Abstract
In this study we report changes in HDL concentration and composition in acute and convalescent Kawasaki disease. Notable reductions in plasma HDL-cholesterol (0.54 ± 0.2 mmol/L, normal level 0.7-1.81 mmol/L) and apolipoprotein A-I (apoA-I) (56 ± 28 mg/dL, normal level 141 ± 22 mg/dL) were observed in all 24 patients studied during the acute phase of Kawasaki disease. These changes were accompanied by the marked appearance of serum amyloid A (SAA) protein in the plasma, associated with HDL3-like lipoprotein particles. The distribution of apoA-I was analyzed in five patients and showed a significant increase in lipid-free apoA-I in the bottom fraction (28.8 ± 4.1%, normal range 10-15%), suggesting displacement of apoA-I from the HDL particles by SAA. Within 2 wk after acute Kawasaki disease, levels of HDL-cholesterol and apoA-I returned to the normal range, and SAA disappeared from the plasma. The HDL of patients with Kawasaki disease was markedly enriched in triglyceride even in the absence of changes in total plasma triglyceride. The core composition of HDL returned to the normal range more slowly than the plasma HDL-cholesterol and apoA-I levels. This suggests that Kawasaki disease has a profound effect on the lipoprotein profile acutely and a more subtle sustained effect on the HDL composition. We interpret these changes as manifestations of the acute phase response in Kawasaki disease.
Similar content being viewed by others
Main
Kawasaki disease is an acute febrile illness of early childhood characterized by multiple clinical and biochemical features of inflammation and histopathologic features of vasculitis. The etiologic agent(s) of Kawasaki disease remains unknown(1). Coronary artery aneurysms are common complications of the disease(1).
Changes in lipoproteins have been demonstrated in several acute inflammatory conditions including Kawasaki disease. Prominent among these changes are decreases in HDL cholesterol and apoA-I(2–5). There is little information in humans on the mechanism of these changes. Animal studies suggest that, after induction of the acute phase response by various stresses, such as injection of endotoxin or other agents including severe physical restraint and trauma, SAA is released from the liver and incorporated into HDL particles. There is a concomitant decline in plasma HDL concentration(6–8). Similar responses have been seen in humans with acute myocardial infarction(9), trauma(10), and infection(11, 12). Recent data have suggested that the decline in HDL in the acute phase of inflammation(8) and other conditions(13) may be either a consequence of, or be accentuated by, the triglyceride enrichment of HDL.
The purpose of this study was to characterize changes in plasma HDL and apoA-I concentrations in children with acute Kawasaki disease, to determine the association of these changes with the plasma SAA content, to compare these changes in Kawasaki disease with those in other acute illnesses of children, and to assess prospectively changes in HDL composition throughout the course of Kawasaki disease.
METHODS
This study was conducted on three cohorts, studied consecutively (see Table 1). Cohort 1 represented a group of 12 children with Kawasaki disease obtained at four time points (before therapy, and at 1, 2, and 12 mo follow-up) and studied retrospectively using frozen sera. Cohort 2 was a comparison group with 17 children presenting with other acute illnesses suspected to be acute infections and also studied retrospectively using frozen sera. Cohort 3 was a group of 12 additional children with Kawasaki disease studied prospectively for lipoprotein composition and SAA. Institutional IRB approval was obtained for the study.
Cohorts. In cohort 1, 12 children with documented Kawasaki disease diagnosed by established criteria and with frozen sera obtained before therapy, and at 1, 2, and 12 mo follow-up were studied(14). Six had developed coronary aneurysms as sequelae of the disease and six had not. There were nine boys and three girls; mean age was 4 y (range 7 mo to 15 y). They presented between the 4th and the 12th illness days.
In cohort 2, 17 children with fever >39 °C for at least 1 d (median duration of fever = 2 d) secondary to a documented acute infection were recruited from the Children's Memorial Hospital Emergency Room. These children had excess sera frozen obtained during routine laboratory studies for work-up of their acute illnesses. These included 10 boys and seven girls with a mean age of 2.5 y (range, 9 mo to 4 y). Acute illnesses were gastroenteritis in six, pneumonia in five, urinary tract infection in two, fever without a source in two, and aseptic meningitis in one.
