Abstract
Nitric oxide (NO) influences cerebral vascular tone both in the normal fetus and in the hypoxemic fetus, but during postnatal life this regulating role of NO seems less prominent. It is therefore possible that under conditions when arterial oxygen content is at postnatal levels NO exerts no action on smooth muscle. We therefore examined the impact of NO on cerebral blood flow and vascular resistance in five near-term lamb fetuses during intrauterine ventilation and oxygenation. Four additional fetuses were pretreated with indomethacin to investigate a posible additional regulatory role of prostaglandins on cerebral vascular resistance. Cerebral blood flow(Qbrain) was measured using radionuclide-labeled microspheres. A tracheal tube was inserted to ventilate the fetus. After recovery, Qbrain and resistance in the cerebral vascular bed(Rcer) were measured during the following subsequent conditions: before and after increasing fetal arterial O2 content by ventilation with air, after inhibition of NO production with Nω-nitro-L-arginine during and after cessation of ventilation, and finally after infusion of L-arginine to increase nitric oxide production. Ventilation decreased Qbrain (95 ± 18 to 47 ± 15 mL/100 g/min) and increased Rcer.Nω-Nitro-L-arginine did not alter Qbrain (52 ± 13 mL/100 g/min) or Rcer during ventilation and oxygenation, indicating no modulating role of NO during higher arterial oxygen content. On cessation of ventilation, Po2 returned to fetal levels and Qbrain increased significantly, but did not return to baseline fetal values (83 ± 7 mL/min). Infusion of L-arginine increased Qbrain to baseline fetal levels (116± 30 mL/min). However, indomethacin pretreatment prevented the rise in cerebral blood flow after cessation of ventilation and after additional L-arginine infusion (Qbrain, 53 ± 20 and 52 ± 4 mL/100 g/min, respectively). These studies indicate that, during postnatal levels of arterial oxygen content, NO does not exert an action on smooth muscle cells of the cerebral resistance vessels as it does at lower arterial(fetal) oxygen content. They further show that prostaglandins are important in facilitating the full expression of NO-induced vasodilation.
Similar content being viewed by others
Main
Cerebral blood flow decreases and cerebral vascular resistance increases in relation to an increase in arterial oxygen content, a process occurring at birth with the onset of ventilation(1). Previously, we have shown that endothelial derived NO modulates tone in cerebral vessels in the fetal lamb, which has a much lower arterial oxygen content compared with the situation after birth. We also showed that NO was involved in cerebral vasodilation induced by fetal hypoxemia(2). Although some recent studies report a regulatory effect of NO on cerebral blood flow postnatally(3, 4), other investigators could not confirm these findings and suggested that NO does not have a key role in regulating cerebral vascular resistance at normal arterial oxygen content in the early postnatal period(5, 6).
To elucidate the role of NO for the regulation of cerebral perfusion during the early postnatal period, the present study was designed. We hypothesized that, under conditions when arterial oxygen content is at postnatal levels, either NO is not produced, or it does not exert an action on smooth muscle and that reducing arterial oxygen content to fetal levels either increases NO production or enhances its action on smooth muscle. A second objective of the present work was to study the additional regulatory role of prostaglandins during these conditions.
Late gestation fetal lambs were ventilated to increase arterial Po2 to normal postnatal levels; NO production was inhibited by administration of NNLA, a competitive analog of L-arginine. In a subset of fetal lambs the study was repeated after pretreatment with indomethacin, an inhibitor of prostaglandin synthesis(7).
METHODS
Animals and surgical preparation. We studied nine fetal sheep with gestational ages ranging from 125 to 136 d and weights ranging from 2980 to 3850 g. All surgical and experimental procedures were approved by the Animal Care and Use Committee at the University of California, San Francisco. The ewe was fasted for at least 24 h before surgery. After local anesthesia with 2% lidocaine, low epidural anesthesia was achieved with 4 mL of 1% tetracaine hydrochloride (Pontocaine HCl; Breon Laboratories, New York, NY). Polyvinyl catheters (1.3-mm inner diameter, 2.3-mm outer diameter) were advanced into the descending aorta and inferior vena cava of the ewe. NaCl 0.9% solution was infused continuously into the ewe, and 50-100 mg of ketamine hydrochloride (Vetalar; Parke-Davis, Morris Plains, NJ) were administered i.v. to the ewe every 10-15 min to maintain sedation.
