival in 60% O2-exposed animals, such that the increase in mortality in 60% O2 became statistically significant (p < 0.05) when compared with U74389G-treated air controls. Animals in both air and O2 that had been treated with U74389G had significantly lower body weights and wet lung weights than vehicle-treated animals (p < 0.05). U74389G caused a marked reduction in lung cell DNA synthesis of air-exposed animals after 14 d, compared with vehicle-treated animals, when assessed by [3H]thymidine autoradiography (Fig. 1, A and C ). This finding is in keeping with previous observations using proliferating cell nuclear antigen immunohistochemistry (33) as an index of DNA synthesis. U74389G did not lead to an increase in lung cell DNA synthesis of 60% O2-exposed animals after 14 d, when assessed by [3H]thymidine autoradiography (Fig. 1D).
[3H]Thymidine autoradiography of lung tissue after exposure to air or 60% O2 for 14 d. Newborn rats received daily i.p. injections of 10 mg/kg U74389G in CS-4 vehicle or CS-4 vehicle alone. Dark- (A, B, C, and D) and bright- (E, F, G, and H) field microscopy is shown. Original magnification, ×200. A, E, air-exposed animals that received vehicle. B, F, 60% O2-exposed animals that received vehicle. Reduced DNA synthesis is evident compared with air-exposed animals. C, G, air-exposed animals that received U74389G. Markedly reduced DNA synthesis is evident compared with vehicle-treated animals. D, H, 60% O2-exposed animals that received U74389G. No recovery of DNA synthesis was observed in U74389G-treated animals.
As previously described (18), the lungs of rat pups exposed to 60% O2 for 14 d have areas of emphysema interspersed with patchy areas of parenchymal thickening and, consistent with these structural changes, abnormal lung mechanics. When compared with these previously reported changes (18), we could not detect any obvious effect of treatment with U74389G on either macroscopic structural changes or on lung mechanics (data not shown).
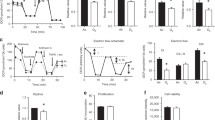
Lipid hydroperoxide formation in lung tissue, as assessed by measurement of lung 8-isoprostane content, was significantly increased (p < 0.05) after exposure to 60% O2 for 10 and 14 d (Fig. 2A). In contrast to gross structural changes, the increase in lipid peroxidation evident after a 14-d exposure to 60% O2 was completely attenuated (p < 0.05) by treatment with U74389G (Fig. 2B).
Total (free and esterified) 8-isoprostane in lung tissue. A, the effect of air (○) or 60% O2 (•) on total 8-isoprostane content in lungs from newborn rats. B, total 8-isoprostane content in lungs after exposure to air (open bars) or 60% O2 (filled bars) for 14 d. Newborn rats received daily i.p. injections of 10 mg/kg U74389G in CS-4 vehicle or CS-4 vehicle alone. Plot points and bars represent mean ± SEM for four litters. *p < 0.05, by one-way ANOVA, for animals exposed to 60% O2 compared with those in air in the same treatment group at d 14. #p < 0.05, by one-way ANOVA, for animals exposed to 60% O2 for 14 d and treated with U74389G in CS-4 vehicle, compared with those in 60% O2 and treated with CS-4 vehicle alone.
Results for whole lung prepro-ET-1 mRNA expression are shown in Figures 3 and 4, A and B . Prepro-ET-1 mRNA expression in air-exposed animals declined significantly after birth (p < 0.05) and remained low, as would be expected with the normal reduction in pulmonary vascular resistance that occurs after delivery. In contrast, animals exposed to 60% O2 had a significantly (p < 0.05) increased expression of prepro-ET-1 mRNA (similar to levels at birth), relative to air-exposed pups, on d 4, 7, and 10. By d 14 of exposure to 60% O2, expression of prepro-ET-1 mRNA had increased to values significantly greater than those present at birth (Fig. 3). Rat pups treated with U74389G for 14 d had no evidence of a 60% O2-mediated increase in prepro-ET-1 mRNA expression (Fig. 4, A and B ) compared with vehicle-treated and untreated animals. ET-1 protein content in lung tissue was assessed by immunohistochemistry (Fig. 5). When compared with air-exposed controls (Fig. 5A), vehicle-treated animals exposed to 60% O2 for 14 d (Fig. 5B) had markedly increased ET-1 immunoreactivity localized to bronchial epithelium and interstitial cells in the lung parenchyma. Animals treated with U74389G did not have this 60% O2-mediated increase in ET-1 immunoreactivity (Fig. 5, C and D ), in keeping with the effect of U74389G on expression of prepro-ET-1 mRNA.
