Abstract
Oral administration of foreign proteins, e.g. cow's milk (CM) proteins, stimulates the immune system and induces humoral and cellular immune response against these antigens in infants. Up-regulation of adhesion molecules is known to be associated with activation of the immune system. The purpose of the study was to examine whether orally administered CM proteins induce elevation in soluble adhesion molecules, i.e. intercellular adhesion molecule-1 (ICAM-1) and L-selectin, in infants. In a double-blind trial, 10 infants received CM-based formula and 10 infants casein hydrolysate formula until the age of 9 mo. The infants of mothers with insulin-dependent diabetes mellitus (IDDM) were recruited into a pilot study of a trial for primary prevention of IDDM by elimination of CM proteins from the diet during early infancy. A cord blood sample and peripheral blood samples were taken at the ages of 3, 6, 9, and 12 mo of age. The levels of soluble ICAM-1 and L-selectin were measured by ELISA. The levels of soluble ICAM-1 were higher at the ages of 3, 6, 9, and 12 mo in infants who received CM-based formula than in infants who received hydrolyzed formula (p = 0.05). Instead, no difference was found in the the levels of soluble L-selectin. The levels of soluble ICAM-1 and L-selectin were higher in all infants when compared with the levels reported in adults or to the levels seen in cord blood. Orally fed CM proteins induce an elevation in soluble ICAM-1 in infants. This may reflect the generation of an immune response against these proteins, because ICAM-1 has an important costimulatory role in lymphocyte activation.
Similar content being viewed by others
Main
Immunity to orally administered proteins has been studied intensively during recent years. Studies performed in animals suggest that early exposure to orally administered proteins initiates an immune response against these antigens, whereas in adult animals the same exposure leads to tolerance(1). Recently we reported that exposure to CM proteins in early infancy provoked both cellular and humoral immune responses, and that the cellular immune response later declined. When the same oral exposure was encountered later in childhood (after 9 mo) it led to weaker T and B cell responses(2). The induction of oral tolerance seems to be an age-dependent phenomenon in man, as well(2, 3). The primary mechanisms by which orally administered antigen induces priming of the immune system or tolerance are, however, poorly understood.
The generation of immune response against exogenous proteins requires binding of the antigen-specific T cell receptor to the peptide/human leukocyte antigen class II complex located on the antigen-presenting cells. In addition, accessory antigen nonspecific signals are required for the effective activation of T lymphocytes(4). Interaction of lymphocyte homing receptors with their endothelial counter receptors not only directs lymphocyte traffic into the tissues, but plays a role in cell-cell interactions that control the immune response. Both LFA-1 and its endothelial counter receptor, ICAM-1, have been shown to provide costimulatory signals for lymphocyte activation(5, 6).
The expression of ICAM-1 correlates with the severity of chronic inflammation in psoriatic lesions as well as in the airway epithelium of patients with asthma(7, 8). In addition, elevated levels of soluble adhesion molecules have been associated with many diseases, such as IDDM, melanoma, graft rejection, septicemia, and systemic lupus erythematosus(9–13).
Because oral exposure to CM proteins is an immunogenic stimulus in infancy and high levels of soluble adhesion molecules are associated with an ongoing immune process, we measured the levels of sICAM-1 and sL-selectin in infants exposed to either whole CM proteins or to hydrolyzed CM proteins.