Through a midline incision in the ewe's abdomen, the pregnant horn of the uterus was exposed, and a uterine incision was made overlying the fetal hind limbs. Local anesthesia was accomplished with 0.5% lidocaine hydrochloride before each fetal skin incision. Also 5 mg of ketamine per kg estimated fetal weight was given i.m. to the fetus. Polyvinyl catheters (0.75-mm inner diameter, 1.2-mm outer diameter) were passed to the fetal descending aorta and inferior vena cava via the hind limb vessels. The fetal skin and uterine incisions were sutured. A second incision was made over the left chest of the fetus. A specially constructed catheter was placed directly in the left atrium(20 gauge i.v. catheter, Becton Dickinson, Mountain View, CA), in those fetuses in which microsphere determined cerebral blood flow determinations were planned during oxygenation and pulmonary ventilation (see“Experimental Procedure”). The fetal chest was sutured in layers. Next, the fetal head, neck, and fore limbs were exposed through the same uterine incision. A 3.5-mm endotracheal tube was connected by an Y-tube to two 100-cm lengths of flexible polyvinyl tubing (3.2-mm inner diameter, 6.4-mm outer diameter). The tubing was filled with 0.9% saline, and the trachea was intubated. A catheter was placed in the amniotic cavity and connected to the ventilation tubing to allow tracheal fluid to flow freely from the uterus to the amniotic cavity postoperatively. Then a polyvinyl catheter (0.75-mm inner diameter, 1.2-mm outer diameter) was inserted via a lingual artery into a carotid artery for infusion of drugs, and another polyvinyl catheter (1.3-mm inner diameter, 2.3-mm outer diameter) was inserted into the right brachial artery and advanced to the brachiocephalic trunk for monitoring arterial blood pressure and for blood sampling. In the midline of the scalp, an incision was made to expose the cranial suture. The sagittal sinus was cannulated with a 20-guage i.v. catheter (Becton Dickinson, Mountain View, CA) for collection of cerebral venous blood. The incision was sutured, and the head was returned to the uterus. The uterus was sutured. The vascular catheters were filled with heparin sodium solution (1000 U/mL) and sealed, and all catheters and ventilation tubing were exteriorized to the maternal flank and protected by a cloth pouch. Antibiotics (400 mg of gentamicin, 1 million U of penicillin) were supplied to the amniotic cavity and maternal vein on the day of surgery and each day thereafter. The animals were allowed 24-48 h to recover from surgery.
Physiologic measurements. During the experiments fetal blood pressures were monitored continuously using Statham P23Db strain-guage transducers and Beckman R 612 (Beckman Instruments, Inc., Palo Alto, CA) or Gould 2800S (Gould, Inc., Cleveland, OH) polygraph. Fetal carotid and sagittal sinus blood pressures were calculated with amniotic cavity pressure as the zero reference. Fetal heart rate, measured by a cardiotachometer, triggered by an arterial pulse wave, was recorded continuously. Blood samples for determination of pH, Po2, Pco2, arterial and sagittal sinus oxygen saturation, and Hb concentration were obtained from the carotid artery and sagittal sinus.
We measured total fetal brain blood flow (Qbrain) using radionuclide-labeled microspheres during a limited number of estimations of brain perfusion (up to five per animal) in both the nonventilation and the ventilation/oxygenation conditions in four fetuses pretreated with indomethacin, and in five animals that did not receive indomethacin (see also“Experimental Procedure”). Qbrain was measured as described previously(8, 9). Briefly, microspheres, 15 μm in diameter and labeled with 114In, 153Gd, 57Co,51 Cr, 113Sn, 85Sr, 95NB, and 46Sc, were injected into the inferior vena caval or left atrial catheters (ventilated and oxygenated fetuses) over a 30-s period, whereas reference samples were obtained at a rate of 4 mL/min from the brachiocephalic trunk for 1.00-1.25 min. After the studies were completed, the brain was removed at the base, weighed, and placed in formalin. The tissue was carbonized in an oven at 350°C, ground into a coarse powder, placed into plastic vials to a uniform height of 3 cm, and counted in a 1000-channel multichannel pulse-height analyzer (Norland Corp., Fort Atkinson, WI). The activity of each isotope in brain tissue was determined by the least squares method(9). Qbrain was calculated by comparing the radioactivity in brain tissue with radioactivity and withdrawal rate of the appropriate reference sample from the brachiocephalic trunk and expressed as milliliters/100 g/min, as described previously(8).
