Abstract
It was previously shown that tracheal obstruction accelerated fetal lung growth and eventually reversed the pulmonary hypoplasia in experimental diaphragmatic hernia. We have successfully developed a reversible tracheal obstruction technique in fetal sheep using balloon occlusion and showed that 3 wk of obstruction induced significant lung growth of the same magnitude as the tracheal ligation. The purpose of this study was to examine the effects of 1 and 3 wk of tracheal occlusion on the alveolar cell population with specific attention to the type II pneumocytes. We first showed that 1 wk of occlusion induced a significant increase in lung weight and in alveolar surface area. We then used the surfactant protein C (SP-C) mRNA as a specific marker of differentiated type II pneumocytes. Total RNA was isolated from fetal sheep lung with or without tracheal occlusion, and Northern blots were hybridized with a cDNA probe specific for the sheep SP-C. The results show a dramatic decrease in SP-C mRNA expression (8.8-fold, p < 0.01). In situ hybridization showed a marked decrease in the density of cells expressing SP-C, as well as the amount of SP-C mRNA expressed by the cells. The effect was present as early as 1 wk of occlusion. The sparseness of type II pneumocytes was further confirmed by electron microscopy. We thus conclude that tracheal obstruction causes a profound decrease in the number of type II pneumocytes in the lungs. Given the crucial role of type II pneumocytes in surfactant production, we could speculate that, if tracheal occlusion is able to accelerate lung growth, the final product is probably surfactant-deficient.
Similar content being viewed by others
Main
Despite prenatal diagnosis and recent advances in neonatal treatment, the mortality and the morbidity of CDH remain above 50%(1). Complications include pulmonary insufficiency from mechanical compression on the lung, pulmonary hypertension, and surfactant deficiency(2). However, the degree of lung hypoplasia seems to be the most important predictor of survival(3).
Congenital laryngeal atresia is a rare malformation in humans that leads to lungs that are larger, contain more alveoli, and appear to have a more mature architecture than expected for gestational age(4). This has been documented by several case reports and confirmed by experimental tracheal ligation in various animal species(5–7). Experimentally, tracheal ligation has been shown to prevent pulmonary hypoplasia in several models: chronic amniotic fluid loss (leading to severe oligohydramnios)(8), bilateral nephrectomy(9), lung liquid drainage(7), and more recently, CDH(9–11).
Using an indwelling balloon catheter, we were able to obtain effective tracheal occlusion without permanent tracheal damage(12). We have previously reported that 21-28 d of occlusion produced an increase in lung weight and volume by 2-3 times(p < 0.001), whereas heart and liver weight remained similar to that of controls. Airspace fraction and radial alveolar counts were raised, and alveolar number per kg of body weight was doubled (p < 0.001). However, the alveolar number per lung volume as well as the DNA and protein content per unit of weight of lung tissue did not change(12). Total lung DNA and protein per g of weight were increased, whereas the DNA/protein ratio did not change. This confirmed true lung hyperplasia, not simply hypertrophy or overdistension.
Based on several morphometric studies(10), it was speculated that the resulting lungs were fully functional. However, using EM, Alcorn et al.(5) mentioned that type II cells were infrequently observed after 3 wk of tracheal ligation in fetal sheep. On the other hand, the concentration of disaturated phosphatidylcholine, a major component of surfactant (produced by type II cells), appeared normal in three human neonates with laryngeal atresia(4). In an experimental model of tracheal ligation in fetal sheep, the concentration of phospholipids and phosphatidylcholine was found to be the same as in controls(13). However, this absence of difference could result from the prematurity of those lambs delivered at 130 d (term = 140-145 d).
The purpose of the current study, therefore, was to investigate the effect of fetal tracheal occlusion on alveolar cell population. We focused our attention on type II pneumocytes, which cover only 5% of the alveolar surface, but are essential to lung function because they produce pulmonary surfactant, a phospholipid and protein complex that reduces surface tension at the air-water interface of alveoli(14). The SP-C mRNA appears to be expressed exclusively by type II cells(15, 16) and was used as a phenotypic indicator of differentiated type II pneumocytes. Moreover, to evaluate the effect of the length of occlusion on lung cell population, experiments were performed on a set of animals with a shorter duration of tracheal occlusion than the one in the group previously described(12). Comparison between animals with 1 and 3 wk of occlusion and untouched littermates was made.