The effect of air (○) or 60% O2 (•) on whole lung mRNA for prepro-ET-1 in newborn rats, as assessed by Northern analysis. All data points represent the mean ± SEM for four litters. Where error bars are not evident, they fall within the plot point. *p < 0.05, by one-way ANOVA, for 60% O2-exposed animals compared with air controls at the same time. #p < 0.05, by one-way ANOVA, for air-exposed animals compared with all other groups. **p < 0.05, by one-way ANOVA, for 60% O2-exposed animals at d 14 compared to d 7 and d 10.
Effect of U74389G on total lung prepro-ET-1 mRNA. A, whole lung prepro-ET-1 mRNA after exposure to air (open bars) or 60% O2 (filled bars) for 14 d. Newborn rats received daily i.p. injections of 10 mg/kg U74389G in CS-4 vehicle (U74389G), CS-4 vehicle alone (VEHICLE), or no intervention (UNTREATED). Bars represent mean ± SEM for four litters. *p < 0.05, by one-way ANOVA, for animals exposed to 60% O2 compared with those in air in the same treatment group. #p < 0.05, by one-way ANOVA, for U74389G-treated animals compared with untreated or vehicle-treated animals exposed to 60% O2. B, Northern blot analysis of whole lung prepro-ET-1 mRNA at birth (DAY 0) and after exposure to air (A) or 60% O2 (O) for 14 d. Newborn rats received either no injections (UNTREATED) or daily i.p. injections of either 10 mg/kg U74389G (U74389G) or vehicle alone (VEHICLE). To correct for differences in loading of RNA and transfer to membranes, all results were normalized to GAPDH mRNA. Transcript sizes are in kilobases.
Immunohistochemistry for ET-1 in lung tissue after exposure to air or 60% O2 for 14 d. Newborn rats received daily i.p. injections of 10 mg/kg U74389G in CS-4 vehicle or CS-4 vehicle alone. Original magnification ×200. A, air-exposed pups that received vehicle. The arrow indicates immunoreactive staining of bronchial epithelium (e). B, 60% O2-exposed pups that received vehicle. Markedly increased immunoreactive staining is evident in bronchial epithelial (e) and parenchymal interstitial (i) cells. C, air-exposed pups that received U74389G. D, 60% O2-exposed pups that received U74389G. Increased immunoreactive staining is not evident in O2-exposed U74389G-treated animals.
Vascular smooth muscle in lung tissue was assessed by α-smooth muscle actin immunohistochemistry (Fig. 6). Vehicle-treated 60% O2-exposed animals (Fig. 6, B and D ) showed marked arterial smooth muscle hypertrophy, and endothelial cell loss, compared with air-exposed controls (Fig. 6, A and C ). Animals treated with U74389G did not have evidence of O2-induced smooth muscle hypertrophy (Fig. 6F). Consistent with these smooth muscle changes, there was significant RVH (Fig. 7), as assessed by the ratio of the right ventricle to the left ventricle and septum dry weights, in 60% O2-exposed vehicle-treated animals at d 14 compared with the similarly treated animals in air. This 60% O2-induced RVH, an index of pulmonary hypertension, was completely attenuated (p < 0.05) in animals treated with U74389G.