METHODS
Subjects. Twenty newborn infants born to mothers with IDDM in the Department of Obstetrics, University of Helsinki, were recruited into the study. This was the pilot stage of a trial for primary prevention of IDDM by elimination of CM proteins from the diet in early infancy. Exclusive breast-feeding was encouraged; thereafter 10 infants received adapted CM-based formula (Enfamil, Mead Johnson, Evansville, IN; group I) and 10 infants casein hydrolysate formula (Nutramigen, Mead Johnson; group II) until the age of 9 mo. The trial was a double-blind, randomized study of two different nutritional supplements: whole CM protein-containing formula versus casein hydrolysate formula with peptides of a molecular mass of less than 1200 D. To ensure that the two formulas could not be distinguished by taste or smell, 25% of the Enfamil powder was replaced by Nutramigen. The mothers of all infants were advised not to give CM-containing infant food or beef to their children during the first 9 mo of life. The infants and their mothers visited the outpatient clinic of the Children's Hospital, University of Helsinki, at the ages of 3, 6, 9, and 12 mo; the dietary advisor had, in addition, phone contact with the mothers. The compliance of family was inquired at each time and only two (one in each group) minor unintentional failures of the diet were recorded. Blood samples were drawn at 3, 6, 9, and 12 mo of age. A cord blood sample was available from 13 infants; nine in group I and four in group II. There was no difference in gestational age, in birth weight, or in the history of infections between the groups. The data on birth weight, gestational age, and weight gain is shown in Table 1. Data with regard to age when the infants started to receive the formula are shown in Table 2, and there was no difference between the groups.
METHODS
Plasma samples from each age group (0, 3, 6, 9, and 12 mo of age) were run in the same plate to assess sICAM-1 and sL-selectin levels. These concentrations were measured by specific ELISA kits purchased from Bender Medsystems, Vienna, Austria. Briefly, diluted plasma samples (1:20 for sICAM-1 assay and 1:200 for sL-selectin assay) were added as duplicates to the polystyrene microtiter plates, which were coated with a specific MAb. A specific horseradish peroxidase-conjugated second MAb was added to the wells, and the plates were incubated for 1 h in the ICAM-1 assay and for 2 h in the L-selectin assay at room temperature. After this first incubation, the wells were washed three times. After incubating the plates with a combination of substrate 1 (tetramethyl-benzidine) and substrate 2 (0.02% buffered hydrogen peroxide), the process was stopped with sulfuric acid, and the absorbances were measured at 450-nm wavelength by Multiskan MS (Labsystems, Helsinki, Finland). A dilution series of sICAM-1 or sL-selectin standards was run in each plate, and a standard curve was calculated. The levels of soluble adhesion molecules as nanograms/mL were calculated from the standard curve.
Statistical analyses. The difference in the levels of soluble adhesion molecules between the groups was analyzed by Mann-Whitney U test and by regression analysis for repeated measurements. The correlation between sICAM and sL-selectin levels was tested by a Spearman correlation test.
RESULTS
All of the infants except one received the test formula before the age of 6 mo (mean age 1.2, range 0-7.5 mo). No difference was observed in the duration of breast feeding nor in the age of starting the substitution formula between the two groups (Table 2). One infant in group I was exposed to the CM formula for the first time at the age of 7.5 mo and was excluded from the analyses performed at the ages of 3 and 6 mo. One child in group I developed CM allergy at the age of 7 mo and was withdrawn from the study at that time. Another child in group II progressed to manifest diabetes at the age of 14 mo.
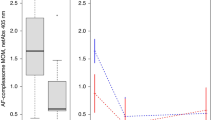
The levels of sICAM-1 were higher throughout the study in infants who received whole CM protein-containing formula (group I) than in infants who received hydrolysate formula (group II): at 3 mo medians (range) were 453(216-578) ng/mL (n = 8) versus 415 (80-546) ng/mL(n = 9) (Mann-Whitney U test p = 0.34), at 6 mo 332 (206-460) ng/mL (n = 8) versus 275 (147-307) ng/mL(n = 9) (p = 0.15), at 9 mo 482 (361-694) ng/mL(n = 8) versus 357 (158-678) ng/mL (n = 8)(p = 0.17), and at 12 mo 514 (374-934) ng/mL (n = 9)versus 365 (174-749) ng/mL (n = 9) (p = 0.04). The levels of sICAM-1 according to the age of the infants are shown inFigure 1. The difference in the levels of sICAM-1 during the follow-up was significant between the two diet groups (p = 0.05, regression analysis). The median levels of sICAM-1 in the cord blood samples did not differ significantly between the groups, being 146 (range 135-222) ng/mL (n = 9) in group I and 130 (119-169) ng/mL (n = 4) in group II.