METHODS
Fetal surgical manipulation. Time-dated pregnant mixed-breed ewes were operated at 117 (3 wk of occlusion) or 126 d of gestation (1 wk of occlusion). Details of the operative procedure have been published(12). Briefly, pregnant ewes were anesthetized, and the fetus were exposed through a hysterotomy. After dissection and puncture of the trachea, a Swan-Ganz catheter was introduced, and the balloon was inflated until tracheal fluid stopped leaking with some resistance on moving the catheter. In the animals with 3 wk of occlusion, the catheter was brought out to measure tracheal and amniotic fluid pressures(12). Subsequently, for the 1-wk occlusion, the catheter was left buried in a s.c. pocket in the ewe to minimize the risk of infection and fetal loss. Untouched twins served as normal controls (the incidence of multiple gestation was over 90%). Postoperatively, the ewes from the 1-wk occlusion group received medroxyprogesterone (300 mg intramuscularly) daily until delivery to prevent abortion in this late gestation experiment. All fetuses were delivered by repeat cesarean section at 137 d for the group with 3 wk of occlusion(12) and at 133 d for the group with 1 wk of occlusion. The difference between the delivery date and the medication of the two groups is explained by the difficulty to perform major fetal surgery during the last 2 wk of gestation. An intervention after 130 d for the 1-wk occlusion group might have resulted in an unacceptably high incidence of premature delivery. However, appropriate controls for the gestational group were used for each group. For the animals analyzed in the current report, the duration of occlusion was precisely 7 or 21 d.
Gross and histologic analysis. Results of gross and histologic analyses of the group with 3 wk of occlusion have been previously reported(12), and preserved samples from the same animals were used in the present study for SP-C mRNA study and EM (three animals with 3 wk of occlusion and their twin controls). Additionally, four animals with 1 wk of occlusion and the untouched controls were analyzed, and the results were reported. Details about the method of analysis were previously published(12). Briefly, the catheter balloon inflation was confirmed by palpation of the trachea before removing it. Total body, lung, heart, liver weights, and kidney weight were recorded. Specimens from the middle lobe and lingula were processed for EM, and some were frozen at -70°C. The lungs were then fixed in formalin at 25 cm of water pressure for 48 h, and the lung volumes were measured using the volume displacement method(17). The lungs were cut in sagittal sections 3-5 mm thick, and random 1 × 1.5-cm blocks were taken from a section, representing each lobe, and embedded in paraffin. Slides were made from each block and stained with hematoxylin and eosin for routine histology, morphometric analysis, and radial alveolar counts. The morphometric analysis was performed using the Macintosh Image 1.44 morphometric analysis program, which allowed us to measure the airspace fraction and to count the number of alveoli per unit of surface area(12). Fifteen readings were taken for each lower and upper lobe for a total of 60 readings per lung. The alveolar density was calculated using the method of Weibel(18) and Weibel and Gomez(19) and expressed in alveoli per cm3. Using that value combined with the measured lung volume and the airspace fraction, it was possible to approximate the total amount of alveoli and the total alveolar surface for the pair of lungs. Radial alveolar counts were taken manually, 10 readings from lower and upper lobes, for a total of 40 readings per pair of lungs(12).
SP-C mRNA analysis. Total RNA was isolated by sedimentation through 5.7 M CsCl. Concentrations of RNA were determined by spectrophotometric absorption at 260 nm. Total RNA was fractionated by formaldehyde gel electrophoresis and transferred onto nylon membranes(MagnaGraph, MSI, Westboro, MA). Northern blot was hybridized with a cDNA probe specific for the sheep SP-C (F. Possmayer, unpublished results) and exposed for autoradiography using X-AR x-ray film (Kodak, Rochester, NY). The membranes were rehybridized with an oligonucleotide probe against the 18 S ribosomal band to normalize for RNA loading. Samples from only three 1-wk occluded animals and their untouched controls were available for the Northern blot analysis.