Effect of U74389G on 60% O2-mediated pulmonary vascular smooth muscle hypertrophy. Immunohistochemistry for α-smooth muscle actin in vessels (v) and airways (a) after exposure to air or 60% O2 for 14 d. Newborn rats received daily i.p. injections of 10 mg/kg U74389G in CS-4 vehicle or CS-4 vehicle alone. Original magnification ×200 (A, B, E, and F) or ×800 (C and D). A, C, air-exposed pups that received vehicle. B, 60% O2-exposed pups that received vehicle. D, 60% O2-exposed animals that received vehicle. Marked vascular smooth muscle hypertrophy and endothelial cell loss is seen. E, air-exposed pups that received U74389G. F, 60% O2-exposed pups that received U74389G. O2-mediated vascular smooth muscle hypertrophy is not evident in U74389G-treated animals.
Right ventricular (RV) dry weight compared with that of left ventricle and septum (LV+S), as an index of RVH, after exposure to air or 60% O2 for 14 d. Newborn rats received daily i.p. injections of 10 mg/kg U74389G in CS-4 vehicle (U74389G) or CS-4 vehicle alone (Vehicle). Plot points represent mean ± SEM for 12 pups from a single litter. *p < 0.05, by one-way ANOVA, for control animals exposed to 60% O2 compared with those in air. #p < 0.05, by one-way ANOVA, for U74389G-treated animals compared with control animals exposed to 60% O2.
To determine whether there could be a direct link between the products of 60% O2-induced lipid peroxidation and the expression of ET-1 after birth, we examined the effect of 8-isoprostane on fetal lung cell prepro-ET-1 mRNA expression. Compared with controls, 8-isoprostane (10, 100, and 1000 nM) caused a significant (p < 0.05) increase in prepro-ET-1 mRNA expression in d 22 rat fetal lung cells (Fig. 8).
Effect of 8-isoprostane on prepro-ET-1 mRNA expression in primary cultures of d 22 mixed rat fetal lung cells. Plot points represent mean ± SEM for three separate experiments. *p < 0.05, by one-way ANOVA, compared with controls. Inset, Northern blot analysis of prepro-ET-1 mRNA expression in control (C) cells and cells exposed to 10 nM 8-isoprostane (8-iso). Transcript size is in kilobases.
DISCUSSION
The ontogeny of ET-1 during fetal and early postnatal life is consistent with it playing an important role in maintaining the normally high fetal pulmonary vascular resistance, which decreases after adaptation of the pulmonary circulation to extrauterine life (36–38). In accordance with our observations in the newborn rat, abnormally elevated levels of ET-1 in plasma and tracheobronchial aspirates of the human newborn have been correlated with persistent pulmonary hypertension and with pulmonary hypertension associated with respiratory distress and BPD (39–43).
ET-1 elicits a number of biologic effects in the lung, including pulmonary vascular and bronchial smooth muscle contraction and proliferation (39, 44), in addition to fibroblast proliferation and chemotaxis (45). Endothelin receptor antagonists have been effective in the prevention, or reversal, of pulmonary hypertension in laboratory animals induced by hypoxia (46, 47) and monocrotaline injection (48). These data are consistent with ET-1 being a critical mediator of the pulmonary vascular pathology of BPD, which includes vasoconstriction, vascular remodeling, and RVH. ET-1 may also contribute to the pathologic changes observed in lung injury through inhibition of apoptosis (49) or enhancement of pulmonary fibrosis (50).
As we have also recently reported elsewhere (33), antioxidants can cause inhibition of lung cell DNA synthesis in air-exposed neonatal rats. Our findings with 60% O2-exposed animals differed somewhat from previous observations with animals exposed to 95% O2 for 7 d, assessed using proliferating cell nuclear antigen immunohistochemistry (33), as well as [3H]thymidine autoradiography (unpublished observations), in which some protection against O2-mediated inhibition of DNA synthesis was observed. This may reflect the differing exposure times, or the very different lung injuries, observed with these exposure protocols. The inhibition of lung cell DNA synthesis observed in air-exposed animals treated with U74389G presumably reflects the critical physiologic role played by reactive O2 species in cell signaling and growth (51). Our current finding that U74389G-treated animals also had attenuated somatic and lung growth is of concern, although a nutritional effect cannot be ruled out. If antioxidant therapies inhibit lung growth, already an adverse consequence of BPD (52), targeted approaches to specific aspects of O2-induced lung injury may be preferable to an antioxidant approach for the newborn.
