Abstract
The effect of orally administered IGF-I on intestinal development was assessed in piglets. Cesarean-derived, colostrum-deprived piglets received formula alone or formula containing 65 nM (500 μg/L) of recombinant human IGF-I. IGF-I intake averaged 200 μg/kg/d. On d 7 and 14 postpartum, piglets were killed, organs were removed and weighed, and tissue and blood samples were collected. The small intestine was divided into 13 segments that were weighed and measured. A sample of each segment was fixed in formalin, and the mucosa was scraped for enzyme analyses. Food intake, body and organ weights, intestinal weight, length, protein, DNA and RNA content did not differ between the treatment groups. Serum IGF-I, IGF-II, and IGF-binding protein profiles and tissue IGF-binding protein mRNA expression were also comparable between the treatment groups. In contrast, intestinal enzymes and villus height were increased by oral IGF-I. Lactase was ≈2-fold higher (p ≤ 0.05) in the jejunum and proximal ileum, and sucrase was ≈50% higher (p ≤ 0.05) in the jejunum of IGF-I-treated animals than in controls. Villus height in the terminal ileum was ≈50% greater in IGF-I-treated animals than in controls(p = 0.03). In conclusion, orally administered IGF-I at 200 μg/kg did not affect whole body or organ growth or serum IGF-I concentrations; however, intestinal disaccharidase activity and ileal villus growth were responsive to orally administered IGF-I, supporting a potential role for milk-borne IGF-I in neonatal intestinal development.
Similar content being viewed by others
Main
Colostrum and milk contain a variety of hormones and peptides that are known to stimulate cellular growth and differentiation both in vivo and in vitro(1, 2). The marked enhancement of porcine neonatal intestinal growth and development upon ingestion of milk or colostrum(3, 4) has led to the speculation that milk-borne hormones and growth factors may be partially responsible for this effect(1, 2). Among the growth factors present in milk are the IGFs. IGF-I and IGF-II are ≈7.5-kD single-chain peptides, consisting of 70 and 67 amino acids, respectively. IGF-I and -II exhibit 70% amino acid homology with each other and a 50% homology with proinsulin(5). The IGF-I amino acid sequence is highly conserved, showing 100% identity between human and porcine species(6). IGFs interact with three distinct receptors, the type I and type II IGF receptors, which have highest affinity for IGF-I and -II, respectively, and the insulin receptor(7). Both type I and II IGF receptors have been detected along the entire length of the neonatal rat and piglet intestine(8–10). In rats, receptor density was five times higher on crypt cells than on cells located on the tips of the villi(8, 10). In the piglet, binding of 125I-IGF-I to intestinal receptors was highest at birth, declined at 3 and 5 d, then recovered by d 21 postpartum(9).
Two previous studies have investigated the effects of oral administration of IGF-I to the piglet for 24(11) to 96 h(12) postpartum; however, neither study reported effects on intestinal enzyme activities. Therefore, the goal of the current study was to determine the effect of longer term (14 d) oral administration of IGF-I on intestinal histomorphology and enzyme activity as well as assessing any effects on whole body or organ growth, serum IGF, and IGFBP profiles. A dose of IGF-I (65 nM, 500 μg/L), which represents a high porcine colostrum concentration(13, 14), was used.
METHODS
Animals and diets. The animal protocol was approved by the Laboratory Animal Care Advisory Committee of the University of Illinois. Newborn, colostrum-deprived piglets (n = 24) were obtained by cesarean section of cross-bred (York-shire-Duroc) sows on d 113 of gestation(mean gestation is 113-115 d). The experiment was conducted in two replicates separated by ≈4 mo. On d 3 postpartum, piglets were administered 100 mg of iron dextran (Ferrodex™ 100; Agri Laboratories Ltd, St. Joseph, MO) by intramuscular injection. Piglets were housed individually in rack cages, which separated piglets from each other by Plexiglas partitions that are adjustable in width (Fig. 1). Piglets were separated from their waste products by rubber-coated perforated stainless steel flooring. Piglets learned to self-feed from rubber nipples mounted in the front Plexiglas partition. All piglets were fed a nutritionally complete commercial piglet formula (Advance Baby Pig Liqui-Wean, Milk Specialties Co., Dundee, IL) comprised of 25% protein, 13% fat, 48% lactose, 6.5% ash, and containing levels of vitamins, minerals, and trace elements, which meet NRC requirements for piglets(15). The powdered diet was reconstituted with deionized water to a final concentration of 183 g/L, which provided 721 kcal/L. The total daily formula intake was calculated as ≈15 mL/kg/h, which was divided into two equal feedings. For the first 4 d postpartum, formula was supplemented with purified porcine IgG (American Protein Corp., Ames, IA) at a concentration of 40 g/L on d 1 and 8 g/L on d 2-4. Formula was provided to the nipple by gravity flow via tubing attached to a 1-L enteral nutrition bag(Flexiflow Easyfeed®) provided by Ross Laboratories (Columbus, OH). Recombinant human IGF-I, generously provided by Genentech, Inc. (South San Francisco, CA), was added to the formula immediately before feeding at a concentration of 65 nM (500 μg/L). A metal pan was affixed to the Plexiglas below the nipple apparatus to collect any formula wastage, which was collected in a bottle below the cage so that formula and IGF-I intake could be accurately determined. Piglets were housed in an isolated suite within the animal care facilities. Airflow to each suite was individually controlled, and the air was high efficiency particulate air-filtered. Room temperature was maintained at 30 °C with 12-h light/dark cycle. Additional heat was provided for the piglets by radiant heaters located at the tops of the cages to maintain a local temperature of ≈34 °C.