In situ hybridization. Complementary RNA probes were labeled with [3H]uracil triphosphate and [3H]cytosine triphosphate(DuPont Canada, Markham, ON, Canada) as previously described(20). The DNA template was digested with RNase-free DNase I (Promega, Madison, WI), and the probe was extracted with an equal volume of phenol/chloroform (1:1), precipitated with ethanol, and resuspended in diethylpyrocarbonate-treated water. Before hybridization, limited alkaline hydrolysis of the RNA probes was performed to reduce the transcript length to 0.1-0.3 kb. Hybridization methods were described previously(20). Hybridization was performed overnight at 54 °C in 50% formamide, 0.3 M NaCl, 10 mM Tris-HCl (pH 8.0), 1 mM EDTA, 1 × Denhardt's solution, 10% dextran sulfate, 0.5 mg/mL yeast tRNA, and 120 ng/mL cRNA probe in a moist chamber containing 0.3 M NaCl, 50% formamide. After hybridization, the sections were treated with RNase(20). The final wash was done in 0.1 × SDS for 30 min at 68 °C. Slides were dipped in NBT-2 photographic emulsion (Kodak), stored for 4 d until development, and counterstained with hematoxylin and eosin before photomicrography. Nonspecific binding and background were evaluated using sense probes on one section from each animal. For each animal, one section from at least three different lobes was analyzed. The number of cells expressing SP-C mRNA was counted on five random fields of each section. Cells were considered positive for SP-C if three or more silver grains were observed over the cell. Additionally, for each section, the number of silver grains per positive cell was determined for 10 randomly selected type II cells.
EM. Tissue sections were fixed with 3% glutaraldehyde in 0.1 M sodium cacodylate buffer and additionally fixed in 2% OsO4 in the same buffer before embedding in Epon 812 (JBS Supplies, Dorval, PQ, Canada). Ultrathin sections were stained with uranyl acetate and lead citrate. Observations and EM pictures were taken with a CX-1200 JEOL electron microscope operating at 60 kV. Random serial electron micrographs were taken from three sets of animals, three fetuses with 3 wk of occlusion and their untouched littermates. Only samples from the first experiment(12) were appropriately fixed for EM analysis. Type II pneumocytes were identified as lamellar body-containing cells. To avoid bias secondary to alveolar distention in the experimental group, the number of type II pneumocytes over the cellular surface of the alveoli, excluding the alveolar space, was measured. The Sigma-Scan Scientific Measurement Program Version 3.90 (Jandel Scientific, Corte Madera, CA) was used to measure lung parenchyma surface on EM pictures with an electronic pen to delimit and exclude the airspace.
Data analysis. Results of lung weight and volume, as well as the alveolar number and alveolar surface, were reported in relation to body weight to correct for differences in fetal size (there is a great variability with multiple gestation). Autoradiograms were analyzed with a Bioimage analyzer using the WBA program (Visage 110S, Millipore, Mississauga, ON, Canada), and results were normalized to the 18 S ribosomal band. Morphometry data and Northern blot results were compared using a t test. For in situ hybridization, results were expressed as mean ± SEM number of cells by field and analyzed by analysis of variance. When indicated, the density of SP-C-positive cells was normalized for the airspace fraction calculated on multiple sections of each lobe of the same animal (as described in the section on histologic analysis) using the equation: density of SP-C positive cell/(1 - airspace fraction). The results of the EM study were reported over the measured cell surface and analyzed with Welsh's t test(21). Statistical significance was accepted atα < 0.05.
RESULTS
We have previously shown that 3 wk of fetal tracheal occlusion induced a significant increase in lung growth(12). In the present study, we first tested the effect of a shorter period of occlusion on fetal lung growth. One of the six ewes that were operated developed ketoacidosis on the first postoperative day, from which it recovered. However, the fetus was found dead at sacrifice, 1 wk later, and was not included in the study. At the postmortem examination, the catheter was found in good place in all fetuses, but one balloon was partly deflated. This was considered inherent to the technique we used, and the animal was included for the analysis.
