Abstract
In 2009, castration-resistant metastatic prostate cancer continues to account for more deaths in US men than any other cancer apart from lung cancer. Although novel targeted molecular, hormonal and immunologic agents are accelerating in their development in this disease, docetaxel and prednisone remain the standard palliative regimen for the majority of men who have progressed despite hormonal therapies. Thus, understanding the practical and often subtle issues of docetaxel initiation, duration of therapy, cessation of therapy and treatment holidays is critical for the informed use of this US Food and Drug Administration-approved regimen. In this review we address these topics in light of prognostic and predictive factors to help guide the rational use of docetaxel chemotherapy in men with this aggressive disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ries LAG, Melbert D, Krapcho M, Stinchcomb DG, Howlader N, Horner MJ et al. (eds). SEER Cancer Statistics Review 1975–2005. National Cancer Institute: Bethesda, MD. http://seer.cancer.gov/csr/1975_2005/. based on November 2007 SEER data submission, posted to the SEER website, 2008.
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ . Cancer statistics, 2009. CA Cancer J Clin 2009; 59: 225–249.
Cancer Research UK. CancerStats Key Facts on Prostate Cancer. Available from: http://info.cancerresearchuk.org/cancerstats/types/prostate/. (last accessed 23 September 2009).
Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Kim KN et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004; 351: 1502–1512.
Petrylak DP, Tangen CM, Hussain MH, Lara Jr PN, Jones JA, Taplin ME et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med 2004; 351: 1513–1520.
Han M, Partin AW, Pound CR, Epstein JI, Walsh PC . Long-term biochemical disease-free and cancer-specific survival following anatomic radical retropubic prostatectomy. The 15-year Johns Hopkins experience. Urol Clin North Am 2001; 28: 555–565.
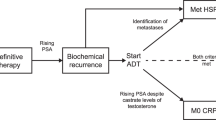
Smith MR, Kabbinavar F, Saad F, Hussain A, Gittelman MC, Bilhartz DL et al. Natural history of rising serum prostate-specific antigen in men with castrate nonmetastatic prostate cancer. J Clin Oncol 2005; 23: 2918–2925.
National Comprehensive Cancer Network. Clinical practice guidelines in oncology: Prostate cancer. National Comprehensive Cancer Network Inc. Version 2; 2009.
Oh WK . Secondary hormonal therapies in the treatment of prostate cancer. Urology 2002; 60 (3 Suppl 1): 87–92.
Tannock IF, Osoba D, Stockler MR, Ernst DS, Neville AJ, Moore MJ et al. Chemotherapy with mitoxantrone plus prednisone or prednisone alone for symptomatic hormone-resistant prostate cancer: a Canadian randomised trial with palliative end points. J Clin Oncol 1996; 14: 1756–1764.
Docetaxel prescribing information. Retrieved 22 May 2008 from http://www.fda.gov/cder/foi/label/2007/020449s045lbl.pdf.
Docetaxel approval [last accessed 23 September 2009]. U.S. Department of Health and Human Services. Available from: www.accessdata.fda.gov/scripts/cder/drugsatfda/index.cfm?fuseaction=Search.DrugDetails.
Berthold DR, Pond GR, Soban F, de Wit R, Eisenberger M, Tannock IF . Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer: updated survival in the TAX 327 study. J Clin Oncol 2008; 26: 242–246.
Collette L, Burzykowski T, Carroll KJ, Newling D, Morris T, Schröder FH et al. Is prostate specific antigen a valid surrogate end point for survival in hormonally treated patients with metastatic prostate cancer? Joint research of the European Organisation for Research and Treatment of Cancer, the Limburgs Universitair Centrum, and AstraZeneca Pharmaceuticals. J Clin Oncol 2005; 23: 6139–6148.
Petrylak DP, Ankerst DP, Jiang CS, Tangen CM, Hussain MH, Lara Jr PN et al. Evaluation of prostate-specific antigen declines for surrogacy in patients treated on SWOG 99-16. J Natl Cancer Inst 2006; 98: 516–521.
Armstrong AJ, Garrett-Mayer E, Ou Yang YC, Carducci MA, Tannock I, de Wit R et al. Prostate-specific antigen and pain surrogacy analysis in metastatic hormone-refractory prostate cancer. J Clin Oncol 2007; 25: 3965–3970 . Erratum in: J Clin Oncol 2008; 26: 820 Reprinted with permission. ©2008 American Society of Clinical Oncology. All rights reserved.
Heidenreich A, Aus G, Bolla M, Joniau S, Matveev VB, Schmid HP et al. EAU guidelines on prostate cancer. Eur Urol 2008; 53: 68–80.
Armstrong AJ, Garrett-Mayer ES, Yang YO, de Wit R, Tannock IF, Eisenberger M . A contemporary prognostic nomogram for men with hormone-refractory metastatic prostate cancer: a TAX327 study analysis. Clin Cancer Res 2007; 13: 6396–6403.