In the prospective study of lipoprotein composition (cohort 3), 12 children with Kawasaki disease had fresh plasma obtained before therapy and seven of these had plasma obtained at follow-up 2-6 wk later. These included eight boys and four girls with a mean age of 4 y (range 1-19 y). Eleven children presented on illness on d 4-11, whereas one child presented on illness on d 36.
Laboratory methods. Retrospective studies used sera previously collected and stored frozen at -20 °C at the Childrens' Memorial Hospital laboratories. The frozen sera were transported on dry ice to the University of Chicago for analyses of HDL cholesterol, apoA-I, and SAA. For the prospective study, plasma samples were collected into tubes containing a preservative “cocktail” (phenylsulfonyl fluoride, 0.001 M in methanol, 0.5 μg of gentamicin sulfate, 50 μg of NaN3, and 1 μg of chloramphenicol per mL of plasma) and stored at 4 °C. The samples were transported on wet ice to the University of Chicago for analyses of triglycerides, cholesterol, apoA-I, and SAA in whole plasma and in lipoprotein subfractions obtained by density gradient centrifugal flotation.
Triglycerides and cholesterol were analyzed using commercially purchased enzymatic kits (Lancer). HDL cholesterol was determined in plasma after precipitation of apolipoprotein B-containing lipoproteins by magnesium chloride and phosphotungstic acid (Lancer). ApoA-I was quantitated by radial immunodiffusion as described previously(15) using polyclonal antibody to human apoA-I. Standardized sera obtained from the Northwest Lipid Research Laboratory (Seattle, WA) were used as reference. Core HDL triglyceride (TG) was determined as the percent of core lipids of HDL [TG/(TG + CE (cholesteryl ester))], assayed in HDL fractions. The HDL subfractions were obtained by an equilibrium density gradient ultracentrifugal flotation(7). In this procedure, 2 mL of plasma were layered at the interface of a 3-20% NaBr gradient. Centrifugation was conducted at 15 °C, 38 000 rpm, 66 h, using a SW 41Ti rotor. The lipoproteins band at their respective equilibrium densities. Density of the fractions were determined based on the density of a reference solution. Fractions collected using a density gradient fractionator with a UV detector (ISCO, Lincoln, NE) were dialyzed (0.01 M Tris, 0.01% EDTA, pH 8.0, with anti-proteases and bacterial inhibitors) and used for further analyses within 2 wk.
SAA in plasma or lipoprotein subfractions was detected as a 12 000-kD band by immunoblotting of proteins transferred from SDS-PAGE. One microliter of plasma or 10 μL of fractions obtained by density gradient centrifugation were applied to each lane of a 10-20% gradient SDS-PAGE. Electrophoresis was conducted at 40 mA/gel using a Mighty Small gel apparatus (Hoeffer Scientific Instruments) and based on the procedure of Weber and Osborne(16). The proteins were electrotransferred to membranes (Immubilon, Millipore) following the procedure of Towbin et al.(17) using a Transphor electrophoresis unit (Hoeffer Scientific Instruments) for 1 h at 360 mA/gel and water-cooled at 10 °C with a circulating bath (Forma Scientific). The specific protein bands were detected using antibody against human SAA obtained from commercial sources (Calbiochem) or our recently produced anti-human SAA, prepared by immunization with recombinant human SAA (our unpublished data), and visualized with a chemiluminescence detection system (ECL, Amersham Corp.) following the protocol outlined by the manufacturer.
Data analysis. For the retrospective study (phase 1), comparisons were made between those patients with and without aneurysms. Then both groups were combined to assess the longitudinal changes in the presence of SAA, HDL cholesterol, and apoA-I concentrations. Follow-up and baseline values were compared by paired t tests. Unpaired t tests were performed to compare the baseline values for children with Kawasaki disease with those with other acute infections. For the prospective studies, descriptive statistics and paired t tests were performed for comparison of acute and follow-up data.