Our findings of a marked effect on ET-1 expression by hyperoxia, and the efficacy of U74389G in its prevention, imply a novel mechanism for ET-1 regulation. We have previously reported that U74389G does not scavenge hydrogen peroxide, and is ineffective at preventing O2-induced cytotoxicity mediated by hydrogen peroxide in distal fetal rat lung epithelial cells (24). These findings indicate that pulmonary hypertension and ET-1 up-regulation are caused, directly or indirectly, by reactive O2 species other than hydrogen peroxide during prolonged exposure of neonatal rats to 60% O2. The extensive increase in immunoreactive ET-1, and its immunolocalization, in the lungs of pups exposed to 60% O2 is consistent with the findings reported for adult rats subjected to another reactive O2 species-mediated lung injury, bleomycin-induced pulmonary fibrosis (53).
Peroxidation of membrane phospholipids by reactive O2 species leads to the production of lipid hydroperoxides (54) and stable, diffusible degradation products such as aldehydes (55) and isoprostanes (22). These products are themselves biologically active, and appear to act as second messengers for a wide range of effects induced by reactive O2 species. Among these, the F2-isoprostanes are a recently described group of prostaglandin F2α isomers formed by reactive O2 species-mediated peroxidation of arachidonic acid, independent of the action of cyclooxygenase. The best-characterized isoprostane produced in vivo is 8-isoprostane, a stable compound found in all biologic fluids that is increased by oxidative stress and antioxidant deficiency (22). 8-Isoprostane is highly vasoactive, causing vasoconstriction in the kidney (56) and retina (57) and increased microvascular permeability in the lung (58).
The bioactive properties of 8-isoprostane, and its marked increase in neonatal rat lung tissue after exposure to 60% O2, suggested to us that it may also contribute directly to the O2-mediated pulmonary hypertension observed in this model. We further postulated that the preventive effect of U74389G on 60% O2-induced ET-1 up-regulation was through inhibition of 8-isoprostane formation. In support of this postulate was the preventive effect of U74389G on lung 8-isoprostane content in vivo and the striking in vitro effect of 8-isoprostane on prepro-ET-1 mRNA production. Recent in vitro evidence, in bovine aortic endothelial cells and the kidney, has also suggested that the biologic activity of 8-isoprostane may be attributable in part to up-regulation of ET-1 (56, 59, 60). The above findings are also consistent with data from Moore et al. (61), who noted that plasma ET-1 levels are markedly increased in the hepatorenal syndrome, a condition in which 8-isoprostane is most likely the causative mediator (62). Our observations are the first, to our knowledge, to link an O2-mediated induction of 8-isoprostane with up-regulation of ET-1 and pulmonary hypertension.
The biologic effects of 8-isoprostane are probably mediated by its interaction with an uncharacterized unique receptor similar to, but distinct from, the thromboxane A2 receptor (56). Work by Lahaie and colleagues (57) has shown that the thromboxane receptor antagonist L670596 attenuated the vasoconstrictor action of 8-isoprostane on retinal vessels, possibly owing to blockade of 8-isoprostane receptors. The dominant role of the thromboxane receptor on 8-isoprostane-induced retinal vasoconstriction reported in this study may also be explained by the observation that ET-1, which is up-regulated by 8-isoprostane, causes phospholipase A2 activation and subsequent thromboxane release through stimulation of the ETB receptor (63, 64). If the same pathways are active in the O2-injured lung, thromboxane receptor antagonists may also be effective in the prevention of O2-induced pulmonary hypertension.
The observations reported above have implications for the development of interventions designed to limit O2-mediated lung injury. First, the observation that a single antioxidant therapy can prevent the pulmonary vascular effects of O2 toxicity in newborn rats, without correcting dysregulated growth of lung interstitial cells, suggests that these outcomes are regulated by different reactive O2 species. Any antioxidant, or combination of antioxidants, intended to completely prevent O2-mediated lung injury will need to have a broad spectrum of activities. Second, the recognition of a reversible up-regulation of ET-1 in O2-mediated pulmonary hypertension suggests that alternative therapeutic approaches, such as endothelin or thromboxane receptor antagonists, may be effective.