As shown in Table 1, the lung weight over the body weight as well as lung volume over the body weight are significantly increased in the animals with 1 wk of occlusion when compared with the untouched fetuses(p < 0.01). The effect was specific to the lungs because no differences were seen with the heart, liver, and kidneys (results not shown). To differentiate between lung growth and lung distention, histologic measurements were done. Table 1 shows that the airspace fraction, the alveolar density, and the radial alveolar count were not different between the experimental versus untouched fetuses. The increase in total alveolar number did not reach the level of significance because of a wide range of value (p = 0.11). However, the total alveolar surface was significantly higher after tracheal ligation. Results of the histologic analysis were from four sets of experimental and untouched animals (one set of specimens was too inadequately fixed to be used).
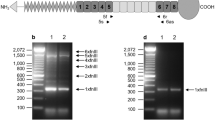
SP-C mRNA assay was first performed on total lung homogenate of three sets of experimental and untouched control animals from the 1-wk occlusion group.Figure 1 shows that 1 wk of tracheal occlusion induces a marked decrease in SP-C mRNA expression compared with that of untouched twins. When normalized for RNA loading with hybridization to the 18 S ribosomal band, SP-C mRNA is decreased by 8.5-fold compared with that of untouched controls(controls versus experimentals, 3.06 ± 0.40 versus 0.36 ± 0.13 of arbitrary units, p < 0.005).
To differentiate between a decrease in SP-C mRNA abundance within individual cells and a decrease in the number of cells expressing it,in situ hybridization studies were done. Samples from both the 3-wk occlusion(12) and the present 1-wk occlusion group were used in this part of the study. Figure 2 shows a typical example of cell-specific SP-C mRNA expression in those lungs. In the untouched animals, we found SP-C mRNA in almost every alveoli (controls). However, after tracheal occlusion the number of cells expressing the SP-C transcript dramatically decreased (experimentals). Table 2 summarizes the results. One week or three weeks of occlusion produced a highly significant decrease in the density of cells expressing the SP-C transcript. When corrected for the difference in airspace fraction between control and experimental groups, the difference remained highly significant (controls versus experimentals, number of cells/field, 1 wk of occlusion: 174± 34 versus 30 ± 7; 3 wk of occlusion: 220 ± 17versus 34 ± 14, p < 0.001). Moreover, as shown in Table 3, the number of silver grains per cell, which reflects the amount of SP-C mRNA in a given cell, was also decreased in the experimental group. In addition, a small, but significant, decrease in the number of silver grains per type II cell was observed in the 1-wk untouched control group as compared with the 3-wk untouched control group(Table 3).
SP-C mRNA-expressing cells are decreased after tracheal occlusion. Lung sections were hybridized with radiolabeled cRNA antisense RNA probes, and hybridization was detected by autoradiography (white grains on dark field). The left panel (Control) is from an untouched fetus, and the right panel (Experimental) is from a fetus with 3 wk of occlusion. Bar = 100 μm.
EM studies were conducted to determine whether tracheal occlusion was accompanied by a decrease in the number of type II cells.Figure 3 shows that, in control animals, a type II pneumocyte could be identified in almost every alveolar space (panel b). In contrast, most of the alveolar spaces are devoid of lamellar body-containing cells after fetal tracheal occlusion (panel a). To measure the number of type II cells present in the lungs of experimental animals versus the lungs of untouched twins, we performed a quantitative analysis of the electron micrographs. A mean of 17.0 ± 4.6 mm2 of alveolar cell surface per animal (excluding the airspace) was analyzed in the experimental group, and no type II pneumocytes could be identified in the three animals (0 type II cells per 51 mm2 of cell surface). In contrast, with a mean 21.7 ± 3.5 mm2 of alveolar cell surface evaluated, 6-7 type II pneumocytes were found in the untouched animals (20 type II cells per 65 mm2 of cell surface). The difference between the two groups was statistically significant (p < 0.02).