Levels of sICAM-1 in infants who received cows' milk-based formula (filled squares) and in infant who receive hydrolyzed formula (open squares) at 3, 6, 9, and 12 mo of age. Medians of the two groups are shown. Levels of sICAM-1 of the individual who was diagnosed with CM allergy at the age of 7 mo (arrows) and of the individual who progressed to manifest IDDM at 14 mo of age(arrowheads) are also indicated.
The levels of sL-selectin did not differ between the diet groups at any age. At 3 mo the median (range) was 2300 (1700-5600) ng/mL (group I)versus 3200 (1120-5600) ng/mL (group II) (p = 0.89 Mann-Whitney U test); at 6 mo, 2100 (1700-5100) ng/mLversus 2200 (1240-2880) ng/mL (p = 0.53); at 9 mo, 2640(1440-9600) ng/mL versus 3800 (1400-11000) ng/mL (p = 0.92); and at 12 mo, 2500 (1720-4600) ng/mL versus 2360 (1540-4080) ng/mL (p = 0.35). The median level of sL-selectin in the cord blood samples was 658 (range 550-950) ng/mL. There was no significant correlation between the sICAM-1 and sL-selectin levels (r = 0.168, Spearman correlation test).
The sICAM-1 and sL-selectin levels in the individual who progressed to manifest diabetes did not differ from the levels of the rest of the study population. This was also true in the case of CM allergy. The data on these two individuals are shown in Figure 1.
DISCUSSION
In the present pilot trial we had the unique opportunity to study the development of an immune response to orally administered antigens in infants. We found higher levels of sICAM-1 in infants who were exposed to whole CM proteins than in infants who received hydrolyzed CM proteins. These hydrolyzed proteins have been shown to have greatly reduced immunogenicity(14). The concentrations of sL-selectin did not differ between the two groups. Interestingly, the difference in sICAM-1 levels between the groups remained unchanged throughout the study until the age of 12 mo, although CM/diet manipulation ceased at 9 mo of age, after which also the rest of the infants were introduced to whole CM proteins. This finding suggests that the immune activation in infants of group II, who started to receive whole CM proteins only after the age of 9 mo, was suppressed when compared with that of group I. This explanation is supported by the earlier observations of a suppressed cellular immune response to CM proteins in group II(2). On the other hand, high levels of sICAM-1 in group I at 12 mo of age could reflect the development of active immunosuppression. We have shown that the proliferative cellular immune response to CM β-lactoglobulin later declined in group I infants, supporting the theory of induction of oral tolerance(2).
In healthy adults the mean level of sICAM-1 varies between 100 and 200 ng/mL(15). We observed that sICAM-1 concentrations were clearly higher in all infants, the mean being 393 ng/mL. This is in agreement with earlier findings of higher levels of sICAM-1 in healthy infants and young children compared with older children and adults(16, 17). In the cord blood samples the levels of ICAM-1 were significantly lower. The elevated levels of sICAM-1 may result from a myriad of primary immune responses activated by contacts with new antigens. In adults, comparable elevated levels of soluble adhesion molecules have been shown in chronic inflammatory diseases such as rheumatoid arthritis and vasculitis(18). Infants are continuously exposed to new oral antigens and infectious agents. In Finland, infants are also vaccinated according to standards of the national program: bacillus Calmette-Guérin (tuberculosis) at birth; pertussis-diphtheria-tetanus at 3, 4, and 5 mo; Haemophilus influenzae type B at 4 and 6 mo, and poliovirus at 6 and 12 mo of age. There was no difference in the vaccination schedule between the two diet groups studied (data not shown).
The results of our study suggest that maturation of the immune system in the infants exposed to new antigens provokes an elevation in sICAM-1. Lymphocytes, monocytes, and vascular endothelium all have ICAM-1 surface molecules. The elevated levels of sICAM-1 found in group I could arise from monocytes which act as antigen-presenting cells in immune activation, or they might also arise from activated lymphocytes. The endothelium seems a less likely source of sICAM-1.