To ventilate the fetus, the tubes connected to the endotracheal tube were opened, and tracheal fluid was allowed to drain. Exosurf (Burroughs Wellcome), an artificial surfactant, was instilled into the trachea to facilitate pulmonary ventilation, and the tubes were connected to a positive pressure ventilator(1).
Experimental procedure. During steady state (heart rate, MCBP), a first set of five fetuses were studied in the following order: 1) without ventilation (FET-Po2 condition); 2) during pulmonary ventilation with room air or, occasionally, with added oxygen, to obtain blood gases normally seen in (preterm) newborns (Po2 45-100 mm Hg; OXY condition); 3) after slow (0.6 mg/kg/min) infusion of 15 mg/kg NNLA directly into the cerebral circulation via the lingual artery to block NO production (NNLA-OXY condition)(5); 4) after returning to normal fetal Po2 levels (NNLA-FET-Po2 condition); and 5) 10 min after infusion of L-arginine in a dose of 150 mg/kg into the lingual artery (LA-FET-Po2 condition).
A second set of four fetuses was pretreated with indomethacin sodium trihydrate (3 mg/kg, administered directly into the cerebral circulation by infusing it into the lingual artery over a 10-min period). Indomethacin produced a modest transient increase in arterial blood pressure, which returned to pretreatment values within 30-60 min. After reaching steady state, these fetuses were subjected to the same experimental procedure.
During each condition, the following variables were measured when heart rate and MCBP had reached steady state: Qbrain, MCBP, heart rate, blood gases and pH, blood O2 saturation, and Hb concentration .Rcer was expressed as the ratio between MCBP and Qbrain, and CMRo2 was calculated CO2 and CMRo2 were determined as follows. CO2 was calculated as: g Hb/dL× 1.36 mL O2/g Hb ×% saturation of Hb with O2. CMRo2 was calculated as: (arterial CO2 - sagittal sinus CO2) × brain blood flow; the result is expressed in mL of O2/100 g brain/min.
Statistical analysis. All data are summarized as means ± 1 SD. Differences between mean values during the different conditions were assessed by one-factor ANOVA for repeated measurements, followed by the Student-Newman-Keuls test when a significant difference was found. A p value <0.05 was considered statistically significant.
RESULTS
Physiologic Measurements
Table 1 shows the Pao2, Paco2, and MCBP values during each experimental condition in fetuses that were not pretreated with indomethacin and in those that received indomethacin. Paco2, pH, heart rate, or Hb (pH, heart rate, and Hb not shown) were always within physiologic ranges and showed no significant differences in the various conditions in both groups. Pao2, as expected, increased significantly during ventilation and oxygenation (p < 0.05). Although MCBP was higher after NO inhibition with NNLA in fetuses not treated with indomethacin, differences reached statistical significance only without ventilation and oxygenation (NNLA-FET-Po2 condition); L-arginine administration caused a drop to values obtained before NO inhibition with NNLA. In indomethacin-pretreated fetuses MCBP was higher compared with non-indomethacin-treated fetuses, increased significantly after NO inhibition with NNLA, and remained higher during the subsequent conditions.