Abbreviations
- BPD:
-
bronchopulmonary dysplasia
- ET-1:
-
endothelin-1
- GAPDH:
-
glyceraldehyde 3-phosphate dehydrogenase
- RVH:
-
right ventricular hypertrophy
References
Northway WH Jr, Rosan RC, Porter DY 1967 Pulmonary disease following respirator therapy of hyaline membrane disease. N Engl J Med 276: 357–368.
Bonikos DS, Bensch KG, Northway WH, Edwards DK 1976 Bronchopulmonary dysplasia: the pulmonary pathological sequel of necrotizing bronchiolitis and pulmonary fibrosis. Human Pathol 7: 643–666.
Jacob SV, Coates AL, Lands LC, MacNeish CF, Riley SP, Hornby L, Outerbridge EW, Davis GM, Williams RL 1998 Long-term pulmonary sequelae of severe bronchopulmonary dysplasia. J Pediatr 133: 193–200.
Saugstad OD 1998 Oxygen radical disease in neonatology. Semin Neonatol 3: 229–238.
Gill AB, Weindling AM 1995 Raised pulmonary artery pressure in very low birthweight infants requiring supplemental oxygen at 36 weeks after conception. Arch Dis Child 72: F20
Subhedar NV, Hamdan AH, Ryan SW, Shaw NJ 1998 Pulmonary artery pressure: early predictor of chronic lung disease in preterm infants. Arch Dis Child 78: F20
Goodman G, Perkin RM, Anas NG, Sperling DR, Hicks DA, Rowen M 1988 Pulmonary hypertension in infants with bronchopulmonary dysplasia. J Pediatr 112: 67–72.
Giaid A, Yanagisawa M, Langleben D, Michel RP, Levy R, Shennib H, Kimura S, Masaki T, Duguid WP, Stewart DJ 1993 Expression of endothelin-1 in the lungs of patients with pulmonary hypertension. N Engl J Med 328: 1732–1739.
Yanagisawa M, Kurihara H, Kimura S, Tomobe Y, Kobayashi M, Mitsui Y, Yazaki Y, Goto K, Masaki T 1988 A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature 332: 411–415.
Mattoli S, Mezzetti M, Riva G, Allegra L, Fasoli A 1992 Specific binding of endothelin on human bronchial smooth muscle cells in culture and secretion of endothelin-like material from bronchial epithelial cells. Am J Respir Cell Mol Biol 3: 145–151.
Soma S, Takahashi H, Muramatsu M, Oka M, Fukuchi Y 1999 Localization and distribution of endothelin receptor subtypes in pulmonary vasculature of normal and hypoxia-exposed rats. Am J Respir Cell Mol Biol 20: 620–630.
Goldie RG, D'Aprile AC, Self GJ, Rigby PJ, Henry PJ 1996 The distribution and density of receptor subtypes for endothelin-1 in peripheral lung of the rat, guinea-pig and pig. Br J Pharmacol 117: 729–735.
Hagiwara H, Nagasawa T, Yamamoto T, Lodhi KM, Ito T, Takemura N, Hirose S 1993 Immunochemical characterization and localization of endothelin ETB receptor. Am J Physiol 264: R777
Elton TS, Oparil S, Taylor GR, Hicks PH, Yang R-H, Jin H, Chen YF 1992 Normobaric hypoxia stimulates endothelin-1 gene expression in the rat. Am J Physiol 263: R1260
Li H, Chen S-J, Chen Y-F, Meng QC, Durand J, Oparil S, Elton TS 1994 Enhanced endothelin-1 and endothelin receptor gene expression in chronic hypoxia. J Appl Physiol 77: 1451–1459.
Kanse SM, Takahashi K, Lam HC, Rees A, Warren JB, Porta M, Molinatti P, Ghatei M, Bloom SR 1991 Cytokine stimulated endothelin release from endothelial cells. Life Sci 48: 1379–1384.
