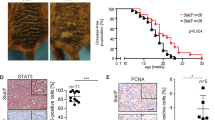
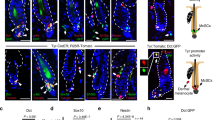
Abstract
Tumor initiation and metastasis formation in many cancers have been associated with emergence of a gene expression program normally active in embryonic or organ-specific stem cells. In particular, the stem cell transcription factor Sox2 is not only expressed in a variety of tumors, but is also required for their formation. Melanoma, the most aggressive skin tumor, derives from melanocytes that during development originate from neural crest stem cells. While neural crest stem cells do not express Sox2, expression of this transcription factor has been reported in melanoma. However, the role of Sox2 in melanoma is controversial. To study the requirement of Sox2 for melanoma formation, we therefore performed CRISPR-Cas9-mediated gene inactivation in human melanoma cells. In addition, we conditionally inactivated Sox2 in a genetically engineered mouse model, in which melanoma spontaneously develops in the context of an intact stroma and immune system. Surprisingly, in both models, loss of Sox2 did neither affect melanoma initiation, nor growth, nor metastasis formation. The lack of a tumorigenic role of Sox2 in melanoma might reflect a distinct stem cell program active in neural crest stem cells and during melanoma formation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Sarkar A, Hochedlinger K . The Sox family of transcription factors: versatile regulators of stem and progenitor cell fate. Cell Stem Cell 2013; 12: 15–30.
Avilion AA, Nicolis SK, Pevny LH, Perez L, Vivian N, Lovell-Badge R . Multipotent cell lineages in early mouse development on SOX2 function. Genes Dev 2003; 17: 126–140.
Takahashi K, Yamanaka S . Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006; 126: 663–676.
Masui S, Nakatake Y, Toyooka Y, Shimosato D, Yagi R, Takahashi K et al. Pluripotency governed by Sox2 via regulation of Oct3/4 expression in mouse embryonic stem cells. Nat Cell Biol 2007; 9: 625–635.
Favaro R, Valotta M, Ferri ALM, Latorre E, Mariani J, Giachino C et al. Hippocampal development and neural stem cell maintenance require Sox2-dependent regulation of Shh. Nat Neurosci 2009; 12: 1248–1256.
Taranova OV . SOX2 is a dose-dependent regulator of retinal neural progenitor competence. Genes Dev 2006; 20: 1187–1202.
Ferri ALM . Sox2 deficiency causes neurodegeneration and impaired neurogenesis in the adult mouse brain. Development 2004; 131: 3805–3819.
Bylund M, Andersson E, Novitch BG, Muhr J . Vertebrate neurogenesis is counteracted by Sox1-3 activity. Nat Neurosci 2003; 6: 1162–1168.
Arnold K, Sarkar A, Yram MA, Polo JM, Bronson R, Sengupta S et al. Sox2+ adult stem and progenitor cells are important for tissue regeneration and survival of mice. Cell Stem Cell 2011; 9: 317–329.
Ambrosetti D, Favaro R, Nicolis SK, Mansukhani A, Basilico C . The transcription factor Sox2 is required for osteoblast self-renewal. Cell Death Differ 2010; 17: 1345–1353.
Vanner RJ, Remke M, Gallo M, Selvadurai HJ, Coutinho F, Lee L et al. Article quiescent Sox2+cells drive hierarchical growth and relapse in sonic hedgehog subgroup medulloblastoma. Cancer Cell 26: 33–47.
Boumahdi S, Driessens G, Lapouge G, Rorive S, Nassar D, Le Mercier M et al. SOX2 controls tumour initiation and cancer stem-cell functions in squamous-cell carcinoma. Nature 2014; 511: 246–253.
Ferone G, Song J, Sutherland KD, Proost N, Gargiulo G, Berns A et al. SOX2 is the determining oncogenic switch in promoting lung squamous cell carcinoma from different cells of origin article SOX2 is the determining oncogenic switch in promoting lung squamous cell carcinoma from different cells of origin. Cancer Cell 2016; 30: 519–532.
Li H, Collado M, Villasante A, Matheu A, Lynch CJ, Cañamero M et al. p27Kip1 directly represses Sox2 during embryonic stem cell differentiation. Cell Stem Cell 2012; 11: 845–852.
Kareta MS, Gorges LL, Hafeez S, Benayoun BA, Marro S, Zmoos A-F et al. Inhibition of pluripotency networks by the Rb tumor suppressor restricts reprogramming and tumorigenesis. Cell Stem Cell 2015; 16: 39–50.
Ikushima H, Todo T, Ino Y, Takahashi M, Miyazawa K, Miyazono K . Article autocrine TGF-b signaling maintains tumorigenicity of glioma-initiating cells through Sry-related HMG-Box factors. Stem Cell 2009; 5: 504–514.
Gangemi RMR, Griffero F, Marubbi D, Perera M, Capra MC, Malatesta P et al. SOX2 silencing in glioblastoma tumor-initiating cells causes stop of proliferation and loss of tumorigenicity. Stem Cells 2009; 27: 40–48.
Favaro R, Appolloni I, Pellegatta S, Sanga AB, Pagella P, Gambini E et al. Sox2 is required to maintain cancer stem cells in a mouse model of high-grade oligodendroglioma. Cancer Res 2014; 74: 1833–1844.
Sarkar A, Huebner AJ, Sulahian R, Sadreyev R, Shivdasani RA, Sarkar A et al. Sox2 suppresses gastric tumorigenesis in mice article Sox2 suppresses gastric tumorigenesis in mice. Cell Rep 2016; 16: 1–13.
Laga AC, Zhan Q, Weishaupt C, Ma J, Frank MH, Murphy GF . SOX2 and nestin expression in human melanoma: an immunohistochemical and experimental study. Exp Dermatol 2011; 20: 339–345.