Patterns of Q brain and R cer during Experimental Conditions
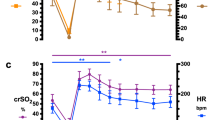
Non-indomethacin-pretreated fetuses. Figure 1, A and B, summarizes the patterns of Qbrain and Rcer in the fetuses during the various conditions. Ventilation and oyxgenation (OXY condition) of the fetuses produced a significant decrease in Qbrain (96 ± 18 to 47 ± 15 mL/100 g/min, p < 0.05), and increase in Rcer(0.63 ± 0.11 to 1.22 ± 0.35 mm Hg/mL/100 g/min, p < 0.05). Blockade of NO production by NNLA during ventilation and oxygenation(NNLA-OXY condition) had no significant effect on Qbrain or Rcer (Qbrain, 52 ± 12 mL/100 g/min;Rcer, 1.22 ± 0.39 mm Hg/mL/100 g/min). After cessation of ventilation and oxygenation (NNLA-FET-Po2 condition), there was a significant increase in Qbrain and decrease in Rcer (Qbrain, 82 ± 7 mL/100 g/min;Rcer, 0.78 ± 0.06, p < 0.05). After L-arginine infusion (LA-FET-Po2 condition) to abolish the inhibitory effect of NO, Qbrain increased and Rcer decreased further (FET-Po2-condition: Qbrain, 116± 30 mL/100 g/min; Rcer, 0.52 ± 0.19 mm Hg/mL/100 g/min, p < 0.05). CMRO2 did not change significantly after ventilation and oxygenation (3.63 ± 1.05versus 2.48 ± 0.70 mL of O2/100 g/min), and also was not significantly different after NNLA (3.88 ± 0.69 and 4.26 ± 1.44 mL of O2/100 g/min, respectively).
Indomethacin pretreated fetuses. Figure 2, A and B, summarizes the patterns of Qbrain and Rcer during the various conditions in indomethacin-pretreated fetuses. Ventilation and oyxgenation (OXY condition) of the fetuses produced a significant decrease in Qbrain (89 ± 13 to 55 ± 24 mL/100 g/min, p < 0.05), and consequent increase in Rcer (0.73 ± 0.18 to 1.32 ± 0.20 mm Hg/mL/100 g/min, p < 0.05). Thus indomethacin pretreatment had no effect on this response. Blockade of NO production by NNLA during ventilation and oxygenation (NNLA-OXY condition) also had no significant effect on Qbrain or Rcer, although Qbrain and Rcer tended to decrease and increase, respectively: Qbrain, 40 ± 17 mL/100 g/min;Rcer, 1.82 ± 0.59 mm Hg/mL/100 g/min. After cessation of ventilation and oxygenation (NNLA-FET-Po2 condition), there was no significant change in Qbrain and Rcer(Qbrain, 57 ± 28 mL/100 g/min; Rcer, 123 ± 0.41 mm Hg/mL/100 g/min). After additional L-arginine infusion(LA-FET-Po2 condition) Qbrain and Rcer values did not change during this condition after indomethacin pretreatment (Qbrain, 53 ± 9 mL/100 g/min; Rcer, 1.40 ± 0.15 mm Hg/mL/100 g/min). CMRO2 did not change significantly after ventilation and oxygenation(4.76 ± 2.90 versus 3.92 ± 1.73 mL of O2/100 g/min), and also was not significantly different after NNLA or indomethacin administration (3.12 ± 0.53 and 3.74 ± 0.71 mL of O2/100 g/min, respectively).
DISCUSSION
Major changes in blood flow patterns and in blood flow to individual organs occur in relation to birth(1), which appears to be stimulated by an increase in arterial oxygen content associated with ventilation(2, 4).
Blood flow to the brain falls dramatically after the increase in arterial oxygen content(1). Cerebral vessels in the fetus are normally influenced by endothelium-derived NO. In observations in lambs of similar gestational age to those in this study, we noted that administration of the competitive blocker of NO release, NNLA, caused cerebral vasoconstriction, and in addition, limited the increase in cerebral flow usually resulting from acute fetal hypoxemia(2). Fetal cerebral blood flow is also affected by prostaglandin mechanisms; indomethacin, a prostaglandin synthesis inhibitor, produces cerebral vasoconstriction in late gestational fetal lambs(7).