Halabi S, Vogelzang NJ, Kornblith AB, Ou SS, Kantoff PW, Dawson NA et al. Pain predicts overall survival in men with metastatic castration-refractory prostate cancer. J Clin Oncol 2008; 26: 2544–2549.
Berthold DR, Pond GR, Roessner M, de Wit R, Eisenberger M, Tannock IF, on behalf of the TAX-327 Investigators. Treatment of hormone-refractory prostate cancer with docetaxel or mitoxantrone: relationships between prostate-specific antigen, pain, and quality of life response and survival in the TAX-327 study. Clin Cancer Res 2008; 14: 2763–2767.
Armstrong AJ, Eisenberger MA, Garrett-Mayer ES, Ou Yang Y, Carducci MA, De Wit R et al. Analysis of prostate-specific antigen decline as a surrogate for overall survival in metastatic hormone-refractory prostate cancer (HRPC). J Clin Oncol 2007; 25 (Suppl 18): (abstract 2009).
Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Miller MC et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med 2004; 351: 781–791.
Moreno J, De Bono JS, Shaffer D, Montgomery B, Miller MC, Tissing H et al. Multi-center study evaluating circulating tumor cells (CTCs) as a surrogate for survival in men treated for castration refractory prostate cancer (CRPC). J Clin Oncol 2007; 25 (Suppl 18): (abstract 5016).
Danila DC, Heller G, Gignac GA, Gonzalez-Espinoza R, Anand A, Tanaka E et al. Circulating tumor cell number and prognosis in progressive castration-resistant prostate cancer. Clin Cancer Res 2007; 13: 7053–7058.
De Bono JS, Scher HL, Montgomery RB, Parker C, Miller MC, Tissing H et al. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res 2008; 14: 6302–6309.
Attard G, Swennenhuis JF, Olmos D, Reid AHM, Vickers E, A’Hern R et al. Characterization of ERG, AR and PTEN gene status in circulating tumor cells from patients with castration-resistant prostate cancer. Cancer Res 2009; 69: 2912–2918.
Smirnov DA, Zweitzig DR, Foulk BW, Miller MC, Doyle GV, Pienta KJ et al. Global gene expression profiling of circulating tumor cells. Cancer Res 2005; 65: 4993–4997.
George DJ, Halabi S, Shepard TF, Sanford B, Vogelzang NJ, Small EJ et al. The prognostic significance of plasma interleukin-6 levels in patients with metastatic hormone-refractory prostate cancer: results from Cancer and Leukemia Group B 9480. Clin Cancer Res 2005; 11: 1815–1820.
Halabi S, Small EJ, Kantoff PW, Kattan MW, Kaplan EB, Dawson NA et al. Prognostic model for predicting survival in men with hormone-refractory metastatic prostate cancer. J Clin Oncol 2003; 21: 1232–1237.
Smaletz O, Scher HI, Small EJ, Verbel DA, McMillan A, Regan K et al. Nomogram for overall survival of patients with progressive metastatic prostate cancer after castration. J Clin Oncol 2002; 20: 3972–3982.
Armstrong AJ, Halabi S, Tannock IF, George DJ, DeWit R, Eisenberger M . Development of risk groups in metastatic castration-resistant prostate cancer (mCRPC) to facilitate the identification of active chemotherapy regimens. J Clin Oncol 2009; 27 (Suppl 15): (abstract 5137).
George DJ, Halabi S, Shepard TF, Vogelzang NJ, Hayes DF, Small EJ et al. Prognostic significance of plasma vascular endothelial growth factor levels in patients with hormone-refractory prostate cancer treated on Cancer and Leukemia Group B 9480. Clin Cancer Res 2001; 7: 1932–1936.
Ghossein RA, Scher HI, Gerald WL, Kelly WK, Curley T, Amsterdam A et al. Detection of circulating tumor cells in patients with localized and metastatic prostatic carcinoma: clinical implications. J Clin Oncol 2005; 13: 1195–1200.
Lin AM, Ryan CJ, Small EJ . Intermittent chemotherapy for metastatic hormone refractory prostate cancer. Crit Rev Oncol Hematol 2007; 61: 243–254.
Halabi S, Vogelzang NJ, Ou SS, Owzar K, Archer L, Small EJ . Progression-free survival as a predictor of overall survival in men with castrate-resistant prostate cancer. J Clin Oncol 2009; 27: 2766–2771.
Hussain M, Goldman B, Tangen C, Higano CS, Petrylak DP, Wilding G et al. Prostate-specific antigen progression predicts overall survival in patients with metastatic prostate cancer: data from Southwest Oncology Group Trials 9346 (Intergroup Study 0162) and 9916. Clin Oncol 2009; 27: 2450–2456.