Harrison VJ, Ziegler T, Bouzourene K, Suciu A, Silacci P, Hayoz D 1998 Endothelin-1 and endothelin-converting enzyme-1 gene regulation by shear stress and flow-induced pressure. J Cardiovasc Pharmacol 31: S38
Han RNN, Buch S, Tseu I, Young J, Christie NA, Frndova H, Lye SJ, Post M, Tanswell AK 1996 Changes in structure, mechanics, and insulin-like growth factor-related gene expression in the lungs of newborn rats exposed to air or 60% oxygen. Pediatr Res 39: 921–929.
Koppel R, Han RNN, Cox D, Tanswell AK, Rabinovitch M 1994 α1-Antitrypsin protects neonatal rats from pulmonary vascular and parenchymal effects of oxygen toxicity. Pediatr Res 36: 763–770.
Saugstad OD 1997 Bronchopulmonary dysplasia and oxidative stress: are we closer to an understanding of the pathogenesis of BPD?. Acta Paediatr 86: 1277
Freeman BA, Topolosky MK, Crapo JD 1982 Hyperoxia increases oxygen radical production in rat lung homogenates. Arch Biochem Biophys 216: 477–484.
Roberts LJ II, Morrow JD 1997 The generation and actions of isoprostanes. Biochim Biophys Acta 1345: 121–135.
Hall ED, McCall JM, Means ED 1994 Therapeutic potential of the lazaroids (21-aminosteroids) in acute central nervous system trauma, ischemia and subarachnoid hemorrhage. Adv Pharmacol 28: 221–268.
Luo XP, Christie NA, McLaughlin MA, Belcastro R, Sedlackova L, Cabacungan J, Freeman BA, Tanswell AK 1999 H2O2 mediates O2 toxicity in cultured fetal rat distal lung epithelial cells. Free Radic Biol Med 26: 1357–1368.
Khalil A, Fortun A, Hebert S, Jay-Gerin J, El Abbouyi A, Wallach J, Fulop T Jr 1998 Novel 21-aminosteroid U-74389G inhibits low-density lipoprotein peroxidation induced by ·OH, and O2·-free radicals. Life Sci 63: 769–779.
Braughler JM, Burton PS, Pregenzer JF, Jacobsen EJ, VanDoornik FJ, Tustin JM, Ayer DE, Bundy GL 1988 Novel membrane localized iron chelators as inhibitors of iron-dependent lipid peroxidation. Biochem Pharmacol 37: 3853–3860.
Hall ED, McCall JM 1994 Antioxidant actions of lazaroids. Methods Enzymol 234: 548–555.
Frank L, McLaughlin GE 1993 Protection against acute and chronic hyperoxic inhibition of neonatal rat lung development with the 21-aminosteroid drug U74389F. Pediatr Res 33: 632–638.
Terada Y, Tomita K, Nonoguchi H, Yang T, Marumo F 1993 Expression of endothelin-3 mRNA along rat nephron segments using polymerase chain reaction. Kidney Int 44: 1273–1280.
Fulton RM, Hutchinson EC 1952 Ventricular weight in cardiac hypertrophy. Br Heart J 14: 413–420.
Chomsczynski P, Sacchi N 1987 Single-step method of RNA isolation by acid-guanidium-thiocyanate-phenol-chloroform extraction. Anal Biochem 162: 156–159.
Hsu SM, Raine L, Fanger H 1981 Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabelled antibody (PAP) procedures. J Histochem Cytochem 29: 577–580.
Luo XP, Sedlackova L, Belcastro R, Cabacungan J, Lye SJ, Tanswell AK 1999 Effect of the 21-aminosteroid U74389G on oxygen-induced free radical production, lipid peroxidation and inhibition of lung growth in neonatal rats. Pediatr Res 46: 215–223.
Tanswell AK, Tzaki MG, Byrne PJ 1986 Hormonal and local factors influence antioxidant enzyme activity of rat fetal lung cells in vitro. Exp Lung Res 11: 49–59.
Snedecor GW, Cochran WG 1980 Statistical Methods. Iowa State University Press, Ames, IA, 215–295.
Giaid A, Polak JM, Gaitonde V, Hamid QA, Moscosco G, Legon S, Uwanogho D, Roncalli M, Shinmi O, Sawamura T, Kimura S, Yanagisawa M, Masaki T, Springall DR 1991 Distribution of endothelin-like immunoreactivity and mRNA in the developing and adult human lung. Am J Respir Cell Mol Biol 4: 50–58.