Both ICAM-1 and its counter-receptor LFA-1 play a crucial role in lymphocyte activation by providing costimulatory signals. Anti-ICAM-1 and anti-LFA-1 antibodies inhibit a mixed lymphocyte reaction and T cell-mediated antibody production of B cells in animal models(5, 6). Roep et al.(15) reported that anti-ICAM-1 and anti-LFA-1 MAb suppressed autoreactive T cell proliferation. They speculated that the elevated levels of ICAM-1 in the serum of subjects at risk for developing IDDM may down-regulate inflammation. The elevated levels of soluble adhesion molecules in all infants may reflect an ongoing immune activation and may have a physiologic role in down-regulation of inflammation in infancy. The even higher levels of sICAM-1 in infants receiving whole CM proteins associate with the development of systemic immune response to these proteins in early infancy. On the other hand, introduction of CM proteins may increase the permeability of the intestine to other oral antigens, thereby enhancing the immune reactions to food antigens in general. This kind of effect has been shown in experimental animals(19, 20). Thus, a high level of sICAM-1 in plasma may not only reflect the immune response produced by CM antigens but by other oral antigens, too.
L-Selectin expressed by granulocytes, lymphocytes, and monocytes is responsible for initial leukocyte attachment to inflamed endothelium and high endothelial venules of peripheral lymph nodes. After leukocyte activation, L-selectin is rapidly shed from the cell surface(21). A mean level of 1.6 ± 0.8 μg/mL has been found in normal blood donors(22). The mean levels of sL-selectin were twice as high in all infants, with no difference appearing between the study groups. Accordingly, L-selectin may not have such an important role in lymphocyte activation as ICAM-1 and LFA-1(5, 6).
We showed that the levels of soluble adhesion molecules in healthy infants were higher than the values reported in adults. This finding may reflect maturation of the immune response to new antigens encountered in infancy. In addition, we found that oral administration of CM proteins in early infancy is a strong immunogenic stimulus which induces a systemic immune response to these antigens(2) as well as elevation of sICAM-1. The elimination of CM proteins from the diet of infants during the first months of life results in lower levels of sICAM-1, even after CM is started at a later age, showing that the later introduction of immunogenic antigens into the diet results in a less marked stimulation of the immune system.
Abbreviations
- CM:
-
cow's milk
- ICAM-1:
-
intercellular adhesion molecule-1
- IDDM:
-
insulin-dependent diabetes mellitus
- LFA-1:
-
lymphocyte function-associated antigen-1
- sICAM-1:
-
soluble ICAM-1
- sL-selectin:
-
soluble L-selectin
References
Strobel S, Ferguson A 1984 Immune responses to fed protein antigens in mice. 3. Systemic tolerance or priming is related to age at which antigen is first encountered. Pediatr Res 18: 588–594
Vaarala O, Saukkonen T, Savilahti E, Klemola T, Akerblom HK 1995 Development of immune response to cow milk proteins in infants receiving cow milk formula or hydrolysed formula. J Allergy Clin Immunol 96: 917–923
Husby S, Mestecky J, Moldoveanu Z, Holland S, Elson CO 1994 Oral tolerance in humans T cell but not B cell tolerance after antigen feeding. J Immunol 152: 4663–4670
Springer TA 1990 Adhesion receptors of the immune system. Nature 346: 425–434
Fischer A, Durandy A, Sterkers G, Griscelli C 1986 Role of LFA-1 molecule in cellular interactions required for antibody production in humans. J Immunol 136: 3198–3203