Cage for housing piglets. Piglets were housed individually in rack cages that held six piglets per rack. Piglets were separated from each other by solid Pexiglas partitions and from their waste products by rubber-coated perforated stainless steel flooring. A nipple feeding apparatus attached to the front of the cage allowed piglets to self-feed at libitum, and a small metal pan below the nipple collected wastage into a plastic bottle below, so that accurate formula and IGF-I intake measurements could be obtained.
Sample collection. On d 7 and 14 postpartum, piglets were killed by electrocution and exsanguination. In addition, 7 (n = 3) and 14-d-old piglets (n = 3) were obtained from litters housed at the University of Illinois Swine Research Center to serve as sow-reared controls. These piglets were sampled approximately 1 y after the first replicate; however, they were genetically identical and were from similar parity sows. Blood samples were collected in nonheparinized tubes, and serum was obtained by centrifugation at 3500 × g for 15 min at 4°C. Visceral organs were removed and weighed, and samples were frozen in liquid nitrogen. The entire small intestine from the pyloric sphincter to the ileocecal valve was removed. The intestine was separated from the mesentery and laid out in five turns so that it formed six segments of equal length, as described by Zijlstra et al.(16). The small intestine was divided into 13 segments of approximately equal length, corresponding to the duodenum (segments 1 and 2), jejunum (segments 3-10), and ileum (segments 11-13). Segments were flushed with ice-cold saline, weighed, and measured. A 2-cm sample from each segment was fixed in buffered surgical grade 10% formalin (Surgipath Medical Industries, Inc., Richmond, IL). The remaining segment was opened longitudinally, and mucosa was scraped from the lumenal surface using a glass microscope slide. Mucosal samples were immediately frozen in liquid nitrogen.
Plasma analyses. IGF-I and IGF-II RIA. Serum IGF-I and -II concentrations were measured by heterologous RIA as previously described(13). To dissociate the IGF-I from IGFBP, plasma samples (0.5 mL) were separated over a 0.9 × 100-cm column containing Sephadex G-50 (fine) in 0.2 mol/L formic acid (Pharmacia Biotech Inc., Piscataway, NJ)(13). Fractions containing the IGF-I peptide were collected into 50-mL tubes containing 0.25 mL of RIA buffer(0.03 mol/L sodium phosphate, 0.25% BSA, 0.02% sodium azide, pH 7.5) were frozen and lyophilized (Flexi-Dry, FTS Systems, Stone Ridge, NY). IGF-I recovery from the column has been previously determined in our laboratory to be >90%. The lyophilized IGF peptide fractions were resolubilized in RIA buffer and diluted 1:4 before assay. IGF-I was measured using125 I-recombinant human IGF-I as the radioligand and a polyclonal anti-human IGF-I antibody distributed through the National Hormone and Pituitary Program. IGF-II was measured using 125I-recombinant human IGF-II as radioligand and a monoclonal anti-rat IGF-II antibody (Upstate Biotechnology Inc., Lake Placid, NY). After an overnight incubation, bound radioactivity was precipitated by centrifugation at 3000 × g for 30 min after the addition of 250 μL of 1% bovine IgG (Sigma Chemical Co., St. Louis, MO) and 1.0 mL of 20% polyethylene glycol (Sigma). For each peptide, samples were analyzed in a single assay with intraassay coefficients of variation of 3% for IGF-I and 5% for IGF-II.
SDS-PAGE and Western ligand blot analysis of plasma IGFBP. Molecular forms of serum IGFBP were characterized by Western ligand blotting as previously described(13). Plasma samples (4 μL) were mixed with sample buffer (10% glycerol, 2% SDS, and 0.625 mol/L Tris-HCl, pH 6.8), and proteins were separated through 4% stacking (0.1% SDS, 0.125 mol/L Tris-HCl, pH 6.8) and 12% running (0.1% SDS, 0.375 mol/L Tris-HCl, pH 8.8) polyacrylamide gels at 65 V, 4 °C overnight (Hoefer Scientific Instruments, San Francisco, CA). Proteins were transferred to nitrocellulose(0.45 μm, Micron Separations Inc., Westborough, MA) at 200 mA for 1 h using a Buchler semidry blotting unit (Labconco Corp., Kansas City, MO). Membranes were sequentially blocked with TBS (0.15 mol/L sodium chloride, 0.01 mol/L Tris-HCl, pH = 7.4) containing 3% Tergitol NP-40 (Sigma), TBS containing 1% BSA (Sigma), and TBS containing 0.1% Tween (Sigma), then were incubated overnight with 0.45 μCi of 125I-IGF-I. IGFBP were visualized by autoradiography (Eastman Kodak, Rochester, NY) at -70 °C for 4 d. Bands were quantified using the Foto/Analyst II Visionary System and Collage software (Fotodyne, New Berlin, WI).