We raised the question whether or not both NO and prostaglandins may influence cerebral vascular changes at the time of birth. To examine the possible roles of NO and prostaglandins in cerebral circulatory changes during different levels of arterial oxygen content, we observed the effect of inhibition of NO release and of an indomethacin-induced inhibition of prostaglandin synthesis on late gestation fetal lambs in utero, before and after increasing arterial oxygen content by ventilation with air.
In these studies, we confirmed our previous observations that cerebral blood flow falls when arterial oxygen levels are increased by ventilating the fetus(1). Although NNLA did not modify this decrease in cerebral blood flow, it had an effect on the recovery of cerebral blood flow when Pao2 returned to fetal levels after ventilation was stopped. Previously, we have observed that, in normal fetal lambs, cerebral blood flow increases to control levels when Pao2 values fall after cessation of ventilation and oxygenation(2). However, in those fetuses receiving NNLA but not indomethacin, return to fetal Pao2 values was associated with a significant rise in brain blood flow, but it did not return to baseline values, at the same Pao2 values as before NNLA administration (FET-Po2). L-Arginine infusion restored flows eventually to baseline levels. Because Paco2 has potent vasoactive properties on the (fetal) cerebral vascular bed, we have to consider the possibility that changes in Paco2 might have influenced the results of the present study. There was indeed a nonsignificant but consistent small decrease in Paco2 during ventilation and oxygenation compared with nonventilation and oxygenation conditions. It seems, however, not very probable that this small drop in Paco2 caused the rather huge decreases in brain blood flow induced by ventilation and oxygenation.
In the fetuses pretreated with indomethacin and also given NNLA, there was no significant rise of cerebral blood flow when Pao2 values returned to fetal levels after stopping ventilation. Furthermore, infusion of L-arginine did not significantly increase flows. These findings suggest that both NO and prostaglandin effects on cerebral flow are affected by ventilation and oxygenation. The partial restoration of cerebral flow on return to fetal Pao2, in the animals not receiving indomethacin, indicates that prostaglandin effects are inhibited with oxygenation and restored at fetal Pao2 levels. The failure of NNLA to produce a further reduction in cerebral blood flow after ventilation suggests that oxygenation abolished any NO effect on cerebral vessels.
Prostaglandins have been shown to exert a vasodilatory effect on cerebral vessels. Leffler et al.(10) have suggested that prostaglandin has a permissive role in the cerebral vasodilation associated with hypercapnia, and prostanoids also influence the cerebral vascular responses to opioids in newborn piglets(11). Moore et al.(12) have shown that both NO and prostanoids are involved in the increase in cerebral blood flow resulting from isoflurane administration to pigs. We have shown that indomethacin causes a reduction in cerebral blood flow in the fetal lamb(7). Although we attributed the decrease in brain perfusion to inhibition of vasodilatory prostaglandins, indomethacin has also been known to exert its vasoconstrictive action via other mechanisms. It has been suggested that indomethacin has a direct effect on smooth muscle cells by inhibition of the influx of calcium(13), whereas Konig et al.(14) suggested an indomethacin-induced inhibition of histamine release. Moreover, rather recently it has also been suggested that the free radical scavenging effect of cyclooxygenase inhibitors may be the mechanism by which the cerebral vascular bed constricts after indomethacin treatment(15, 16). Nonetheless, the rather high dose of indomethacin used in the present study will undoubtedly have blocked prostaglandin synthesis, as in fact was shown by Chemtob et al.(17) who reported that high dose indomethacin infusions in newborn piglets induced decreases in systemic and cerebrovascular prostaglandin concentrations. In our studies, we could not indicate which prostanoids are responsible for the cerebral vascular changes. Also, we are unable to state whether oxygenation limits production of prostanoids, affects their action on the vessel, or modulates their possible permissive role in vascular dilatory response to some other agent. Chemtob et al.(18) showed in newborn piglets that prostaglandin E, prostaglandin F2α, and prostaglandin I2 had a vasodilatory action on the cerebral resistance vessels, whereas thromboxane B2 had a merely vasoconstrictive action. Ment et al.(19) showed in a study on newborn puppies that hypoxia increases the production of vasodilatory prostaglandins, suggesting a relationship between prostanoids and arterial oxygen tension. The inability of L-arginine infusion to restore cerebral blood flow to baseline fetal levels in those animals that received indomethacin raises the possibility that prostanoids may be important in enhancing or permitting NO vasodilation.
