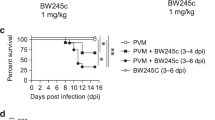
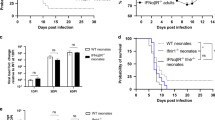
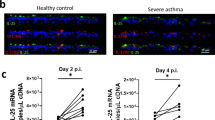
Abstract
Respiratory viral infections have been associated with airway obstruction and hyperresponsiveness, and exacerbations of asthma. Although virus-induced asthma is thought to be precipitated by airway inflammation, the clinical efficacy and rationale for using antiinflammatory treatment during such exacerbations remains controversial. The purpose of this study was to use a well characterized animal model of respiratory viral illness to test the hypothesis that the inflammatory response to viral infection is responsible for the development of airway dysfunction. Adult rats were inoculated with either Sendai virus or sterile vehicle and treated with daily injections of dexamethasone or saline. At postinoculation d 4, 5, or 6, rats were evaluated for airway obstruction, hyperresponsiveness, inflammation, and lung viral titers. Saline-treated infected rats had significant airway obstruction(increased resistance, decreased dynamic compliance), hyperresponsiveness(i.v. methacholine), and inflammation (increased bronchoalveolar lavage leukocytes) compared with noninfected controls. In contrast, dexamethasone-treated infected rats had no increase in bronchoalveolar lavage leukocytes and significantly smaller changes in airway physiology, but had increased lung viral titers compared with saline-treated infected rats. We conclude that glucocorticoid suppression of the inflammatory response to respiratory viral infection largely prevents virus-associated airway dysfunction.
Similar content being viewed by others
Main
Viral respiratory infections commonly precipitate asthma exacerbation in humans, especially in children younger than 6 y of age(1). Furthermore, even in individuals without asthma, viral respiratory infections have been associated with abnormal physiology and airway hyperresponsiveness(2–6). Glucocorticoid therapy is commonly used for prevention and treatment of virus-induced exacerbations of asthma, but controversy persists regarding its efficacy(7, 8), the uncertainty having been renewed by negative findings in a recent controlled trial in children(9).
Airway inflammation is a prominent feature of asthma(10, 11), and of respiratory viral infections(12–14). Although it is likely that virus-induced airway inflammation contributes to airway dysfunction during acute viral illness, it is not known to what extent the airway dysfunction may be attenuated by suppressing the inflammatory response. We hypothesized that airway dysfunction during acute viral illness is caused by the inflammatory response, rather than direct viral effects on airway epithelium. Previously we reported that adult rats infected with Sendai virus exhibit airway obstruction and hyperresponsiveness analogous to virus-induced airway dysfunction in humans(15). This animal model allows sufficient control of variables to address the basic mechanisms underlying virus-induced airway dysfunction. The purpose of this study was to test whether suppression of inflammation, but not viral replication, with dexamethasone would attenuate virus-induced airway obstruction and hyperresponsiveness in rats.
METHODS
Preparation of animals. All methods and procedures were approved by the Animal Care and Use Committee of the University of Wisconsin. Forty-three adult male-specific pathogen-free Sprague-Dawley derived rats (CD strain, Charles River Breeding Laboratories, Raleigh, NC), weighing an average of 0.295 kg, were used for these experiments. All animals were allowed food and water ad libitum.
General experimental design. Rats were inoculated with parainfluenza type 1 (Sendai) virus or sham inoculated with sterile vehicle. Dexamethasone (0.5 mg/kg) or saline was injected s.c. daily, with the initial dose given 12 h after inoculation to avoid the potential interference with viral infection noted when steroid treatment was started before inoculation(16). Physiologic evaluations and BAL were performed on postinoculation d 4, 5, or 6, 24 h after the last dexamethasone or saline injection. Measurements included daily weights, lung mechanics (respiratory system resistance and dynamic compliance), responsiveness to i.v. methacholine, and BAL total and differential inflammatory cell counts. An additional group of six infected rats of comparable age was evaluated for viral titers and lung histology at 5 d postinoculation.