RESULTS
In the first part of the study involving frozen sera, all 12 children studied had abundant SAA present when they initially presented with acute Kawasaki disease. One month later, only 1/12 had detectable SAA in the sera. SAA was not detected in any of the follow-up sera. There were no significant differences in HDL cholesterol or apoA-I concentrations between those with and those without aneurysms (data not shown), although statistical power to detect such a difference was small.
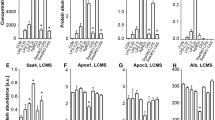
The sequential changes in HDL cholesterol and apoA-I at the four measurement points are shown in Figures 1 and 2. Both HDL cholesterol and apoA-I were significantly below the normal range during the acute illness and increased over time. All children had values ≥0.9 mmol/L at final follow-up. The child with SAA detectable at 1 mo had the lowest HDL cholesterol and apoA-I levels at that time.
Changes in HDL cholesterol levels. HDL cholesterol was assessed in frozen plasma obtained from children with Kawasaki disease at presentation and follow-up visits. Values are expressed as mean ± SD, p < 0.001 admission vs later periods. Normal range = 0.78-1.81 mmol/L(23).
HDL cholesterol and apoA-I levels of children with Kawasaki disease were compared with children with other acute febrile illnesses. In 15/17 such children SAA was present at clinical presentation (Table 1). However, their serum HDL cholesterol and apoA-I levels were significantly higher than those with acute Kawasaki disease [HDL cholesterol: 0.88 ± 0.47 versus 0.47 ± 0.23 mmol/L (p < 0.01); apoA-I: 89.7 ± 28 versus 61.6 ± 19.6 (p < 0.01)]. These comparisons were also significant when the two with acute infections who lacked SAA were excluded from the analyses (p < 0.05). The much larger range for HDL cholesterol and apoA-I levels among those with acute infections suggests that the lipoprotein response to acute inflammation varies considerably.
In cohort 3, prospective assessment of lipid and apolipoprotein values in children with Kawasaki disease was undertaken using freshly drawn, nonfrozen plasma. Table 2 shows the results for assessment of total cholesterol, triglycerides, HDL cholesterol, core triglyceride in HDL, and apoA-I at acute presentation. Total cholesterol and triglycerides were within normal limits. However, the HDL-cholesterol (0.54 ± 0.21 mmol/L) and apoA-I (56 ± 27 mg/dl) values appeared to be notably below the normal levels (0.78-1.81 mmol/L and 141 ± 22 mg/dL reported for HDL-cholesterol and apoA-I, respectively, by the National Cholesterol Education Program(23). The HDL cholesterol values for cohort 3 are similar to those seen in cohort 1 of the study (0.47 ± 0.23 versus 0.54 ± 0.21 mmol/L). A higher percentage of apoA-I was found in the non-lipoprotein bottom fraction. Surprisingly, although plasma triglycerides were within normal limits, the percentage of core triglyceride in the HDL was markedly elevated. Analyses of convalescent samples from the same patients (Table 3) showed significant increases of the HDL cholesterol and apoA-I levels toward the normal range. Plasma triglyceride values did not change significantly at follow-up. However, the percentage of HDL core triglyceride, which was significantly elevated at presentation, decreased but was still above the normal range at follow-up (2-6 wk). There were no significant changes in the other lipid or protein components of HDL.
One child was excluded from the above analysis because of the presence of an acute otitis media at the 6-wk follow-up assessment. It is of note that this child continued to have SAA in HDL at follow-up, with a decline in HDL cholesterol (1.27 to 0.96 mmol/L and no change in apoA-I (107 mg/dL at both assessments). This child with an intercurrent acute illness was the only subject in the study who did not have an increase in HDL cholesterol from the acute presentation to the first follow-up visit.