RNA extraction and Northern analysis. Total cellular RNA was isolated from liver (50 mg) by the guanidinium isothiocyanate-phenol chloroform method(17) using a commercial reagent(Tri-Reagent, Molecular Research Center, Inc., Cincinnati, OH). RNA purity yield was determined spectrophoto-metrically by the ratio of absorbance of the sample at 260 and 280 nm. RNA quality was verified by agarose gel electrophoresis, staining of the 18 and 28 S ribosomal RNA with ethidium bromide, and visualization by UV transillumination. RNA samples (15 μg) were denatured by heating at 65 °C for 10 min in loading buffer [1 × MOPS buffer (0.020 mol/L MOPS, 0.005 mol/L sodium acetate and 0.0005 mol/L EDTA, pH 7.0), 50% formamide, and 2.2 M formaldehyde] and were subsequently separated on a 1.2% agarose-formaldehyde gel by electrophoresis at room temperature for 4-5 h at 100 V. The separated RNA was capillary transferred to nitrocellulose (0.45 μm, Micron Separations Inc.) using 10 × SSC buffer (1.5 M NaCl, 0.15 sodium citrate). The blot was then baked for 1 h at 80 °C and was hybridized overnight with [32P]dCTP labeled cDNA probes for rat IGFBP-1(18), IGFBP-2(19), and IGFBP-4(20) in hybridization buffer (1% BSA, 7% SDS, 0.45 mol/L sodium phosphate, 0.25 mol/L sodium chloride, and 0.001 mol/L EDTA, pH 7.2). Blots were hybridized with a cDNA for β-actin (American Type Culture Collection, Bethesda, MD) as a control for loading differences. IGFBP mRNA were visualized by autoradiography for 3-7 d at -70 °C, and bands were quantified using the Foto/Analyst II Visionary System and Collage software (Fotodyne, New Berlin, WI).
Intestinal analyses. Mucosal RNA, DNA, and protein content. RNA and DNA were determined by the method of Schmidt and Thannhauser(21), as modified by Munro and Fleck(22). DNA content was determined by the indole procedure as described by Ceriotti(23). Mucosa (25-50 mg) was homogenized in 8.0 mL of distilled water, and the nucleic acids were precipitated by the addition of 4.0 mL of 0.6 N PCA. The homogenate was centrifuged at 3,000 × g for 15 min, and the supernatant was discarded. Pellets were washed twice with 4.0 mL of 0.2 N PCA and then solubilized in 6.0 mL of 0.3 N KOH for 1 h at 37 °C. Next, 2.0 mL of solubilized solution and 1.2 mL of 1.2 N PCA were added to duplicate test tubes. After standing in ice 10 min, precipitated DNA was pelleted by centrifugation at 3000 × g for 10 min. Supernatants were saved, and the pellets were washed in 3.0 mL of 0.2 N PCA and recentrifuged. The supernatants were combined, and RNA content was determined spectrophotometrically at 250 nm (Beckman DU-64, Arlington Heights, IL). The remaining pellets were resolubilized with 1.0 mL of 0.3 N KOH in a 37 °C water bath for 30 min. Distilled water (2 mL) was added to each tube, followed by a 30-min incubation at 37 °C. Then 1 mL of 0.04% indole (4 °C) and 1.0 mL of concentrated HCl were added to each tube, and the tubes were heated in a boiling water bath for 20 min. Samples were then extracted twice with 4.0 mL of chloroform and centrifuged at 1000 × g for 10 min, and DNA content in the resulting supernatant was measured at 490 nm. Mucosal protein content was determined by a modification of the Lowry protein assay(24).
Disaccharidase activity. Mucosal scrapings (200 mg) from each segment were homogenized in 2 mL of homogenization buffer containing protease inhibitors (0.45 mol/L sodium chloride, 0.001 mol/L phenylmethylsulfonyl fluoride, 0.002 mol/L iodoacetic acid). Sucrase (EC 3.2.1.10)- and lactase (EC 3.2.1.23-62)-specific activities were determined by the method of Thulin et al.(25). Enzyme activities were determined by incubating intestinal homogenates with the appropriate disaccharide(lactose or sucrose) for 30 min at 37 °C. Liberated glucose was assayed by coupling the reaction to glucose-6-phosphate dehydrogenase and measuring the formation of NADPH at 340 nm. Leucine aminopeptidase (EC 3.4.11.1) activity was determined using a commercially available kit (Procedure No. 251, Sigma) based upon the method of Martinek et al.(26). Enzyme activities were expressed on the basis of homogenate protein content.
Histomorphology. Cross-sections of the formalin-fixed odd-numbered intestinal segments were embedded in paraffin, thinly sliced with a microtome, and stained with hematoxylin. Crypt depth, villus height, crypt-villus ratios, and muscularis thickness were measured using a Videometric 150 image analysis system (American Innovision, Inc., San Diego, CA).
Statistical analysis. Analyses were performed using the general linear modeling procedure within SAS (Version 6.09, SAS Institute, Cary, NC), nesting each segment within treatment and using least squares means to obtain a p value for comparison between control and treatment groups for each segment. Significance was assigned at p < 0.05. All data are expressed as mean ± SD.