Endo A, Ayasuwa M, Minato M, Takada M, Takahashi S, Harada K 1997 Endothelium-derived relaxing and contracting factors during the early neonatal period. Acta Paediatr 86: 834–836.
Haegerstrand A, Hemsén A, Gillis C, Larsson O, Lundberg JM 1989 Endothelin: presence in human umbilical vessels, high levels in fetal blood and potent constrictor effect. Acta Physiol Scand 137: 541–542.
Barnes PJ 1994 Endothelins and pulmonary diseases. J Appl Physiol 77: 1051–1059.
Niu JO, Munshi UK, Siddiq MM, Parton LA 1998 Early increase in endothelin-1 in tracheal aspirates of preterm infants: correlation with bronchopulmonary dysplasia. J Pediatr 132: 965–970.
Allen SW, Chatfield BA, Koppenhafer SA, Schaffer MS, Wolfe RR, Abman SH 1993 Circulating immunoreactive endothelin-1 in children with pulmonary hypertension: association with acute hypoxic pulmonary vasoconstriction. Am Rev Respir Dis 148: 519–522.
Kojima T, Isozaki-Fukuda Y, Takedatsu M, Ono A, Hirata Y, Kobayashi Y 1992 Plasma endothelin-like immunoreactivity levels in neonates. Eur J Pediatr 151: 913–915.
Christou H, Adatia I, Van Marter LJ, Kane JW, Thompson JE, Stark AR, Wessel DL, Kourembanas S 1997 Effect of inhaled nitric oxide on endothelin-1 and cyclic guanosine 5′-monophosphate plasma concentrations in newborn infants with persistent pulmonary hypertension. J Pediatr 130: 603–611.
Zamora MA, Dempsey EC, Walchak SJ, Stelzner TJ 1993 BQ123, an ETA receptor antagonist, inhibits endothelin-1-mediated proliferation of human pulmonary artery smooth muscle cells. Am J Respir Cell Mol Biol 9: 429–433.
Peacock AJ, Dawes KE, Shock A, Gray AJ, Reeves JT, Laurent GJ 1992 Endothelin-1 and endothelin-3 induce chemotaxis and replication of pulmonary artery fibroblasts. Am J Respir Cell Mol Biol 7: 492–499.
Underwood DC, Bochnowicz S, Osborn RR, Louden CS, Hart TK, Hay DWP 1998 Chronic hypoxia-induced cardiopulmonary changes in three rat strains: inhibition by the ET receptor antagonist SB 217242. J Cardiovasc Pharm 31: S453
Oparil S, Chen S-J, Meng QC, Elton TS, Yano M, Chen Y-F 1995 Endothelin-A receptor antagonist prevents acute hypoxia-induced pulmonary hypertension in the rat. Am J Physiol 268: L95
Miyauchi T, Yorikane R, Sakai S, Sakurai T, Okada M, Nishikibe M, Yano M, Yamaguchi I, Sugishita Y, Goto K 1993 Contribution of endogenous ET-1 to the progression of cardio-pulmonary alterations in rats with monocrotaline-induced pulmonary hypertension. Circ Res 73: 887–897.
Wu-Wong JR, Chiou WJ, Dickinson R, Opgenorth TJ 1997 Endothelin attenuates apoptosis in human smooth muscle cells. Biochem J 328: 733–737.
Giaid A, Michel RP, Stewart DJ, Sheppard M, Corrin B, Hamid Q 1993 Expression of endothelin-1 in lungs of patients with cryptogenic fibrosing alveolitis. Lancet 341: 1550–1554.
Suzuki YJ, Forman HJ, Sevanian A 1997 Oxidants as stimulators of signal transduction. Free Radic Biol Med 22: 269–285.
Margraf LF, Tomashefski JF, Bruce MC, Dahms BB 1991 Morphometric analysis of the lung in bronchopulmonary dysplasia. Am Rev Respir Dis 143: 391–400.