Viral inoculation. Rats were inoculated with parainfluenza type 1 (Sendai) virus strain P3193 by use of an aerosol exposure apparatus(Glas-Col, Terre Haute, IN) as described previously(15, 17–19). The apparatus introduced an aerosol of approximately 108 plaque-forming units of virus in 2 mL of chorioallantoic fluid into the animal chamber, and the rats breathed the virus-containing aerosol for 15 min. Control rats were inoculated with sterile chorioallantoic fluid in a similar manner. Animals were housed in microisolater cages after inoculation, and virus and control groups were studied at different times to ensure that control rats would have no viral exposure.
Instrumentation and measurement of pulmonary mechanics. Cannulae were placed in the trachea, femoral vein, and femoral artery of each rat after anesthesia was induced with urethane (1.2 g/kg intraperitoneally; Sigma Chemical Co., St. Louis, MO). Rats were placed in a constant-pressure rodent plethysmograph, pretreated with propranolol (2 mg/kg i.v.; Sigma Chemical Co.) to suppress adrenergic modulation of methacholine challenges, and then paralyzed with succinylcholine chloride (4 mg/kg i.v. initially and 2 mg/kg thereafter as needed; Sigma Chemical Co.). Rats were ventilated mechanically at 80 breaths/min with the tidal volume based on body weight and adjusted to maintain a normal arterial Pco2 of 4.5-6.0 kPa. Lungs were inflated to 2.9-kPa pressure each minute to prevent atelectasis, and accumulated secretions were removed from the airways by suctioning as necessary.
Resistance was calculated by the isovolume method at 50% tidal volume(model 6 Pulmonary Mechanics Analyzer, Buxco Electronics, Sharon, CT), using inflation pressure measured at the tracheal cannula, and flow and volume obtained from the plethysmograph transducer signal(15). Cdyn was determined from inflation pressure and tidal volume at points of zero flow. Pressure and flow signals were in phase to at least 7 Hz under test conditions. Adiabatic/isothermal transients were negligible in the range of flow rates encountered in these studies. Rrs was obtained from the computed resistance by subtracting the resistance of the tracheal cannula and its connector.
Methacholine challenge. Methacholine chloride (Sigma Chemical Co.) was injected as 1 mL/kg of increasing concentrations of the agent, followed by a flush of 0.3 mL of saline, both completed in 5 s. Responses to i.v. methacholine were recorded as the change in resistance after administration of the agent. Sensitivity to methacholine was determined as the log dose (nmol/kg) required to increase resistance by 20 Pa mL-1 s(PD20) by interpolation of the dose-response curve(15, 17).
Bronchoalveolar lavage and cell count. After physiologic measurements, rats were killed by exsanguination and air embolus. The thoracic cavity was then exposed with a midline incision along the sternum and the lungs were inflated to total lung capacity with Hanks' balanced salt solution without calcium and magnesium; the lavage was repeated for a total of five exchanges. Lavage fluid was centrifuged, and the cell pellet was resuspended in 1 mL of buffer. Total BAL cell counts were measured with a Hemo-W cell counter (Coulter Electronics, Hialeah, FL). Cytocentrifuge cell samples of lavage fluid were made with Cytospin (Shandon Lipshaw Inc., Pittsburgh, PA) and stained with Diff-Quik (Baxter Healthcare, Miami, FL). The differential cell count was determined from 200 stained cells.
Lung viral titers. Viral titers were measured in a separate group of six infected rats 5 d postinoculation. After exsanguination, the left mainstem bronchus was tied, and the left lung was removed, frozen in liquid N2, and stored at -80 °C. Plaque assays for infectious virus were performed on lung homogenates, using Madin-Darby bovine kidney cells(20). Right lungs were fixed by tracheal perfusion with 10% buffered formalin and were then evaluated for inflammatory changes. A semiquantitative inflammation score was assigned for each lung section in a blinded manner by a pathologist (W.L.C.), with scores ranging from 1 (mild bronchitis) to 3 (moderately severe, erosive, and suppurative bronchiolitis and interstitial pneumonia).