RESULTS
Formula and IGF-I intake. Mean formula intake per kg of body weight was relatively constant from d 3 to 14 postpartum and did not differ between the treatment groups (control piglets 382 ± 37 mL/kg/d; IGF-I piglets 381 ± 27 mL/kg/d). We previously determined that the sow milk replacer is devoid of endogenous IGF-I and IGFBP(24). The purified IgG preparation that was added to the milk replacer on the first 4 d postpartum did contain low levels of IGF-I. The 40 g/L solution provided on d 1 postpartum contained 0.95 nmol/L (7.2 μg/L) IGF-I and the 8 g/L solution provided on d 2-4 postpartum contained 0.23 nmol/L (1.74 μg/L), which resulted in a mean IGF-I intake of 0.10 ± 0.02 nmol/kg/d (0.73± 0.14 μg/kg/d) in the control piglets on d 1-4 postpartum. Mean IGF-I intake of the IGF-I supplemented group was 26.2 ± 2.6 nmol/kg/d(198 ± 19.7 μg/kg/d) from d 1 to 14 postpartum.
Body and organ growth. Initial body weight, body weight gain(g/d), or final body weights did not differ between the treatment groups(Table 1). Body weights of sow-reared piglets on d 7 and 14 postpartum were comparable to formula-fed piglets. Organ weights of formula-fed piglets were unaffected by oral IGF-I; however, differences between sow-reared and formula-fed piglets were observed. Formula-fed piglets had heavier kidneys on d 7 and kidneys, liver, and spleen on d 14 compared with sow-reared piglets. Intestinal weight (g/kg) and length (cm/kg) were unaffected by oral IGF-I; however, intestines of sow-reared piglets were longer on d 7 and lighter on d 14 than were formula-fed piglets.
Serum IGF-I, IGF-II, and IGFBP profiles. Serum IGF-I and -II concentrations of sow-reared and formula-fed piglets on d 7 and 14 postpartum are summarized in Table 2. Serum IGF-I and -II concentrations were similar to those previously reported in the literature(12, 28, 29). No effect of oral IGF-I on serum IGF-I or -II concentrations were apparent either on d 7 or 14. In addition, there were no significant differences in serum IGF-I or -II concentrations between the two formula-fed groups compared with the sow-reared piglets.
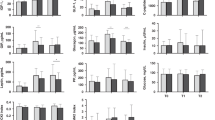
Representative Western ligand blots of serum IGFBP profiles of sow-reared and formula- and formula + IGF-I- fed piglets on d 7 and 14 are shown in Figure 2, and densitometric analyses of the IGFBP bands are summarized in Table 3. Five IGFBP bands were apparent at 48, 46, 32.5, 28, and 24 kD. The serum IGFBP are comparable to those reported in neonatal piglets by McCusker et al.(29). The 48- and 46-kD bands represent two glycosylated variants of IGFBP-3. The 32.5- and 28-kD bands have been immunologically identified as IGFBP-2 and IGFBP-1, respectively(29). The 24-kD band most likely represents IGFBP-4 based on the similar molecular mass to IGFBP-4, which has been purified from porcine follicular fluid(30). No differences in IGFBP-1 were noted among the groups at either d 7 or 14. On both d 7 and 14, IGFBP-2 was higher in sow-reared piglets than both artificially fed groups, whereas IGFBP-3 at d 4 was lower in sow-reared than the formula + IGF-I group.
Western ligand blot of plasma IGF binding proteins. A representative autoradiograph of a Western ligand blot of serum IGFBP in sow-reared piglets and piglets fed formula ± IGF-I for 7(top) or 14 (bottom) days postpartum. Plasma samples (4μL) were separated by SDS-PAGE, Western ligand blotted using125 I-IGF-I, and exposed to film for 4 d. Bands with apparent molecular masses of 48, 46, 32.5, 28, and 24 kD were visualized. Densitometric analysis of autoradiographs is summarized in Table 3.
Tissue IGFBP mRNA expression. Hepatic IGFBP mRNA expression was examined to determine whether oral IGF-I influenced IGFBP expression (Fig. 3). IGFBP-1 was expressed at very low levels, was detected only in formula-fed piglets on d 7 postpartum, and did not differ between two treatment groups (Table 4). IGFBP-2 mRNA was greater in sow-reared piglets than in piglets fed formula + IGF-I on d 7 postpartum (p < 0.05). No effect of IGF-I on IGFBP-2 or -4 expression in formula-fed piglets at either d 7 or 14 was detected.
Hepatic IGFBP mRNA expression. Autoradiographs of Northern blots of hepatic IGFBP-1, -2, and -4, and β-actin expression in 7- and 14-d postpartum sow-reared piglets and piglets fed formula ± IGF-I. Bands were visualized by autoradiograph for 3-7 d, and densitometric analyses of autoradiographs are as summarized in Table 4.
Intestinal RNA, DNA, and protein content. RNA, DNA, and protein content in the duodenum (segment 1), jejunum (segments 3, 5, 7, and 9) and ileum (segments 11 and 13) of control and IGF-I-treated piglets are summarized in Table 5. Statistical comparisons were limited to within segment comparisons at each age. No significant effect of orally administered IGF-I on mucosal RNA, DNA, or protein content was detected.
Intestinal enzyme activities. Sucrase- and lactase-specific activities were determined throughout the intestine of d 7 and 14 postpartum sow-reared and formula-fed piglets. Sucrase activity (Fig. 4, a and b) was highest in the distal duodenum and proximal jejunum and increased ≈2-fold between d 7 and 14 (p < 0.05). On d 7, sucrase activity in the proximal jejunum (segment 3) of sow-reared piglets was higher than in piglets fed formula alone (p < 0.05), but not in piglets fed formula + IGF-I. In intestinal segment 4, sucrase activity of sow-reared piglets was higher than in both formula-fed groups (p< 0.05) (Fig. 4a). By d 14, the highest sucrase activity was observed in piglets fed formula + IGF-I. Mean sucrase activity was higher in segments 3 through 7 of IGF-I-treated piglets than in piglets fed formula alone, whereas sucrase activity in sow-reared piglets was intermediate between the two formula-fed groups (Fig. 4b).