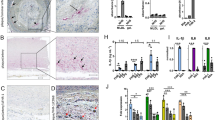
Analyses of the lipoprotein particles obtained by density gradient centrifugation of plasma showed that SAA was present in the HDL of all 12 children with acute Kawasaki disease and was absent in all seven patients with convalescent specimens at follow-up (data not shown). The SAA was present in HDL with peak levels at the density region of HDL3 (fractions 23-28, d > 1.125 g/mL) (Fig. 3). Barely detectable levels were found in the non-lipoprotein associated bottom fractions (fractions 29-30, d > 1.21 g/mL) confirming that SAA circulates in plasma bound to HDL3-like particles.
Western immunoblotting showing the presence of SAA in the various lipoprotein fractions obtained by density gradient centrifugal flotation of plasma. Density of the fractions is indicated in the bottom. Odd numbered fractions were assessed. Results are shown for fractions 17-30. There were no detectable SAA in fractions 1-15.
DISCUSSION
Earlier studies showed diminished HDL cholesterol and apoA-I in association with the acute phase response(6–12), including decreases of HDL-cholesterol in the acute phase of Kawasaki disease(2–5). The present data show: 1) that children with Kawasaki disease have extremely low serum HDL cholesterol and apoA-I at the time of the acute illness, 2) that SAA is present in the acute stage and is associated mainly with HDL particles of HDL3 density, 3) that HDL and apoA-I concentrations rapidly increase toward the normal range and SAA disappears after resolution of the acute stage of Kawasaki disease, and 4) that SAA is present at the time of other acute illnesses in childhood in association with variable changes in HDL and apoA-I. Whether the severity of the lipoprotein changes in Kawasaki disease is disease specific or related to the duration of inflammation cannot be assessed in this study.
This is the first report of apoA-I levels and of the correlations among apoA-I, HDL cholesterol and SAA in Kawasaki disease. The previous studies that assessed lipoprotein responses in Kawasaki disease have shown that HDL-cholesterol is low during the acute phase of the illness and rises subsequently(2–5). Most studies including the present one suggest that these values return to normal after complete recovery; however, Newburger et al.(3) reported that HDL cholesterol levels may be depressed for more than 1 y after resolution of acute Kawasaki disease. We found that although HDL and apoA-I concentrations are within the normal range 1-2 mo after acute Kawasaki disease, further increases in both parameters occur for up to 1 y (Figs. 1 and 2). Even with the early normalization of HDL and apoA-I concentrations in convalescent Kawasaki disease, HDL composition is not fully normalized early, as reflected by the sustained enrichment in HDL triglyceride at 2 wk of convalescence (Table 2). Miwata et al.(18) analyzed the SAA concentrations in acute viral infections of children and showed that SAA is significantly higher in the acute phase of patients with Kawasaki disease compared with other illnesses. Although the responses to a variety of infections are variable, the degree of reduction in HDL and apoA-I levels in our Kawasaki disease subjects is more marked than in other acute illnesses. Our results suggest that the decline in HDL cholesterol is part of the acute phase response as confirmed by the presence of SAA, and that normal levels are again achieved by 2-12 mo after the acute illness.
The precise mechanism of the decline of HDL during acute illnesses such as Kawasaki disease remains unknown. The factors that account for the decline in HDL-cholesterol and apoA-I may not be precisely the same. The decline in HDL-cholesterol may relate to a change in the formation or catabolism of HDL and its apoproteins as well as the replacement of core cholesteryl ester by triglyceride. Triglyceride enrichment of acute phase HDL is observed here and in studies of rabbits and non-human primates(8). This occurs even in the absence of an elevation of plasma triglyceride, perhaps due to a change in the mass of cholesteryl ester transfer protein or its directional activity. The effect of the acute phase response on the cholesteryl ester transfer protein mass or activity is yet to be fully investigated. The sustained triglyceride enrichment of HDL after an episode of Kawasaki disease is unexplained. Due to the limited amount of samples, the triglyceride content of HDL in the other acute illnesses was not assessed.