Data analysis. All statistical analyses were done using Systat version 5.03 software (Systat, Inc., Evanston, IL). Kruskal-Wallis one-way analysis of variance was used to detect differences among the four study groups; when significant, planned pairwise comparisons were conducted with the Mann-Whitney test. Data from postinoculation d 4, 5, and 6 were pooled for all variables except BAL lymphocytes, for which d 4 was excluded due to prior observations that virus-induced increases in lymphocytes do not occur before d 5. Mann-Whitney was also used to test for differences in viral titers. A p value ≤ 0.05 was considered to be significant.
RESULTS
Inflammatory response and viral titers. There was an increase in total BAL inflammatory cells recovered from the virus-inoculated saline-treated group compared with sham-inoculated controls, with a significant increase in the number of neutrophils and macrophages (Fig. 1). Total BAL lymphocyte counts were significantly elevated in the untreated, virus-inoculated rats on d 5 and 6 compared with untreated controls (Fig. 1). In contrast, rats treated with dexamethasone during viral infection had no significant influx of inflammatory cells into the airways, having BAL cell counts similar to those of the noninfected rats (Fig. 1).
Viral titers measured at postinoculation d 5 were 1-2 logs higher in the lungs of dexamethasone-treated rats than in lungs of untreated rats of the same inoculation batch (Fig. 2). Semiquantitative inflammation scores were lower in the dexamethasone-treated animals (Fig. 2), consistent with the BAL findings.
Pulmonary physiology. The virus-infected group had significant airway obstruction, as measured by differences in lung mechanics compared with sham-inoculated controls. Rats in the infected group had a reduction in Cdyn and an increase in Rrs (Fig. 3) compared with those of the noninfected control group. Dexamethasone treatment significantly attenuated virus associated deviations in Rrs, but it was less effective in preventing virus-induced changes in Cdyn (Fig. 3). Rrs and Cdyn were not significantly changed by dexamethasone treatment in noninfected rats (Fig. 3).
Methacholine responsiveness. Significant airway hyperresponsiveness to i.v. methacholine was present in saline-treated rats of the virus-inoculated group compared with rats of the saline-treated sham-inoculated control group (Fig. 4). Dexamethasone treatment significantly attenuated, but did not abolish, development of virus-associated airway hyperresponsiveness to methacholine (Fig. 4). Methacholine responsiveness was not significantly altered by dexamethasone treatment in noninfected rats (Fig. 4).
DISCUSSION
Respiratory viral illnesses in rats produce acute airway pathology and under some conditions also produce chronic airway abnormalities. In the 1st wk after inoculation with Sendai virus, previously healthy rats develop an acute bronchiolitis and pneumonitis characterized by neutrophilic inflammation, airway obstruction, airway hyperresponsiveness, and altered gas exchange; toward the end of the 1st wk the viral titers drop precipitously, coincident with lymphocytic infiltration and the appearance of neutralizing antibodies(15, 20–22). These manifestations of acute viral illness in the airways of previously healthy rats are consistent with those observed during acute respiratory viral illness in nonasthmatic humans(2–6) and are qualitatively similar for rats ranging in age from weanlings to adults, and among different strains of rats. Adult rats typically resolve all their virus-associated changes in airway morphology, histology, and physiology within 4-8 wk(15). However, after a single respiratory viral illness in the 1st mo of life, rats may develop chronic airway abnormalities that have similarities to human asthma, including chronic episodic airway obstruction with a reversible component(23), airway hyperresponsiveness(17, 24), chronic airway inflammation(21, 22, 24), and bronchiolar wall thickening with subepithelial fibrosis(19, 24). There appears to be a genetic predisposition to the development of these postviral asthma-like features, in that certain inbred strains, such as the Brown Norway, develop the postviral airway pathology readily, whereas another inbred strain, the Fisher 344, recovers from the infection without significant airway sequelae(22, 24). Treatment of rats with 3 d of high dose dexamethasone at a point several weeks after viral illness effectively suppresses the airway obstruction and inflammation, but the asthma-like syndrome returns within 3 wk of the last steroid dose(23, 25). The current study differs from these previous studies in that it addresses the effect of glucocorticoid treatment on the acute, rather than the chronic, airway manifestations of respiratory viral illness in rats.