Boyd AW, Wawryk SO, Burns GF, Fecondo JV 1988 Intercellular adhesion molecule 1 (ICAM-1) has a central role in cell-cell contact-mediated immune mechanisms. Proc Natl Acad Sci USA 85: 3095–3099
Lisby S, Ralfkiaer E, Rothlein R, Vejlsgaard GL 1989 Intercellular adhesion molecule-I (ICAM-I) expression correlated to inflammation. Br J Dermatol 120: 479–484
Wegner CD, Gundel RH, Reilly P, Haynes N, Letts G, Rothlein R 1990 Intercellular adhesion molecule-1 (ICAM-1) in the pathogenesis of asthma. Science 247: 456–459
Adams DH, Hubscher J, Shaw R, Rothlein R, Neuberger JM 1989 Intercellular adhesion molecule 1 on liver allografts during rejection. Lancet 2: 1122–1124
Gearing AJH, Newman W 1993 Circulating adhesion molecules in disease. Immunol Today 14: 506–512
Lampeter ER, Kishimoto TK, Rothlein R, Mainolfi EA, Bertrams J, Kolb H, Martin S 1992 Elevated levels of circulating adhesion molecules in IDDM patients and in subjects at risk for IDDM. Diabetes 41: 1668–1671
Harning R, Mainolfi EA, Bystryn J-C, Henn M, Merluzzi VJ, Rothlein R 1991 Serum levels of circulating intercellular adhesion molecule 1 in human malignant melanoma. Cancer Res 51: 5003–5005
Shimizu Y, Newman W, Tanaka Y, Shaw S 1992 Lymphocyte interactions with endothelial cells. Immunol Today 13: 106–112
Lee Y-H 1992 Food-processing approaches to altering allergenic potential of milk-based formula. J Pediatr 121: 47–50
Roep BO, Heidenthal E, de Vries RRP, Kolb Hubert, Martin S 1994 Soluble forms of intercellular adhesion molecule-1 in insulin-dependent diabetes mellitus. Lancet 343: 1590–1592
Furukawa S, Imai K, Matsubara T, Yone K, Yachi A, Okumara K, Yabuta K 1992 Increased levels of circulating intercellular adhesion molecule 1 in Kawasaki disease. Arthritis Rheum 35: 672–677
Nash MC, Shah V, Dillon MJ 1995 Soluble cell adhesion molecules and von Willebrand factor in children with Kawasaki disease. Clin Exp Immunol 101: 13–17
Blann AD, McCollum CN 1995 Circulating adhesion molecules in inflammatory and atherosclerotic vascular disease. Immunol Today 16: 251–252
Walker WA 1978 Antigen handling by the gut. Arch Dis Child 53: 527–531
Weaver LT, Laker MF, Nelson R, Lucas A 1987 Milk feeding and changes in the intestinal permeability and morphology in the newborn. J Pediatr Gastroenterol Nutr 6: 351–358
Kishimoto TK, Jutila MA, Butcher EC 1990 Identification of a human peripheral lymph node homing receptor: a rapidly down-regulated adhesion molecule. Proc Natl Acad Sci USA 87: 2244–2248
Schleiffenbaum B, Spertini O, Tedder TF 1992 Soluble L-selectin is present in human plasma at high levels and retains functional activity. J Cell Biol 119: 229–238
Author information
Authors and Affiliations
Additional information
Supported by grants from the Foundation for Diabetes Research, Finland(O.V., H.K.Å.), the Finnish Cultural Foundation (O.V.), Finska Läkaresällskapet (O.V), the Juvenile Diabetes Foundation International Grant 193101 (H.K.Å.), the Sigrid Juselius Foundation(E.S., H.K.Å.), the Novo Nordisk Fund (H.K.Å.), the University of Helsinki (H.K.Å.), and the Dorothea Olivia, Karl Walter, and Jarl Walter Pèrklen Foundation (H.K.Å.). O.V. is a recipient of a Research Fellowship from the Juvenile Diabetes Foundation International J.P. is a recipient of a Postgraduate Fellowship from the University of Helsinki.
Rights and permissions
About this article
Cite this article
Paronen, J., Vaarala, O., Savilahti, E. et al. Soluble Adhesion Molecules and Oral Antigen Feeding in Infants. Pediatr Res 40, 276–279 (1996). https://doi.org/10.1203/00006450-199608000-00014
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199608000-00014
This article is cited by
-
Is the origin of type 1 diabetes in the gut?
Immunology & Cell Biology (2012)