Mutsaers SE, Foster ML, Chambers RC, Laurent GJ, McAnulty RJ 1998 Increased endothelin-1 and its localization during the development of bleomycin-induced pulmonary fibrosis in rats. Am J Respir Cell Mol Biol 16: 611–619.
Porter NA, Caldwell SE, Mills KA 1995 Mechanism of free radical oxidation of unsaturated lipid. Lipids 30: 277–290.
Esterbauer H, Schauer RJ, Scholler H 1991 Chemistry and biochemistry of 4-hydroxynonenal, malondialdehyde and related aldehydes. Free Radic Biol Med 11: 81–128.
Takahashi K, Nammour TM, Fukunaga M, Ebert J, Morrow JD, Roberts LJ II, Hoover RL Badr KF 1992 Glomerular actions of the free radical-generated novel prostaglandin, 8-epi-prostaglandin F2α, in the rat: evidence for interaction with thromboxane A2 receptors. J Clin Invest 90: 136–141.
Lahaie I, Hardy P, Hou X, Hasséssian H, Asselin P, Lachapelle P, Almazan G, Varma DR, Morrow JD, Roberts LJ II, Chemtob S 1998 A novel mechanism for vasoconstrictor action of 8-isoprostaglandin F2α on retinal vessels. Am J Physiol 274: R1406
Vacchiano CA, Tempel GE 1994 Role of nonenzymatically generated prostanoid, 8-iso-PGF2α, in pulmonary oxygen toxicity. J Appl Physiol 77: 2912–2917.
Fukunaga F, Yura T, Badr KF 1995 Stimulatory effect of 8-epi-prostaglandin F2α, an F2-isoprostane, on endothelin-1 release. J Cardiovasc Pharmacol 26: S51
Yura T, Fukunaga M, Khan R, Nassar GN, Badr KF, Montero A 1999 Free-radical-generated F2-isoprostane stimulates cell proliferation and endothelin-1 expression on endothelial cells. Kidney Int 56: 471–478.
Moore K, Wendon J, Frazer M, Karani J, Williams R, Badr KF 1992 Plasma endothelin immunoreactivity in liver disease and the hepatorenal syndrome. N Engl J Med 327: 1774–1778.
Morrow JD, Moore KP, Awad JA, Ravenscraft MD, Marini G, Badr KF, Williams R, Roberts LJ II 1993 Marked overproduction of non-cyclooxygenase derived prostanoids (F2-isoprostanes) in the hepatorenal syndrome. J Lipid Med 6: 417–420.
D'Orleans-Juste P, Claing A, Telemaque S, Maurice M-C, Yano M, Gratton J-P 1994 Block of endothelin-1-induced release of thromboxane A2 from the guinea pig lung and nitric oxide from the rabbit kidney by the selective ETB antagonist, BQ-788. Br J Pharmacol 113: 1257–1262.
Reynolds EE, Mok LLS 1990 Role of thromboxane A2/prostaglandin H2 receptor in the vasoconstrictor response of rat aorta to endothelin. J Pharmacol Exp Ther 252: 915–921.
Author information
Authors and Affiliations
Additional information
Supported by Group grants from the Medical Research Council of Canada, and an equipment grant from the Ontario Thoracic Society. Dr. Tanswell holds the Women's Auxiliary Chair in Neonatal Medicine. Dr. Jankov is supported by a RESTRACOM Fellowship from the Hospital for Sick Children Research Institute.
Rights and permissions
About this article
Cite this article
Jankov, R., Luo, X., Cabacungan, J. et al. Endothelin-1 and O2-Mediated Pulmonary Hypertension in Neonatal Rats: A Role for Products of Lipid Peroxidation. Pediatr Res 48, 289–298 (2000). https://doi.org/10.1203/00006450-200009000-00005
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-200009000-00005
This article is cited by
-
Isoprostanes as physiological mediators of transition to newborn life: novel mechanisms regulating patency of the term and preterm ductus arteriosus
Pediatric Research (2012)
-
Today’s and tomorrow’s imaging and circulating biomarkers for pulmonary arterial hypertension
Cellular and Molecular Life Sciences (2012)