The decline in plasma apoA-I may be the consequence of its decreased synthesis and/or increased catabolism. The marked increase in plasma SAA level and its association with HDL displaces apoA-I from the lipoprotein surface with the free apoprotein likely to be rapidly removed from the plasma. Both purified SAA(19) and recombinant SAA (our unpublished data) displace apoA-I from the HDL in vitro. Displacement of apoA-I by HDL is suggested by the increased percentage of lipid-free apoA-I in the bottom fraction of the patients' plasma (Table 1). The contribution of the HDL triglyceride enrichment to the lowering of plasma apoA-I is not certain. Elevations of plasma triglyceride, as with inhibition of lipoprotein lipase, lead to increased apoA-I catabolism, probably in the kidney(20). Triglyceride enrichment of HDL increases apoA-I catabolism in the perfused rabbit kidney(13). Consistent effects of the acute phase response on apoA-I synthesis have not yet been reported. Our preliminary data suggest that there is a decline in apoA-I mRNA in the livers of endotoxin-treated animals.
Acute changes in lipids and lipoproteins are characteristic of infections and chronic inflammatory diseases(11, 12, 21). Several studies have shown declines in total cholesterol and HDL cholesterol levels in response to bacterial and viral infection. The present study confirms that these changes occur in many acute pediatric illnesses; most interesting was our case of a child with acute otitis media at the 6-wk follow-up for Kawasaki disease who continued to have evidence of an acute phase response. These results emphasize that the use of lipid profiles to assess cardiovascular risk is not reliable during acute illnesses and possibly for several weeks thereafter.
Recent studies of the pathogenesis of atherosclerosis have shown that HDL is important in inhibiting the role of oxidized LDL in initiating arterial wall injury(22). HDL particles that have incorporated SAA do not perform this inhibitory function. Therefore, it is tempting to speculate that the lipoprotein changes secondary to the acute phase response of Kawasaki disease may be an additional factor that contributes to the evolution of the coronary artery injury seen in this disease. However, further testing of this hypothesis must await a satisfactory animal model of this disease and more information about smooth muscle cell responses to both oxidized LDL and SAA-containing HDL particles.
Abbreviations
- SAA:
-
serum amyloid A
- apoA-I:
-
apolipoprotein A-I
References
Shulman ST, De Inocencio J, Hirsch R 1995 Kawasaki disease. Pediatr Clin North Am 42: 1205–1222.
Okada T, Harada K, Okuni M 1982 Serum HDL-cholesterol and lipoprotein fraction in Kawasaki disease (acute mucocutaneous lymph node syndrome). Jpn Circ J 46: 1039–1044.
Newburger JW, Burns JC, Beiser AS, Loscalzo J 1991 Altered lipid profiles after Kawasaki syndrome. Circulation 84: 625–631.
Solo E, Pesonen E, Viikari J 1991 Serum cholesterol levels during and after Kawasaki disease. J Pediatr 119: 557–561.
Inoue O, Sugimura T, Kato H 1993 Long term lipid profiles in patients with Kawasaki disease. In: Takahashi M and Taubert K (eds) Proceedings of the Fourth International Symposium on Kawasaki Disease. American heart Association, Dallas TX, pp 305–309.
Parks JS, Rudel LL 1985 Alteration of high density lipoprotein subfraction distribution with induction of serum amyloid A protein (SAA) in the nonhuman primate. J Lipid Res 26: 82–91.
Cabana VG, Siegel JN, Sabesin SM 1989 Effects of the acute phase response on the concentration and density distribution of plasma lipids and apolipoproteins. J Lipid Res 30: 39–49.
Cabana VG, Lukens JR, Rice KS, Hawkins TJ, Getz GS 1996 HDL content and composition in acute phase response in three species. J Lipid Res 37: 2662–2674.
Fahie-Wilson M, Mills R, Wilson K 1987 HDL-cholesterol and the acute phase response following myocardial infarction and acute pancreatitis. Clin Chem Acta 167: 197–209.
Eriksen N, Benditt EP 1984 Trauma, high density lipoproteins, and serum amyloid A. Clin Chim Acta 140: 139–149.
Alvarez C, Ramos A 1986 Lipids, lipoproteins, and apoproteins in serum during infection. Clin Chem 32: 142–145.