Small intestinal sucrase specific activity. Mean sucrase-specific activity (μmol glucose/min/g protein) throughout the small intestine of sow-reared, formula-fed, and formula + IGF-I fed piglets on d 7(a) and d 14 (b) postpartum. Segments 1 and 2 represent duodenum, segments 3-10 jejunum and 11-13 represent ileum. Different superscripts among the treatment groups within each segment indicate significant differences at p < 0.05.
Lactase activity (Fig. 5, a and b) was relatively evenly distributed throughout the piglet duodenum and jejunum, with lower activity observed in the ileum (segments 11-13). On d 7, lactase activity was higher in intestinal segments 2, 3, 4, 6, and 10 of sow-reared piglets than of formula-fed piglets (p < 0.05). No effect of oral IGF-I on lactase activity was observed on d 7 (Fig. 5a). By d 14, however, lactase activity in piglets receiving oral IGF-I was higher (p ≤ 0.05) throughout the jejunum and ileum, with the exception of segment 6, than in piglets fed formula alone (Fig. 5b). Lactase activity on d 14 postpartum in piglets fed formula + IGF-I was comparable to d 7 levels, whereas lactase activity in control animals had declined ≈33%. In addition, lactase activity of IGF-I-treated piglets was similar to that of sow-reared piglets. It is interesting to note that lactase activity in sow-reared piglets was biphasic, with peaks observed in the duodenum and proximal jejunum (segments 2 and 3) and in the distal jejunum (segments 8-10). This pattern of lactase expression had been previously observed in sow-reared piglets(32), but was not apparent in either formula-fed group.
Small intestinal lactase-specific activity. Mean lactase specific activity (μmol glucose/min/g protein) throughout the small intestine of sow-reared, formula-fed, and formula + IGF-I-fed piglets on d 7(a) and d 14 (b) postpartum. Segments 1 and 2 represent duodenum, segments 3-10 jejunum, and 11-13 ileum. Different superscripts among the treatment groups within each segment indicate significant differences at p < 0.05.
Intestinal histomorphology. Intestinal morphology of formula-fed piglets was assessed on d 14 postpartum (Fig. 6). Mean villus height in the terminal ileum (segment 13) was greater(p = 0.03) in IGF-I-treated piglets than in piglets fed formula alone. There was also a tendency (p = 0.11) for villus length to be increased more proximally in the ileum (segment 11), whereas no effect of oral IGF-I on villus height in any other region of the intestine was detected. In addition, no differences in crypt depth, crypt-to-villus ratio, or muscularis thickness were observed.
DISCUSSION
The intestinal structure and digestive enzyme development of the neonatal piglet is comparable to that of the human infant(33, 34), making it an ideal model to study the potential role of milk-borne growth factors in the neonate. Previous studies in piglets have shown that consumption of formula supplemented with either insulin(35, 36) or epidermal growth factor(16) stimulated intestinal enzyme activity. The ability of IGF-I to modulate intestinal enzyme activity in piglets has not been previously investigated; however, oral administration of IGF-I for 6 d to neonatal rats increased intestinal disaccharidase activity(10), suggesting IGF-I may also be a mediator of intestinal enzyme activity. A recent study in neonatal piglets fed 458 nmol of IGF-I/kg of body weight (3500 μg/kg) demonstrated significant increases in small intestinal mucosal growth compared with piglets fed formula alone(12). This dose compares with the estimated IGF-I intake of 5-100 μg/kg in piglets consuming mature porcine milk or colostrum, respectively(12). The current study was designed to determine the effect of oral administration of a high porcine colostral concentration of IGF-I (65 nM or 500 μg/L)(14, 15) on intestinal development in the piglet. The ingested dose of IGF-I in this study was approximately twice that of colostrum, 26 nmol of IGF-I/kg of body weight/d (200 μg/kg/d); however, it is important to note that piglets were exposed to this dose throughout the 14-d study, whereas a naturally suckling piglet would consume this amount of IGF-I for only the first 1-2 d postpartum.
Final body weight, growth rate, and organ weights were unaffected by oral IGF-I, which is consistent with previous studies in which orally administered IGF-I exerted little or no systemic effect(11, 12). In the study of Burrin et al.(12) there was a tendency (p < 0.10) for piglets fed 458 nmol of IGF-I/kg/d to gain more weight and have heavier livers and spleens(p < 0.10) than did control piglets. However, due to the level of IGF-I that was administered in that study, it is unlikely that IGF-I at porcine milk concentrations is a significant regulator of whole body growth in the neonatal pig.