NO has been shown to modulate cerebral vascular resistance in fetal lambs; it contributes to maintaining a degree of vasodilation in the normal fetus and is also important in mediating the increase in cerebral blood flow resulting from fetal hypoxemia(2). It has also recently been reported that NO is involved in the posthypoxic-ischemic vasodilation of fetal lamb cerebral vascular bed: NO synthase inhibition attenuates the delayed vasodilation after fetal cerebral ischemia(20). Although some recent studies report an effect of NO synthase inhibitors on cerebral blood flow postnatally(3, 4, 21), other studies in newborn piglets have shown that NO does not have a key role in regulating cerebral vascular resistance postnatally(5, 6, 22, 23). We also noted that administration of NNLA had no significant effect on cerebral vascular resistance after ventilation and oxygenation in the fetal lamb.
In summary, under conditions when Pao2 is at postnatal levels, either NO is not produced, or it does not exert an action on smooth muscle reducing Pao2 to fetal levels either increases NO production, or enhances its action on vascular smooth muscle is supported by the present study. It further suggests that prostanoids are also important in facilitating the full expression of NO-induced vasodilation.
Abbreviations
- Co2:
-
blood oxygen content
- CMRo2:
-
cerebral metabolic rate for oxygen
- FET-Po2:
-
fetal normoxemia
- LA-FET-Po2:
-
fetal normoxemia after L-arginine infusion
- MCBP:
-
mean carotid blood pressure
- NNLA:
-
Nω-nitro-L-arginine
- NNLA-OXY:
-
pulmonary ventilation and oxygenation after NNLA infusion
- NNLA-FET-Po2:
-
fetal normoxemia after NNLA infusion
- NO:
-
nitric oxide
- OXY:
-
pulmonary ventilation and oxygenation
- R cer :
-
total cerebral vascular resistance
- Q brain :
-
microspheres determined brain blood flow
- Pao2:
-
pressure of arterial O2
- Paco2:
-
pressure of arterial CO2
References
Iwamoto HS, Teitel DF, Rudolph AM 1987 Effects of birth-related events on blood flow distribution. Pediatr Res 22: 634–640.
Van Bel F, Sola A, Roman C, Rudolph AM 1995 Role of nitric oxide in regulation of cerebral circulation in the lamb fetus during normoxemia and hypoxemia. Biol Neonate 68: 200–210.
Greenberg RS, Helfaer MA, Kirsch JR, Moore LA, Traystman RJ 1994 Nitric oxide synthase inhibition with NG-mono-methyl-L-arginine reversibly decreases cerebral blood flow in piglets. Crit Care Med 22: 384–392.
Greenberg RS, Helfaer MA, Kirsch JR, Traystman RJ 1995 Effect of nitric oxide synthase inhibition on postischemic cerebral hyperemia. Am J Physiol 269:H341–H347.
Busija DW, Leffler CW, Wagerle LC 1990 Mono-L-arginine-containing compounds dilate piglet pial arterioles via an endothelium-derived relaxing factor-like substance. Circ Res 67: 1374–1380.
Takei Y, Edwards AD, Lorek A, Peebles DM, Belai A, Cope M, Delpy DT, Reynolds EOR 1993 Effects of N-ω-nitro-L-arginine methyl ester on the cerebral circulation of newborn piglets quantified in vivo by near-infrared spectroscopy. Pediatr Res 34: 354–359.
Van Bel F, Bartelds B, Teitel DF, Rudolph AM 1995 Effect of indomethacin on cerebral blood flow and oxygenation in the normal and ventilated fetal lamb. Pediatr Res 38: 243–250.
Heymann MA, Payne BD, Hoffman JIE, Rudolph AM 1977 Blood flow measurements with radionuclide-labeled particles. Prog Cardiovasc Dis 20: 55–79.