The results of the current study support the hypothesis that airway dysfunction during acute viral respiratory infection is caused predominantly by corticosteroid-sensitive mechanisms, such as the inflammatory response. In the present study, dexamethasone treatment completely prevented the increased influx of inflammatory cells into the airways during acute viral illness, and markedly attenuated the virus-associated airway obstruction and hyperresponsiveness. In contrast, lung viral titers were increased in the dexamethasone-treated rats, suggesting that suppression of the inflammatory response, although effective for prevention of physiologic dysfunction of the airways, might also compromise viral clearance. The glucocorticoid dose used in this study was large, but analogous to doses commonly used to treat low birth weight human infants(26) and adults with severe acute exacerbations of asthma(27).
Acute respiratory viral infections have been associated with exacerbations of asthma, particularly in children, but also in adults(1, 28). Although the mechanisms of virus-induced airway dysfunction have not been defined precisely, viral infections cause an acute inflammatory response in the airways that could alter airway structure and function by a multitude of pathways(3, 29–31). Although glucocorticoids might be expected to inhibit the development of virus-induced airway dysfunction based on their antiinflammatory effects(32), the efficacy of steroids in virus-induced exacerbations of asthma remains controversial(7, 8). Contributing to the controversy are the variables of dose, duration, and timing of steroid therapy(7, 8), as well as the uncertainties of which exacerbations are caused by viral illness, even in the presence of viral-like symptoms(33). Our current study in rats allowed precise control of these variables and demonstrated unequivocal efficacy of glucocorticoid therapy in the prevention of airway dysfunction during acute respiratory viral infection. Although airway dysfunction due to acute viral illness in previously healthy airways cannot be equated with exacerbations of preexisting airway disease, it is likely that some of the pathophysiologic mechanisms responsible for the acute manifestations also contribute to virus-associated exacerbations of asthma.
In these experiments there was a potent uncoupling effect of dexamethasone on the transduction of the viral infection/replication signal to the inflammatory cell infiltration response. Although this study did not address the mechanism responsible for the uncoupling of virus-induced inflammation, the latter is likely due to glucocorticoid-mediated inhibition of the expression of key cytokines and adhesion molecules in the affected airway epithelium. In this regard, respiratory viral infection in mice has been reported to increase pulmonary expression of NF-κB a transcriptional factor that is involved in the activation of immunoregulatory genes in response to immune stimulators(34). Recent work has identified NF-κB as a primary target for the inhibitory effects of glucocorticoids(35, 36), which might explain not only why glucocorticoid therapy may be a potent inhibitor of virus-induced inflammation when administered during the early stages of viral infection, but also why glucocorticoid treatment may be much less effective when begun after the expression of proinflammatory cytokines and adhesion molecules has already occurred.
Although glucocorticoid treatment was efficacious in altering virus-associated airway dysfunction, a potential concern in the use of glucocorticoids during viral illness was the increase of pulmonary viral titers associated with steroid treatment; a number of previous observations are noteworthy in this regard. Children who developed respiratory syncytial virus-confirmed respiratory illness while on chronic systemic glucocorticoid therapy had larger peak viral titers and more prolonged viral shedding in nasal washes compared with those who had no steroid treatment and normal immune systems at the time of infection(37). However, there were no apparent differences in the clinical course of the viral illness, whether or not the children were on glucocorticoid therapy(37–39). In experimental rhinovirus infection, glucocorticoid treatment increased nasal viral titers significantly, but it had no significant effect on infection rate or duration of nasal viral shedding(40, 41). Other studies reporting effective glucocorticoid treatments for prevention of virus-induced asthma in children similarly had no evidence for an alteration of the length or severity of viral illness due to treatment, although no viral studies were done(42, 43). Local glucocorticoid treatment via nasal insufflation in Cotton rats infected with parainfluenza-3 caused a marked decrease in lung pathology, but also a log increase in viral titers(44), consistent with the findings in our current study. In contrast, experimental respiratory syncytial virus infection of gnotobiotic calves treated with dexamethasone resulted in not only increased peak viral titers and duration of shedding, but enhanced lung lesions in the treated animals(45). Whether steroid treatment during acute viral illness in rats affects the ultimate recovery from the infection remains a question to be addressed in future studies.