Type II pneumocytes are sparsely seen after tracheal occlusion. Photomicrography shows the typical appearance of lung parenchyma of a fetus with 3 wk of tracheal occlusion (panel a) or the untouched twin (panel b). A type II pneumocyte (arrows) could be identified in most alveoli of the untouched animal, whereas they are sparsely seen in the experimental animal. Al, alveolar space; bar = 2μm.
DISCUSSION
Fetal tracheal obstruction has been shown to induce a dramatic increase in lung growth(5, 9–12, 22), and has been proposed to reverse CDH in utero(10, 22). The general morphology and histology of lungs subjected to occlusion appears normal(12). Moreover, a specific study looking at the development of the vasculature confirmed that it is adequately developed together with the alveoli(10). Therefore, it was speculated that tracheal obstruction exerts its effect by enhancing normal mechanisms of pulmonary development. However, a decrease in the density of type II cells after tracheal ligation was first observed by Alcorn et al.(5) in 1977. It was generally concluded that this represented only a relative decrease given the substantial increase in the other cell types.
In this report, we show that 1 wk of tracheal occlusion is sufficient to increase lung size and alveolar surface area per g of body weight. A slight but nonsignificant increase in airspace fraction is observed and is thought to be secondary to the thinning of the alveolar septa(5, 10). Alveolar distention may also contribute to some degree; however, the absence of variation in the alveolar density and radial alveolar count combined with the increase in alveolar surface suggest that the observed changes in lung size are a reflection of lung growth and not pure lung distention. These results are not unexpected, because 1 wk of fetal tracheal obstruction is enough to reverse lung hypoplasia induced by lung liquid drainage(7). A direct comparison with our previous report on 3 wk of fetal tracheal occlusion(12) is difficult because the animals were delivered at different times, and only one of the two groups received medroxyprogesterone.
We have clearly demonstrated that tracheal occlusion as short as 1 wk induced a dramatic decrease in the type II cell population in these lungs. As mentioned in “Results,” the change observed is still highly significant when corrected for the slightly increased airspace fraction seen with tracheal occlusion. On the other hand, it could be hypothesized that the number of type II cells remained constant, whereas the remainder of the lung grew. Because the final lung size is not only determined by the treatment but also by the size of the fetus, it is not possible to evaluate, for each individual animal of the experimental group, the percentage of lung overgrowth related to the treatment. However, the 20-fold decrease in the density of SP-C-positive cells is out of proportion to the 3-fold increase in lung size observed with 3 wk of occlusion(12). Therefore, these observations represent an absolute decrease in type II pneumocytes in those lungs. Given the crucial role of those cells in surfactant production, we could speculate that the quasi-disappearance of the type II pneumocytes could impair seriously the function of the lungs at birth.
Lung maturation is usually assessed either by surfactant analysis or standard pulmonary function tests. Measuring the surfactant activity in tracheal liquid would have been difficult to interpret because of a 5-10-fold difference in fluid recovery between experimental and control animals (our unpublished observation). Alternatively, surfactant could have been extracted from lung tissue(23). However, the method is not very sensitive, and we speculated that the levels of surfactant in animals killed at 133 d would have been below the levels of detection. In the same perspective, standard pulmonary function tests were not performed because the interpretation would have been difficult given the important differences in lung sizes(12). Therefore, we relied on indirect indices of lung maturation. Analysis of SP-C mRNA does not only allow precise quantification of differentiated type II cells in these lungs, but also gives information on the functional adequacy of the type II pneumocytes. The significant decrease in SP-C mRNA content of type II cells, as assessed by the number of silver grains per positive cell, suggests that tracheal occlusion not only affected the process of cell differentiation during lung development but also the final maturation of the cell. This effect could be a reflection of specific inhibition of SP-C gene expression or part of a global effect of tracheal occlusion on type II cells. However, the results of EM indicate that the decreased density of cells expressing SP-C mRNA after tracheal occlusion is largely the consequence of fewer type II pneumocytes.