Bear RW, Payne BA, Verrier ED, Vlahakes GJ, Molodowitch D, Uhlig PN, Hoffman JIE 1984 Increased number of myocardial blood flow measurements with radionuclide-labeled microspheres. Am J Physiol 246:H418–H434.
Leffler CW, Mirro R, Pharris LJ, Shibata m 1994 Permissive role of prostacyclin in cerebral vasodilation to hypercapnia in newborn pigs. Am J Physiol 267: H285–H291.
Armstead WM, Mirro R, Busija DW 1990 Prostanoids modulate opioid cerebrovascular responses in newborn pigs. J Pharmacol Exp Ther 255: 1083–1090.
Moore LE, Kirsch JR, Helfaer MA, Tobin JR 1994 Nitric oxide and prostanoids contribute to isoflurane-induced cerebral hyperemia in pigs. Anesthesiology 80: 1328–1337.
Northover BJ 1971 Mechanism of the inhibitory action of indomethacin on smooth muscle. Br J Pharmacol 41: 540–551.
Konig W, Brom J, Schonfeld A, Knoller J, Stuning M 1987 Effect of tenoxicam and indomethacin on the release of histamine, prostaglandin E2 and leukotrienes from various cells. Arzneimittelforschung 37: 296–299.
Kennedy TP, Rao NV, Noah W, Micheal JR, Jafri MH Jr, Gurtner GH, Hoidal JR 1990 Ibuprofen prevents oxidant lung injury and in vitro lipid peroxidation by chelating iron. J Clin Invest 86: 1565–1573.
Pourcyrous M, Leffler CW, Bada HS, Korones SB, Busija DW 1993 Brain superoxide anion generation in asphyxiated piglets and the effect of indomethacin at therapeutic dose. Pediatr Res 34: 366–369.
Chemtob S, Beharry K, Barna T, Varma DR, Aranda JV 1991 Differences in effects in the newborn piglet of various nonsteroidal anti-inflammatory drugs on cerebral blood flow but not on cerebrovascular prostaglandins. Pediatr Res 30: 106–111.
Chemtob S, Beharry K, Rex J, Varma DR, Aranda JV 1990 Changes in cerebrovascular prostaglandins and thromboxane as a function of systemic blood pressure. Cerebral blood flow autoregulation of the newborn. Circ Res 67: 674–682.
Ment LR, Stewart WB, Duncan CC, Pitt BR, Cole JS 1986 Beagle puppy model of perinatal cerebral infarction. Regional cerebral prostaglandin changes during acute hypoxemia. J Neurosurg 65: 851–855.
Marks KA, Mallard CE, Roberts I, Williams CE, Gluckman PD, Edwards AD 1996 Nitric oxide synthase inhibition attenuates delayed vasodilation and increases injury after cerebral ischemia in fetal sheep. Pediatr Res 40: 185–191.
Thompson BG, Pluta RM, Girton ME, Oldfield EH 1996 Nitric oxide mediation of chemoregulation but not autoregulation of cerebral blood flow in primates. J Neurosurg 84: 71–78.
Meadow W, Rudinsky B, Bell A, Lozon M, Randle C, Hipps R 1994 The role of prostaglandins and endothelium-derived relaxation factor in the regulation of cerebral blood flow and cerebral oxygen utilization in the piglet: Operationalizing the concept of an essential circulation. Pediatr Res 35: 649–656.
Patel J, Pryds O, Roberts I, Harris D, Edwards AD 1996 Limited role for nitric oxide in mediating cerebrovascular control of newborn piglets. Arch Dis Child 75: F82–F86.
Author information
Authors and Affiliations
Additional information
Supported in part by U.S. Public Health Service Grant HL 35842 and by the Netherlands Organization for Scientific Research (NWO). F.v.B. is a recipient of the Ter Meulenfund of the Netherlands Academy of Arts and Sciences.
Rights and permissions
About this article
Cite this article
Van Bel, F., Sola, A., Roman, C. et al. Perinatal Regulation of the Cerebral Circulation: Role of Nitric Oxide and Prostaglandins. Pediatr Res 42, 299–304 (1997). https://doi.org/10.1203/00006450-199709000-00009
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199709000-00009