Finally, the ability to demonstrate such a marked attenuation of virus-induced physiologic and inflammatory airway changes in this animal model underscores the potential of the model for defining relevant pathways that could be altered similarly with agents having fewer side effects compared with glucocorticoids.
Abbreviations
- Rrs:
-
respiratory system resistance
- Cdyn:
-
dynamic respiratory system compliance
- PD20:
-
log dose (nmol/kg) i.v. methacholine required to increase resistance by 20 Pa mL-1 s
- BAL:
-
bronchoalveolar lavage
References
Pattemore PK, Johnston SL, Bardin PG 1992 Viruses as precipitants of asthma symptoms. I. Epidemiology. Clin Exp Allergy 22: 325–336.
Lemanske RF, Dick EC, Swenson CA, Vrtis RF, Busse WW 1989 Rhinovirus upper respiratory infection increases airway hyperreactivity and late asthmatic reactions. J Clin Invest 83: 1–10.
Bardin PG, Johnston SL, Pattemore PK 1992 Viruses as precipitants of asthma symptoms. II. Physiology and mechanisms. Clin Exp Allergy 22: 809–822.
Hall WJ, Hall CB, Speers DM 1978 Respiratory syncytial virus infection in adults: clinical, virologic and serial pulmonary function studies. Ann Intern Med 88: 203–205.
Hall WJ, Douglas RG, Hyde RW, Roth FK, Cross AS, Speers DM 1976 Pulmonary mechanics after uncomplicated influenza A infection. Am Rev Respir Dis 113: 141–148.
Blair HT, Greenberg SB, Stevens PM, Bilunos PA, Couch RB 1976 Effects of rhinovirus infection on pulmonary function of healthy human volunteers. Am Rev Respir Dis 114: 95–102.
Weinberger M 1988 Corticosteroids for exacerbations of asthma: current status of the controversy. Pediatrics 81: 726–729.
Shapiro GG 1995 Steroids and asthma. Pediatrics 96: 347–348.
Grant CC, Duggan AK, DeAngelis C 1995 Independent parental administration of prednisone in acute asthma: a double-blind, placebo-controlled, crossover study. Pediatrics 96: 224–229.
Laitinen LA, Laitinen A, Haahtela T 1992 A comparative study of the effects of an inhaled corticosteroid, budesonide, and aβ-2-agonist, terbutaline, on airway inflammation in newly diagnosed asthma: a randomized, double-blind, parallel-group controlled trial. J Allergy Clin Immunol 90: 32–42.
Shelhamer JH, Levine SJ, Wu T, Jacoby DB, Kaliner MA, Rennard SI 1995 NIH conference. Airway inflammation. Ann Intern Med 123: 288–304.
Cypcar D, Busse WW 1993 Role of viral infections in asthma. Immunol Allergy Clin North Am 13: 745–767.
Busse WW, Lemanske RF, Dick EC 1992 The relationship of viral respiratory infections and asthma. Chest 101: 385S–388S.
Calhoun WJ, Dick EC, Schwartz LB, Busse WW 1994 A common cold virus, rhinovirus 16, potentiates airway inflammation after segmental antigen bronchoprovocation in allergic subjects. J Clin Invest 94: 2200–2208.