We were somewhat surprised to see a difference in the abundance of the SP-C mRNA in cells between the 1- and 3-wk control groups. This difference is likely secondary to the earlier delivery in the 1-wk occlusion group (133 d versus 137 d), which could result in less mature type II cells. This could also result from the use of medroxyprogesterone in the 1-wk group only. However, the slight difference observed between the two groups does not alter the significance of the data concerning the effect of the occlusion.
The validity of the controls could be discussed. Sham operation of the twin fetus was attempted in the previous study(12); however, this had resulted in fetal death or premature delivery in five consecutive cases. To save time, money, and animal lives, we decided to use untouched twins as controls. They have gone through the stress of hysterotomy but were not subjected to direct surgery. We could not formally exclude that the stress of the surgery contributed to the reduction in the number of type II cells. On the other hand, if the technique of tracheal occlusion becomes applied to human fetuses, the gold standard will not be the sham operation but the normal newborn infant. Therefore, the effect of fetal manipulation will be as important as the specific effect of the occlusion.
The mechanism by which tracheal obstruction causes increased lung growth is still largely unknown. A 180% increase in IGF-II mRNA was recently observed after 7 d of fetal tracheal obstruction in sheep(24). Additionally, antisense oligonucleotides for platelet-derived growth factor B and its receptor inhibit the mechanical strain-induced fetal lung cell growth in vitro(25). Increase in IGF-II, platelet-derived growth factor B, or other unidentified growth factors are certainly involved in the lung growth induced by tracheal occlusion; these growth factors could also affect type II cell differentiation(26). On the other hand, an increase in the number of type II pneumocytes per unit of surface area is observed in lung hypoplasia either induced by chronic lung fluid drainage(5) or by CDH(27). It was hypothesized that, in hypoplastic lungs, the differentiation of cuboidal type II cells into squamous type I cells was retarded(27). Inversely, we could explain the diminution of type II cells after tracheal occlusion by a more rapid natural differentiation of type II cells into type I cells(14). Given the magnitude of the type II cells disappearance, the observed decrease is probably the result of both mechanisms: an inhibition of the differentiation of undifferentiated cells into type II cells and a stimulation of the differentiation of type II cells into type I cells.
The clinical relevance of our findings remains undetermined. One could argue that a surfactant-deficient lung is preferable to a hypoplastic lung because the disease could be rapidly reversed with exogenous surfactant. However, the problem differs considerably from the immature lung of the premature infant. In the latter situation, some type II pneumocytes are present but not yet producing sufficient surfactant. In a primate model, premature delivery induces a rapid maturation of the surfactant system, resulting in an increase of SP-C mRNA beyond the level found in full-term controls within the first 24 h(28). On the other hand, in a type II cell-depleted lung, as we observed with tracheal occlusion, a prolonged delay may be necessary to allow time for type II cell division and maturation before surfactant production is initiated. During this time, irreversible lung damage could result from a forced ventilation applied to a noncompliant lung. Our observation is reinforced by preliminary results suggesting that tracheal obstruction failed to correct the surfactant deficiency seen with CDH(29). However, the case of CDH is more complex. The morphologic studies suggest that the number of type II cells per unit of volume is increased in those hypoplastic lung(5); however, physiologic studies have shown that the production of surfactant is deficient(30, 31), suggesting an inhibition of the type II cells. Therefore, tracheal occlusion could have a further negative effect on the activity of the type II cells.
This study raises important concerns about the maturity of these lungs that should be addressed before widespread human trial. Therefore, research needs to be directed to techniques that will combine tracheal occlusion with methods promoting lung maturation such as antenatal maternal steroid administration(32). Moreover, long-term postnatal studies are needed to evaluate the capacity of those lungs to promote normal gas exchange.