No differences in serum IGF-I or IGF-II concentrations were noted between the two formula-fed groups or when compared with sow-reared piglets. This lack of effect of oral IGF-I administration on serum IGF-I concentration is consistent with the findings of Burrin et al.(12) and supports the findings of two recent studies in which 125I-IGF-I was orally administered to neonatal piglets(27) or rats(37). We administered 25 μCi (100 ng) of 125I-IGF-I to newborn colostrum-deprived piglets and collected blood samples for 4 h(27). Approximately 3-5% of the radioactivity detected in serum was immunoreactive with an IGF-I antibody; however, it was calculated that the radioactive 125I-IGF-I contained in serum, 4 h after oral administration, constituted less that 0.025% of the circulating pool of IGF-I in the piglets(27). Similarly, less than 4% of a dose of125 I-IGF-I was recovered in serum 30 min after orogastric administration to suckling rats(37). In the same study, over 40% of the orally administered 125I-IGF-I was recovered within the gastrointestinal wall and lumenal contents, of which a significant proportion was structurally intact and receptor-active 30 min after administration, which supports the ability of IGF-I to be physiologically active within the gastrointestinal tract(37).
Serum IGFBP profiles and liver IGFBP mRNA expression were also determined. Serum IGFBP are thought to mediate IGF action by limiting the interaction of IGF-I with its cell surface receptors and incresing its circulating half-life(38). In the piglet, serum IGFBP profiles and hepatic IGFBP mRNA expression undergo distinct ontogenic changes(28), and it was unknown whether milk-borne IGF-I affected the normal developmental expression of IGFBP. Consistent with the IGF-I and -II concentrations, no effect of oral IGF-I on circulating IGFBP profiles was observed among the formula-fed piglets. However, serum IGFBP profiles between sow-reared and formula-fed piglets did differ. On d 7 and 14, IGFBP-2 was higher and IGFBP-3 was lower in sow-reared than in formula-fed piglets. Nutritional status and GH are two factors that are known to regulate serum IGFBP. Malnutrition leads to a reduction in serum IGFBP-3 and an increase in serum IGFBP-1 and -2, whereas GH raises serum IGFBP-3 and down-regulates IGFBP-1 and -2(29, 31, 38). It is possible that sow-reared piglets had lower serum GH concentrations than formula-fed piglets; however, this remains to be determined. On d 7, hepatic IGFBP-2 mRNA was highest in sow-reared piglets, which is consistent with the increased levels of serum IGFBP-2 in these piglets compared with formula-fed piglets. By d 14, however, IGFBP-2 mRNA was lowest in sow-reared piglets, although their serum IGFBP-2 remained higher than the other two groups.
With respect to intestinal growth, results from this and previous studies(11, 12) suggest that the magnitude of the response to IGF-I is affected both by the dose administered and the duration of the exposure. No significant differences in intestinal weight and length, protein, or DNA or RNA content were observed in this study or in a previous study in which piglets were administered 58 nmol of IGF-I/kg/d for 24 h(11). The 24-h treatment was likely insufficient to detect an increase in mucosal mass, because an increase in bromodeoxyuridine labeling in the crypts of IGF-I- and IGF-II-treated piglets was observed, suggesting that a longer period of treatment could have resulted in significant increases in intestinal villus growth(11). This was clearly demonstrated by the study of Burrin et al.(12) in which consumption of 458 nmol/kg/d for 4 d markedly increased small intestinal weight, protein, and DNA content compared with piglets fed unsupplemented milk replacer(12). Although overall small intestinal weight and length were not increased in response to oral IGF-I in the current study, villus height was increased by 40-60% in the distal ileum of IGF-I-treated animals, suggesting that IGF-I was promoting intestinal growth at the cellular level. Burrin et al. also reported significant increases in villus height in both the jejunum (174% of control) and ileum (134% of control) of piglets fed 458 nmol of IGF-I/kg/d. It is interesting to note that, in the current study and that of Burrin et al.(12), the growth-promoting effect of orally administered IGF-I was limited to increasing villus height; crypt depth and muscularis thickness did not differ between the treatment groups.
This study is the first to demonstrate a positive effect of oral IGF-I on intestinal disaccharidase activity in the piglet. The specific activities of sucrase in the jejunum and lactase in the jejunum and proximal ileum were elevated in piglets receiving IGF-I compared with that of controls. These results are consistent with those of Young et al.(10), in which low doses of IGF-I (1 μg/d) administered orally to neonatal rats increased jejunal brush border lactase and sucrase activity, without stimulating intestinal cellular growth. Likewise, orally administered insulin has been shown to stimulate lactase- and maltase-specific activity in neonatal piglets(34, 35). The mechanism underlying the increase in intestinal disaccharidases in response to orally administered IGF-I remains to be elucidated, but could involve direct modulation of lactase gene expression or the posttranslational processing of the enzyme(39). Alternatively, it is possible that IGF-I could indirectly enhance intestinal disaccharidase activity either by increasing cell number or increasing the residence time of fully differentiated villus cells. This could be achieved either by enhancing mitotic activity in the crypts(11) or by inhibiting apoptosis(40, 41).
In summary, orally administered IGF-I exerts local effects within the intestine. It is likely that providing IGF-I in a milk-based formula was advantageous, because casein protects IGF-I from gastric and intestinal digestive secretions(42). Orally administered IGF-I does not influence circulating concentrations of IGF-I, even at pharmacologic doses(12). In addition, the inability of colostral levels of IGF-I (500 μg/L) consumed for 14 d to influence body weight or organ weights suggests that milk-borne IGF-I is not a major regulator of neonatal growth. Within the intestine, ingestion of 26 nmol/kg/d for 14 d increased ileal growth and sucrase and lactase activities in piglets without significantly increasing small intestinal weight, length, protein, or DNA content. These results imply that orally administered IGF-I may be efficacious in stimulating neonatal gastrointestinal development and that its actions would be limited to the intestine without exerting undersirable systemic effects.