Sorkness R, Clough JJ, Castleman WL, Lemanske RF 1994 Virus-induced airway obstruction and parasympathetic hyperresponsiveness in adult rats. Am J Respir Crit Care Med 150: 28–34.
Kimsey PB, Goad MEP, Zhi-Bo Z, Brackee G, Fox JG 1989 Methyl prednisolone acetate modulation of infection and subsequent pulmonary pathology in hamsters exposed to parainfluenza-1 Virus (Sendai). Am Rev Respir Dis 140: 1704–1711.
Sorkness R, Lemanske RF, Castleman WL 1991 Persistent airway hyperresponsiveness after neonatal viral bronchiolitis in rats. J Appl Physiol 70: 375–383.
Castleman WL, Sorkness RL, Lemanske RF, McAllister PK 1990 Viral bronchiolitis during early life induces increased numbers of bronchiolar mast cells and airway hyperresponsiveness. Am J Pathol 137: 821–831.
Castleman WL, Sorkness RL, Lemanske RF, Grasee G, Suyemoto MM 1988 Neonatal viral bronchiolitis and pneumonia induces bronchiolar hypoplasia and alveolar dysplasia in rats. Lab Invest 59: 387–396.
Castleman WL, Brundage-Anguish LJ, Kreitzer L, Neunschwander SB 1987 Pathogenesis of bronchiolitis and pneumonia induced in neonatal and weanling rats by parainfluenza (Sendai) virus. Am J Pathol 129: 277–286.
Castleman WL, Owens SB, Brundage-Anguish LJ 1989 Acute and persistent alterations in pulmonary inflammatory cells and airway mast cells induced by Sendai virus infection in neonatal rats. Vet Pathol 26: 18–25.
Sorden SD, Castleman WL 1995 Virus-induced increases in airway mast cells in Brown Norway rats are associated with enhanced pulmonary viral replication and persisting lymphocytic infiltration. Exp Lung Res 21: 197–213.
Kumar A, Sorkness RL, Kaplan MR, Lemanske RF 1997 Chronic, episodic, reversible airway obstruction after viral bronchiolitis in rats. Am J Respir Crit Care Med 155: 130–134.
Uhl EW, Castleman WL, Sorkness RL, Busse WW, Lemanske RF, McAllister PK 1996 Parainfluenza virus-induced persistence of airway inflammation, fibrosis, and dysfunction associated with TGF-β1 expression in Brown Norway rats. Am J Respir Crit Care Med 154: 1834–1942.
Sheth KK, Sorkness RL, Clough JJ, McAllister PK, Castleman WL, Lemanske RF 1994 Reversal of persistent postbronchiolitis abnormalities with dexamethasone in rats. J Appl Physiol 76: 333–338.
Durand M, Sardesai S, McEvoy C 1996 Effects of early dexamethasone therapy on pulmonary mechanics and chronic lung disease in very low birth weight infants: a randomized, controlled trial. Pediatrics 95: 584–590.
Corbridge TC, Hall JB 1995 The assessment and management of adults with status asthmaticus. Am J Respir Crit Care Med 151: 1296–1316.
Johnston SL, Pattemore PK, Sanderson G, Smith S, Campbell MJ, Josephs LK, Cunningham A, Robinson BS, Myint SH, Ward ME, Tyrrell DAJ, Holgate ST 1996 The relationship between upper respiratory infections and hospital admissions for asthma: a time-trend analysis. Am J Respir Crit Care Med 154: 654–660.
Fraenkel DJ, Bardin PG, Sanderson G, Lampe F, Johnston SL, Holgate ST 1995 Lower airways inflammation during rhinovirus colds in normal and in asthmatic subjects. Am J Respir Crit Care Med 151: 879–886.
Cannon MJ, Openshaw PJM, Askonas BA 1988 Cytotoxic T cells clear virus but augment lung pathology in mice infected with respiratory syncytial virus. J Exp Med 168: 1163–1168.
Hegele RG, Hayashi S, Hogg JC, Pare PD 1995 Mechanisms of airway narrowing and hyperresponsiveness in viral respiratory tract infections. Am J Respir Crit Care Med 151: 1659–1664.