Scher HI, Halabi S, Tannock I, Morris M, Sternberg CN, Carducci MA et al. Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the Prostate Cancer Clinical Trials Working Group. J Clin Oncol 2008; 26: 1148–1159.
Scher HI, Warren M, Heller G . The association between measures of progression and survival in castrate-metastatic prostate cancer. Clin Cancer Res 2007; 13: 1488–1492.
Armstrong AJ, Garrett-Mayer ES, De Wit R, Ou Yang Y, Tannock I, Eisenberger M . Limitations of the current progression-free survival (PFS) definition in hormone-refractory prostate cancer (HRPC): benefit associated with continuation of docetaxel beyond TAX 327 protocol-defined disease progression. Proc Am Soc Clin Oncol Prostate Cancer Symposium 2007; (abstract 223).
Armstrong AJ, Garrett-mayer E, de Wit R, Tannock I, Eisenberger M . Prediction of survival following first-line chemotherapy in men with castration-resistant metastatic prostate cancer. Clin Cancer Res; e-pub ahead of print, 15 December 2009
Ramiah V, George DJ, Armstrong AJ . Clinical endpoints for drug development in prostate cancer. Curr Opin Urol 2008; 18: 303–308.
Berry W, Dakhil S, Gregurich M, Asmar L . Phase II trial of single-agent weekly docetaxel in hormone-refractory, symptomatic, metastatic carcinoma of the prostate. Semin Oncol 2001; 28: 8–15.
Beer TM, Pierce WC, Lowe BA, Henner WD . Phase II study of weekly docetaxel in symptomatic androgen-independent prostate cancer. Ann Oncol 2001; 12: 1273–1279.
Joshua AM, Nordman I, Venkataswaran R, Clarke S, Stockler MR, Boyer MJ . Weekly docetaxel as second line treatment after mitoxantrone for androgen-independent prostate cancer. Intern Med J 2005; 35: 468–472.
Beer TM, Garzotto M, Henner WD, Eilers KM, Wersinger EM . Intermittent chemotherapy in metastatic androgen-independent prostate cancer. Br J Cancer 2003; 89: 968–970.
Beer TM, Ryan CW, Venner PM, Petrylak DP, Chotta G, Ruether J et al. Intermittent chemotherapy in metastatic androgen-independent prostate cancer (AIPC): initial results from ASCENT. American Society of Clinical Oncology Multi-disciplinary Prostate Cancer Symposium 2006; (abstract 4518).
Miller K, Wulfing C, Lehmann J, Johannsen M, Heidenreich A, Hegele A et al. Weekly docetaxel plus estramustine for hormone-refractory prostate cancer (HRPC) with intermittent repetition: preliminary results of a multicenter phase II study (AUO AP33/02). J Clin Oncol 2005; 23 (Suppl 16): (abstract 4613).
Small EJ, Reese DM, Um B, Whisenant S, Dixon SC, Figg WD . Therapy of advanced prostate cancer with granulocyte macrophage colony-stimulating factor. Clin Cancer Res 1999; 5: 1738–1744.
Dreicer R, See WA, Klein EA . Phase II trial of GM-CSF in advanced prostate cancer. Invest New Drugs 2001; 19: 261–265.
Rini BI, Weinberg V, Bok R, Small EJ . Prostate-specific antigen kinetics as a measure of the biologic effect of granulocyte–macrophage colony-stimulating factor in patients with serologic progression of prostate cancer. J Clin Oncol 2003; 21: 99–105.
Schellhammer PF, Higano C, Berger ER, Shore N, Small E, Penson D et al. A randomized, double-blind, placebo-controlled, multi-center,. American Urological Association annual meeting 2009: (abstract LBA9).
Acknowledgements
Editorial support was provided by Ghzaleh Masnavi, PhD, at Adelphi Communications and funded by Sanofi-aventis US.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors were fully responsible for all content and editorial decisions, and did not receive financial support or compensation related to the development of the paper.
Rights and permissions
About this article
Cite this article
Armstrong, A., George, D. Optimizing the use of docetaxel in men with castration-resistant metastatic prostate cancer. Prostate Cancer Prostatic Dis 13, 108–116 (2010). https://doi.org/10.1038/pcan.2009.62
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pcan.2009.62
Keywords
This article is cited by
-
Chemotherapy for prostate cancer: when should a urologist refer a patient to a medical oncologist?
Prostate Cancer and Prostatic Diseases (2013)
-
De dilemma’s van de nieuwe behandelmogelijkheden voor castratieresistent prostaatcarcinoom (CRPC); een filosofische kijk op de toekomst
Tijdschrift voor Urologie (2012)
-
Second-line chemotherapy in metastatic docetaxel-resistant prostate cancer: a review
Medical Oncology (2012)