Abbreviations
- SP-C:
-
surfactant protein C
- CDH:
-
congenital diaphragmatic hernia
- EM:
-
electron microscopy
References
Lund DP, Mitchell J, Kharasch V, Quigley S, Kuehn M, Wilson JM 1994 Congenital diaphragmatic hernia: the hidden morbidity. J Pediatr Surg 29: 258–262
Laberge J-M, Sigalet DL, Guttman FM 1995 Congenital diaphragmatic hernia. In: Nyhus LM, Condon RE (eds) Hernia, 4th Ed. JB Lippincott, Philadelphia, pp 555–566
Antunes MJ, Greenspan JS, Cullen JA, Holt WJ, Baumgart S, Spitzer AR 1995 Prognosis with preoperative pulmonary function and lung volume assessment in infants with congenital diaphragmatic hernia. Pediatrics 96: 1117–1122
Wigglesworth JS, Desai R, Hislop AA 1987 Fetal lung growth in congenital laryngeal atresia. Pediatr Pathol 7: 515–525
Alcorn D, Adamson TM, Lambert TH 1977 Morphologic effects of chronic tracheal ligation and drainage in the fetal lamb lung. J Anat 123: 649–660
Jost A, Policard A 1948 Contribution expérimentaleá l'étude du développment prénatal du poumon chez le lapin. Arch Anat Microsc Morphol Exp 37: 323–332
Nardo L, Hooper SB, Harding R 1995 Lung hypoplasia can be reversed by short-term obstruction of the trachea in fetal sheep. Pediatr Res 38: 690–696
Adzick NS, Harrison MR, Glick PL, Villa RL, Finkbeiner W 1984 Experimental pulmonary hypoplasia and oligohydramnios: Relative contributions of lung fluid and fetal breathing movements. J Pediatr Surg 19: 658–665
Wilson JM, DiFiore JW, Peters CA 1993 Experimental fetal tracheal ligation prevents the pulmonary hypoplasia associated with fetal nephrectomy: possible application for congenital diaphragmatic hernia. J Pediatr Surg 28: 1433–1439
DiFiore JW, Fauza DO, Salvin R, Wilson JM 1995 Experimental fetal tracheal ligation and congenital diaphragmatic hernia: a pulmonary vascular morphometric analysis. J Pediatr Surg 30: 917–924
Hedrick MH, Estes JM, Sullivan KM, Bealer JF, Kitterman JA, Flake AW, Adzick NS, Harrison MR 1994 Plug the lung until it grows (PLUG): a new method to treat congenital diaphragmatic hernia in utero. J Pediatr Surg 29: 612–617
Hashim E, Laberge J-M, Chen M-F, Quillen EW Jr 1995 Reversible tracheal obstruction in the fetal sheep: effects on tracheal fluid pressure and lung growth. J Pediatr Surg 30: 1172–1177
Moessinger AC, Harding R, Adamson TM, Singh M, Kiu GT 1990 Role of lung fluid volume in growth and maturation of the fetal sheep lung. J Clin Invest 86: 1270–1277
Finkelstein JN 1990 Physiological and toxicologic responses of alveolar type II cells. Toxicology 60: 41–52
Hawgood S, Clements JA 1990 Pulmonary surfactant and its apoproteins. J Clin Invest 86: 1–6
Horowitz S, Watkins RH, Auten R, Mercier CE, Cheng ER 1991 Differential accumulation of surfactant protein A, B, and C mRNAs in two epithelial cell types of hyperoxic lung. Am J Respir Cell Mol Biol 5: 511–515
Scherle W 1970 A simple method for volumetry of organs in quantitative stereology. Mikroskopie 26: 57–60
Weibel ER 1973 A simplified morphometric method for estimating diffusing capacity in normal and emphysematous human lungs. Am Rev Respir Dis 107: 579–588
Weibel ER, Gomez DM 1962 A principle for counting tissue structures on random sections. J Appl Physiol 17: 343–348
Piedboeuf B, Johnston CJ, Watkins RH, Hudak BB, Lazo JS, Cherian MG, Horowitz S 1994 Increased expression of tissue inhibitor of metalloproteinases (TIMP-I) and metallothionein in murine lungs after hyperoxic exposure. Am J Respir Cell Mol Biol 10: 123–132
Winer BJ, Brown DR, Michels KM 1991 Stastistical Principles in Experimental Design, 3rd Ed. McGraw-Hill, New York, pp 66–69