Abbreviations
- IGFBP:
-
insulin-like growth factor-binding protein
- MOPS:
-
morpholinopropanesulfonic acid
- PCA:
-
perchloric acid
- TBS:
-
Tris-buffered saline
References
Grosvenor CE, Picciano MF, Baumrucker CR 1993 Hormones and growth factors in milk. Endocr Rev 14: 710–728.
Donovan SM, Odle J 1994 Growth factors in milk as mediators of infant development. Annu Rev Nutr 14: 147–167.
Widdowson EM, Colombo VE, Artavanis CA 1976 Changes in the organs of pigs in response to feeding for the first 24 h after birth. II. The digestive tract. Biol Neonate 28: 272–281.
Burrin DG, Shulman RJ, Reeds PJ, Davis TA, Gravitt KR 1992 Porcine colostrum and milk stimulate visceral organ and skeletal muscle protein synthesis in neonatal piglets. J Nutr 122: 1205–1213.
Rechler MM, Nissley SP 1991 Insulin-like growth factors. In: Sporn MB, Roberts AB, Hanson LA (eds) Peptide Growth Factors and Their Receptors, Vol 1. Springer-Verlag, New York, pp 263–368.
Tavakkol A, Simmen FA, Simmen RCM 1988 Porcine insulin-like growth factor-I (pIGF-I): complementary deoxyribonucleic acid cloning and uterine expression of messenger ribonucleic acid encoding evolutionarily conserved IGF-I peptides. Mol Endocrinol 2: 674–681.
Oh Y, Müller HL, Neely EK, Lamson G, Rosenfeld RG 1993 New concepts in insulin-like growth factor receptor physiology. Growth Regul 3: 113–123.
Laburthe M, Rouyer-Fessard C, Gammeltoff S 1988 Receptors for insulin-like growth factor I and II in rat gastrointestinal epithelium. Am J Physiol 254:G457–G462.
Schober DA, Simmen FA, Hadsell DL, Baumrucker CR 1990 Perinatal expression of type I IGF receptors in porcine small intestine. Endocrinology 126: 1125–1132.
Young GP, Taranto TM, Jones HA, Cox AJ, Hogg A, Werther GA 1990 Insulin-like growth factors in the developing and mature rat small intestine: receptors and biological actions. Digestion 46: 240–252.
Xu R-J, Mellor DJ, Birtles MJ, Breier BH, Gluckman PD 1994 Effects of oral IGF-I or IGF-II on digestive organ growth in newborn piglets. Biol Neonate 66: 280–287.
Burrin DG, Wester TJ, Davis TA, Amick S, Heath JP 1996 Orally administered insulin-like growth factor-I increases intestinal mucosal growth in formula-fed neonatal pigs. Am J Physiol 270:R1085–R1091.
Donovan SM, McNeil LK, Jiménez-Flores R, Odle J 1994 Insulin-like growth factors and IGF binding proteins in porcine serum and milk throughout lactation. Pediatr Res 36: 159–168.
Simmen FA, Simmen RCM, Reihnart G 1988 Maternal and neonatal somatomedin-C/insulin-like growth factor-I (IGF-I) and IGF binding proteins during early lactation in the pig. Dev Biol 130: 16–20.
National Research Council 1988 Nutrient Requirements of Swine, 9th Ed. National Academy of Sciences Press, Washington, DC
Zijlstra RT, Odle J, Hall WF, Petschow BW, Gelberg HB, Litov RE 1994 Effect of orally administered epidermal growth factor on intestinal recovery of neonatal pigs infected with rotavirus. J Pediatr Gastroenterol Nutr 19: 382–390.
Chomczynski P 1993 A reagent for the single-step simultaneous isolation of RNA, DNA and proteins from cell and tissue samples. BioTechniques 15: 532–537.
Murphy LJ, Seneviratne C, Ballejo G, Croze F, Kennedy TG 1990 Identification and characterization of a rat decidual insulin-like growth factor binding protein complementary DNA. Mol Endocrinol 4: 329–336.
Margot JB, Binkert C, Mary J-L, Landwehr J, Heinrich G, Schwander J 1989 A low molecular weight insulin-like growth factor binding protein from rat: cDNA cloning and tissue distribution of its messenger RNA. Mol Endocrinol 3: 1053–1060.
Shimasaki S, Uchiyama F, Shimonaka M, Ling N 1990 Molecular cloning of the cDNAs encoding a novel insulin-like growth factor binding protein (IGFBP-4) from rat and human. Mol Endocrinol 4: 1451–1458.
Schmidt G, Thannhauser SJ 1945 A method for the determination of deoxyribonucleic acid, ribonucleic acid, and phosphoproteins in animal tissues. J Biol Chem 161: 83–89.
Munro HN, Fleck A 1969 Analysis of tissues and body fluids for nitrogenous constituents. In: Munro HN, Allison JB (eds) Mammalian Protein Metabolism, Vol 3. Academic Press, New York, pp 423–525.
Ceriotti G 1952 A microchemical determination of deoxyribonucleic acid. J Biol Chem 198: 297–303.
Peterson GL 1977 A simplification of the protein assay method of Lowry et al. Anal Biochem 83: 346–356.
