Abstract
Extract: At least 15 apparently inherited disorders of branched chain amino acid catabolism are now known; the 12th in chronological order of discovery is described in this report. It is a partial defect of the pathway of isoleucine oxidation beyond the level of oxidative decarboxylation and prior to the oxidation of propionate. The impairment of isoleucine catabolism appears to be situated at the “thiolase” reaction which converts α-methylacetoacetyl coenzyme A (CoA) to propionyl-CoA and acetyl-CoA.
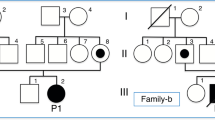
Two pedigrees (B and M) were investigated in detail. A third (S pedigree) has been brought to our attention for analysis of metabolites in urine but we have not performed additional studies in the latter. Each propositus was ascertained because of intermittent, odorless metabolic acidosis usually precipitated by intercurrent infection. Lethargy and coma occurred frequently during the periods of acidosis. One M sib, also presumably affected, died abroad in such an episode. Symptoms can be ameliorated by a low protein diet and careful attention to the management of intercurrent illness.
A large excess of α-methyl-β-hydroxybutyrate and a seemingly smaller excess of α-methylacetoacetate is present at all times in the urine of the three propositions. The M and S propositi also excrete N-tiglylglycine. The amounts of these unusual metabolites increase severalfold during acidosis and after a dietary load of l-isoleucine (75 mg/kg, 3 times daily for 2 days). The urine also contains butanone, particularly during acidosis. The amount of propionate and of glycine and other amino acids in blood and urine is always normal in our patients. Oxidation of l-isoleucine-U-14C to CO2 by cultured skin fibroblasts is about 45% of normal in the B propositus. The precise nature and location of the enzyme defect awaits clarification.
Studies of family members reveal that presumed obligate heterozygotes excrete a small excess of α-methyl-β-hydroxybutyrate at all times; the amount can be increased by L-isoleucine feeding. The condition is apparently inherited in autosomal recessive fashion. It is likely that more than one form of mutant allele is responsible for the condition, as it is found in the three different pedigrees described here.
Speculation: Investigation of “unexplained,” intermittent metabolic acidosis in childhood has led to the discovery of a “new” disorder of branched chain amino acid catabolism. Gas chromatography coupled with mass spectrometry were important aids to the diagnosis. Rapid escalation of acidosis during catabolic episodes encourages one to suspect that specific metabolites, themselves accumulating during episodic illness, may further inhibit the mutant enzyme. A temperature-sensitive mutant enzyme was not identified in cultured skin fibroblasts.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Daum, R., Scriver, C., Mamer, O. et al. An Inherited Disorder of Isoleucine Catabolism Causing Accumulation of α-Methylacetoacetate and α-Methyl-β-hydroxybutyrate, and Intermittent Metabolic Acidosis. Pediatr Res 7, 149–160 (1973). https://doi.org/10.1203/00006450-197303000-00007
Issue Date:
DOI: https://doi.org/10.1203/00006450-197303000-00007
Keywords
This article is cited by
-
2-methylacetoacetyl-coenzyme A thiolase (beta-ketothiolase) deficiency: one disease - two pathways
Orphanet Journal of Rare Diseases (2020)
-
Recent advances in understanding beta-ketothiolase (mitochondrial acetoacetyl-CoA thiolase, T2) deficiency
Journal of Human Genetics (2019)
-
Enzymes involved in branched-chain amino acid metabolism in humans
Amino Acids (2017)
-
Ketone body metabolism and its defects
Journal of Inherited Metabolic Disease (2014)
-
Glial Metabolism of Isoleucine
Neurochemical Research (2009)