Brattsand R, Pipkorn U 1991 Glucocorticoids- experimental approaches. In: Kaliner MA, Barnes PJ, Persson CGA (eds) Asthma: Its Pathology and Treatment. Marcel Dekker, New York, pp 667–709.
Sokhandan M, McFadden ER, Huang YT, Mazanec MB 1995 The contribution of respiratory viruses to severe exacerbations of asthma in adults. Chest 107: 1570–1574.
Jacoby DB, Choi AMK 1994 Responses to influenza virus infections in C57B 1:6 mice and C3H/HeJ mice: differential activation of transcriptional regulatory proteins. Am J Respir Crit Care Med 149:A721.
Scheinman RI, Cogswell PC, Lofquist AK, Baldwin AS 1995 Role of transcriptional activation of I κ B α in mediation of immunosuppression by glucocorticoids. Science 270: 283–286.
Auphan N, DiDonato JA, Rosette C, Helmberg A, Karin M 1995 Immunosuppression by glucocorticoids: inhibition of NF-κ B activity through induction of I κ B synthesis. Science 270: 286–290.
Hall CB, Powell KR, MacDonald NE, Gala CL, Menegus ME, Suffin SC, Cohen HJ 1986 Respiratory syncytial viral infection in children with compromised immune function. N Engl J Med 315: 77–81.
Leer JA, Green JL, Heimlich EM, Hyde JS, Moffet HL, Young GA, Barron BA 1969 Corticosteroid treatment in bronchiolitis. A controlled, collaborative study in 297 infants and children. Am J Dis Child 117: 495–503.
Connolly JH, Field CM, Glasgow JF, Slattery CM, MacLynn DM 1969 A double blind trial of prednisolone in epidemic bronchiolitis due to respiratory syncytial virus. Acta Paediat Scand 58: 116–120.
Farr BM, Gwaltney JM, Hendley JO, Hayden FG, Naclerio RM, McBride T, Doyle WJ, Sorrentino JV, Riker DK, Proud D 1990 A randomized controlled trial of glucocorticoid prophylaxis against experimental rhinovirus infection. J Infect Dis 162: 1173–1177.
Gustafson LM, Proud D, Hendley JO, Hayden FG, Gwaltney JM 1996 Oral prednisone therapy in experimental rhinovirus infections. J Allergy Clin Immunol 97: 1009–1014.
Brunette MG, Lands L, Thibodeau LP 1988 Childhood asthma: prevention of attacks with short-term corticosteroid treatment of upper respiratory tract infection. Pediatrics 81: 624–628.
Connett G, Lenney W 1993 Prevention of viral induced asthma attacks using inhaled budesonide, Arch Dis C. hild 68: 85–87.
Prince GA, Porter DD 1996 Treatment of parainfluenza virus type 3 bronchiolitis and pneumonia in a cotton rat model using topical antibody and glucocorticosteroid. J Infect Dis 173: 598–608.
Thomas LH, Stott EJ, Collins AP, Crouch S, Jebbett J 1984 Infection of gnotobiotic calves with a bovine and human isolate of respiratory syncytial virus. Modification of the response by dexamethasone. Arch Virol 79: 67–77.
Acknowledgements
The authors thank the Pulmonary Function Laboratory of the University of Wisconsin Hospitals and Clinics for blood gas measurements.
Author information
Authors and Affiliations
Additional information
Supported by National Institutes of Health Grants AI-34891 and AI-00995. H.M. was a recipient of an Allen & Hanburys Respiratory Institute Allergy Fellowship Award.
Rights and permissions
About this article
Cite this article
Mehta, H., Sorkness, R., Kaplan, M. et al. Effects of Dexamethasone on Acute Virus-Induced Airway Dysfunction in Adult Rats. Pediatr Res 41, 872–877 (1997). https://doi.org/10.1203/00006450-199706000-00012
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199706000-00012