Thulin JD, Kuhlenschmidt MS, Gelberg HB 1991 Development, characterization and utilization of an intestinal xenograft model for infectious disease research. Lab Invest 65: 719–726.
Martinek RG, Berger L, Broida D 1964 Simplified estimation of leucine aminopeptidase (LAP) activity. Clin Chem 10: 1087–1092.
Donovan SM, Chao JC-J, Zijlstra RT, Odle J 1997 Orally administered iodinated recombinant human insulin-like growth factor-I(125I-rhIGF-I) is poorly absorbed by the neonatal piglet. J Pediatr Gastroenterol Nutr 24: 174–182.
Lee CY, Bazer FW, Etherton TD, Simmen FA 1991 Ontogeny of insulin-like growth factors (IGF-I and IGF-II) and IGF-binding proteins in porcine serum during fetal and postnatal development. Endocrinology 128: 2336–2334.
McCusker RH, Cohick WS, Busby WH, Clemmons DR 1991 Evaluation of the developmental and nutritional changes in porcine insulin-like growth factor-binding protein-1 and -2 serum levels by immunoassay. Endocrinology 129: 2631–2638.
Shimasaki S, Shimonaka M, Zhang H-P, Ling N 1991 Isolation and molecular characterization of three novel novel insulin-like growth factor binding proteins (IGFBP- 4, 5 and 6). In: Spencer EM (ed) Modern Concepts of Insulin-like Growth Factors. Elsevier, New York, pp 343–358.
Zhao X, Unterman TG, Donovan SM 1995 Human growth hormone but not insulin-like growth factor-I enhances recovery from neonatal malnutrition in rats. J Nutr 125: 1316–1327.
Manners MJ, Stevens JA 1972 Changes from birth to maturity in the pattern of distribution of lactase and sucrase activity in the mucosa of the small intestine of pigs. Br J Nutr 28: 113–127.
Aumaitre A, Corring T 1978 Development of digestive enzymes in the piglet from birth to 8 wk. Nutr Metab 22: 244–253.
Moughan PJ, Birtles MJ, Cranwell PD, Smith WC, Pedraza M 1992 The piglet as a model animal for studying aspects of digestion and absorption in milk-fed human infants. World Rev Nutr Diet 37: 40–113.
Shulman RJ 1990 Oral insulin increases small intestinal mass and disaccharidase activity in the newborn miniature pig. Pediatr Res 28: 171–175.
Shulman RJ, Tivey DR, Sunitha I, Dudley MA, Henning SJ 1992 Effect of oral insulin on lactase activity, mRNA, and posttranslational processing in the newborn pig. J Pediatr Gastroenterol Nutr 14: 166–172.
Philipps AF, Radhakrishna R, Anderson GG, McCracken DM, Lake M, Koldovsky O 1995 Fate of insulin-like growth factors I and II administered orogastrically to suckling rats. Pediatr Res 37: 586–592.
Bach LA, Rechler MM 1995 Insulin-like growth factor binding proteins. Diabetes Rev 3: 38–61.
Burrin DG, Dudley MA, Reeds PJ, Shulman RJ, Perkinson S, Rosenberger J 1994 Feeding colostrum rapidly alters enzymatic activity and the relative isoform abundance of jejunal lactase in neonatal pigs. J Nutr 124: 2350–2357.
Hermiston ML, Gordon JI 1995 Organization of the crypt-villus axis and evolution of its stem cell hierarchy during intestinal development. Am J Physiol 268:G813–G822.
Sell CR, Baserga R, Rubin R 1995 Insulin-like growth factor-I and the IGF-I receptor prevent etoposide-induced apoptosis. Cancer Res 55: 303–306.
Xian CJ, Shoubridge CA, Read LC 1995 Degradation of IGF-I in the adult rat gastrointestinal tract is limited by a specific antiserum or the dietary protein casein. J Endocrinol 146: 215–225.
Acknowledgements
The authors thank Cliff Shipley, D.V.M., of the University of Illinois School of Veterinary Medicine for performing cesarean sections and Ruurd Zijlstra for invaluable technical assistance.
Author information
Authors and Affiliations
Additional information
Supported by National Institutes of Health Grant RO1 HD29264 (S.M.D.).
Rights and permissions
About this article
Cite this article
Houle, V., Schroeder, E., Odle, J. et al. Small Intestinal Disaccharidase Activity and Ileal Villus Height Are Increased in Piglets Consuming Formula Containing Recombinant Human Insulin-Like Growth Factor-I. Pediatr Res 42, 78–86 (1997). https://doi.org/10.1203/00006450-199707000-00013
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199707000-00013
This article is cited by
-
Glucose-regulatory hormones and growth in very preterm infants fed fortified human milk
Pediatric Research (2024)
-
Recombinant IGF-1/BP3 protects against intestinal injury in a neonatal mouse NEC model
Pediatric Research (2024)
-
Assessment trial of the effect of enteral insulin on the preterm infant intestinal microbiota
Pediatric Research (2023)
-
Place and direction learning in a spatial T-maze task by neonatal piglets
Animal Cognition (2012)
-
Mammary Specific Transgenic Over-expression of Insulin-like Growth Factor-I (IGF-I) Increases Pig Milk IGF-I and IGF Binding Proteins, with no Effect on Milk Composition or Yield
Transgenic Research (2005)