Sammalkorpi K, Valtonen V, Kerttula Y, Nikkila E, Taskinen MR 1988 Changes in serum lipoprotein pattern induced by acute infections. Metabolism 37: 859–865.
Horowitz BS, Goldberg IJ, Merab J, Vanni TM, Ramakrishnan R, Ginsberg IIN 1993 Increased plasma and renal clearance of an exchangeable pool of apolipoprotein A-I in subjects with low levels of high density lipoprotein cholesterol. J Clin Invest 91: 1743–1752.
Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association 1993 Diagnosis and therapy of Kawasaki disease in children. Circulation 87: 1776–1780 pp
Albers JJ, Wahl PW, Cabana VG, Hazzard WR 1975 Quantitation of human plasma apolipoprotein A-1 of human high density lipoprotein. Relationship to age, sex and lipid levels. Metabolism 24: 633–644.
Weber K, Osborne M 1969 The reliability of molecular weight determination by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem 244: 4406–4412.
Towbin H, Gordon J 1984 Immunoblotting and dot blotting-current status and outlook. J Immunol Methods 7: 313–340.
Miwata H, Yamada Y, Okada M, Kudo T, Kimura H, Morishima T 1993 Serum amyloid A in acute viral infections. Arch Dis Childhood 68: 210–214.
Coetzee GA, AF Strachan, DR van der Westhuyzen, HC Hoppe, MS Jeenah, FC de Beer 1986 Serum amyloid A-containing human high density lipoprotein 3. J Biol Chem 261: 9644–9651
Goldberg IJ, Blaner WS, Vanni VM, Moukides M, Ramakrishnan R 1990 Role of lipoprotein lipase in the regulation of high density lipoprotein apolipoprotein metabolism. J Clin Invest 86: 463–473.
Kumon Y, Suehiro T, Ikeda Y, Hashimoto K, Ohno F 1993 Influence of serum amyloid A protein on high-density lipoprotein in chronic inflammatory disease. Clin Biochem 26: 505–511.
VanLenten BJ, Hama SY, DeBeer FC, Stafforini DM, McIntyre TM, Prescott SM, LaDu BN, Fogelman AM, Navab M 1995 Anti-inflammatory HDL becomes proinflammatory during the acute phase response. Loss of protective effect of HDL against LDL oxidation in aortic wall cell cocultures. J Clin Invest 96: 2758–2767.
National Cholesterol Education Program Report of the Expert Panel on Blood Cholesterol Levels in Children and Adolescents 1991. National Heart, Lung, and Blood Institute Information Center, Bethesda, MD
Srinivasan SR Berenson GS 1995 Serum apolipoproteins A-I and B as markers of coronary artery disease risk in early life: the Bogalusa Heart Study. Clin Chem 41: 159–164.
Author information
Authors and Affiliations
Additional information
Supported by National Institutes of Health Grant HL-15062 (Specialized Center of Research in Atherosclerosis), American Heart Association Grant-in-Aid 96015880 (V.G.C.) (Formation and Interaction of Acute Phase HDL), and the Children's Memorial Hospital Kawasaki Disease Fund.
Rights and permissions
About this article
Cite this article
Cabana, V., Gidding, S., Getz, G. et al. Serum Amyloid A and High Density Lipoprotein Participate in the Acute Phase Response of Kawasaki Disease. Pediatr Res 42, 651–655 (1997). https://doi.org/10.1203/00006450-199711000-00017
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199711000-00017
This article is cited by
-
Plasma Clusterin Levels in Predicting the Occurrence of Coronary Artery Lesions in Patients With Kawasaki Disease
Pediatric Cardiology (2010)
-
Kawasaki disease: a review with emphasis on cardiovascular complications
Insights into Imaging (2010)
-
Acute-Phase Reactants and a Supplemental Diagnostic Aid for Kawasaki Disease
Pediatric Cardiology (2010)
-
Age-Related Acceleration of Endothelial Dysfunction and Subclinical Atherosclerosis in Subjects with Coronary Artery Lesions After Kawasaki Disease
Pediatric Cardiology (2009)