Bealer JF, Skarsgard ED, Hedrick MH, Meuli M, VanderWall KJ, Flake AW, Adzick NS, Harrison MR 1995 The “PLUG” odyssey: adventures in experimental fetal tracheal occlusion. J Pediatr Surg 30: 361–364
Katyal SL, Estes LW, Lombardi B 1977 Method for the isolation of surfarctant from homogenate and lavages of lung of adult, newborn and fetal rats. Lab Invest 36: 585–592
Hooper SB, Han VK, Harding R 1993 Changes in lung expansion alter pulmonary DNA synthesis and IGF-II gene expression in fetal sheep. Am J Physiol 265:L403–L409
Souza P, Sedlackova L, Kuliszewski M, Wang J, Liu J, Tseu I, Liu M, Tanswell AK, Post M 1994 Antisense oligodeoxynucleotides targeting PDGF-B mRNA inhibit cell proliferation during embryonic rat lung development. Development 120: 2163–2173
Ryan RM, Mineo-Kuhn MM, Kramer CM, Finkelstein JN 1994 Growth factors alter neonatal type II alveolar epithelial cell proliferation. Am J Physiol 26:L17–L22
Brandsma AE, Ten Have-Opbroek AA, Vulto IM, Molenaar JC, Tibboel D 1994 Alveolar epithelial composition and architecture of the late fetal pulmonary acinus: an immunocytochemical and morphometric study in a rat model of pulmonary hypoplasia and congenital diaphragmatic hernia. Exp Lung Res 20: 491–515
Minoo P, Segura L, Coalson JJ, King RJ, DeLemos RA 1991 Alterations in surfactant protein gene expression associated with premature birth and exposure to hyperoxia. Am J Physiol 261:L386–L392
O'Toole SJ, Sharma A, Karamanoukian HL, Holm B, Azizkhan RG, Glick PL 1996 Tracheal ligation does not correct the surfactant deficiency associated with congenital diaphragmatic hernia. J Pediatr Surg 31: 546–550
Wilcox DT, Glick PL, Karamanoukian H, Rossman J, Morin FCr, Holm BA 1994 Pathophysiology of congenital diaphragmatic hernia. V. Effect of exogenous surfactant therapy on gas exchange and lung mechanics in the lamb congenital diaphragmatic hernia model. J Pediatr 124: 289–293
Glick PL, Stannard VA, Leach CL, Rossman J, Hosada Y, Morin FC, Cooney DR, Allen JE, Holm B 1992 Pathophysiology of congenital diaphragmatic hernia. II. The fetal lamb CDH model is surfactant deficient. J Pediatr Surg 27: 382–387
Delemos RA, Shermeta DW, Knelson JH 1970 Acceleration of appearance of pulmonary surfactant in the fetal lamb by the administration of corticosteroids. Am Rev Respir Dis 102: 459–461
Author information
Authors and Affiliations
Additional information
Supported by grants from the Quebec Pulmonary Association, the Téléthon de la Recherche sur les Maladies Infantiles, and a Canadian Medical Research Council groups grant. B.P. was supported by an award from the Queen Elizabeth II Research Fund.
Rights and permissions
About this article
Cite this article
Piedboeuf, B., Laberge, JM., Ghitulescu, G. et al. Deleterious Effect of Tracheal Obstruction on Type II Pneumocytes in Fetal Sheep. Pediatr Res 41, 473–479 (1997). https://doi.org/10.1203/00006450-199704000-00004
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199704000-00004
This article is cited by
-
Bronchopulmonary dysplasia is associated with polyhydramnios in a scan for novel perinatal risk factors
Pediatric Research (2023)
-
Fetal Laryngeal Stenosis/Atresia and Congenital High Airway Obstructive Syndrome (CHAOS): A Case Report
Journal of Perinatology (2005)
-
Lung Pathology in Patients with Congenital Diaphragmatic Hernia Treated with Fetal Surgical Intervention, Including Tracheal Occlusion
Pediatric and Developmental Pathology (2003)
-
The History of Congenital Diaphragmatic Hernia from 1850s to the Present
Journal of Perinatology (2002)