Laga AC, Lai C-Y, Zhan Q, Huang SJ, Velazquez EF, Yang Q et al. Expression of the embryonic stem cell transcription factor SOX2 in human skin. Am J Pathol 2010; 176: 903–913.
Santini R, Pietrobono S, Pandolfi S, Montagnani V, D’Amico M, Penachioni JY et al. SOX2 regulates self-renewal and tumorigenicity of human melanoma-initiating cells. Oncogene 2014; 33: 4697–4708.
Girouard SD, Laga AC, Mihm MC, Scolyer RA, Thompson JF, Zhan Q et al. SOX2 contributes to melanoma cell invasion. Lab Invest 2012; 92: 362–370.
Weina K, Wu H, Knappe N, Orouji E, Novak D, Bernhardt M et al. TGF-β induces SOX2 expression in a time-dependent manner in human melanoma cells. Pigment Cell Melanoma Res 2016; 29: 453–458.
Shakhova O, Zingg D, Schaefer SM, Hari L, Civenni G, Blunschi J et al. Sox10 promotes the formation and maintenance of giant congenital naevi and melanoma. Nat Cell Biol 2012; 14.:882–890.
Ackermann J, Frutschi M, Kaloulis K, Mckee T, Trumpp A, Beermann F . Metastasizing melanoma formation caused by expression of activated N-Ras Q61K on an INK4a-deficient background. Cancer Res 2005; 65: 4005–4011.
Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal 2013; 6: pl1.
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2012; 2: 401–404.
Buttner P, Garbe C, Bertz J, Burg G, d’Hoedt B, Drepper H et al. Primary cutaneous melanoma: optimized cutoff points of tumor thickness and importance of Clark’s level for prognostic classification. Cancer 1995; 75: 2499–2506.
Buzaid AC, Ross MI, Balch CM, Soong S, McCarthy WH, Tinoco L et al. Critical analysis of the current American Joint Committee on Cancer staging system for cutaneous melanoma and proposal of a new staging system. J Clin Oncol 1997; 15: 1039–1051.
Carrasco-Garcia E, Santos JC, Garcia I, Brianti M, García-Puga M, Pedrazzoli J et al. Paradoxical role of SOX2 in gastric cancer. Am J Cancer Res 2016; 6: 701–713.
Driskell RR, Giangreco A, Jensen KB, Mulder KW, Watt FM . Sox2-positive dermal papilla cells specify hair follicle type in mammalian epidermis. Development 2009; 136: 2815–2823.
Chen P-L, Chen W-S, Li J, Lind AC, Lu D . Diagnostic utility of neural stem and progenitor cell markers nestin and SOX2 in distinguishing nodal melanocytic nevi from metastatic melanomas. Mod Pathol 2012; 26: 44–53.
Soriano P . Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat Genet 1999; 21: 70–71.
Miyagi S, Masui S, Niwa H, Saito T, Shimazaki T, Okano H et al. Consequence of the loss of Sox2 in the developing brain of the mouse. FEBS Lett 2008; 582: 2811–2815.
Zingg D, Debbache J, Schaefer SM, Tuncer E, Frommel SC, Cheng P et al. The epigenetic modifier EZH2 controls melanoma growth and metastasis through silencing of distinct tumour suppressors. Nat Commun 2015; 6: 6051.
Shakhova O, Sommer L . Testing the cancer stem cell hypothesis in melanoma: the clinics will tell. Cancer Lett 2013; 338: 74–81.
Dankort D, Filenova E, Collado M, Serrano M, Jones K, McMahon M . A new mouse model to explore the initiation, progression, and therapy of BRAFV600E-induced lung tumors. Genes Dev 2007; 21: 379–384.
Cesarini V, Guida E, Todara F, Di Agostina S, Nicolis S, Favaro R et al. Sox2 is not required for melanomagenesis, melanoma growth and melanoma metastasis in vivo. Oncogene; e-pub ahead of print 3 April 2017.
Wong DJ, Liu H, Ridky TW, Cassarino D, Segal E, Chang HY . Module map of stem cell genes guides creation of epithelial cancer stem cells. Cell Stem Cell 2008; 2: 333–344.
Van Keymeulen A, Lee MY, Ousset M, Brohée S, Rorive S, Giraddi RR et al. Reactivation of multipotency by oncogenic PIK3CA induces breast tumour heterogeneity. Nature 2015; 525: 119–123.
Kaufman CK, Mosimann C, Fan ZP, Yang S, Thomas AJ, Ablain J et al. A zebrafish melanoma model reveals emergence of neural crest identity during melanoma initiation. Science 2016; 351: aad2197.
Wakamatsu Y, Endo Y, Osumi N, Weston JA . Multiple roles of Sox2, an HMG-box transcription factor in avian neural crest development. Dev Dyn 2004; 229: 74–86.
Zipser MC, Eichhoff OM, Widmer DS, Schlegel NC, Schoenewolf NL, Stuart D et al. A proliferative melanoma cell phenotype is responsive to RAF/MEK inhibition independent of BRAF mutation status. Pigment Cell Melanoma Res 2011; 24: 326–333.
Civenni G, Walter A, Kobert N, Mihic-Probst D, Zipser M, Belloni B et al. Human CD271-positive melanoma stem cells associated with metastasis establish tumor heterogeneity and long-term growth. Cancer Res 2011; 71: 3098–3109.
Acknowledgements
We thank Annika Klug for help with hematoxylin and eosin stainings and Jessica Häusel for assistance in histology. We would like to thank Daniel Zingg for his help with statistical analysis. Further, we acknowledge the advice by the Cancer Biology PhD program of the University of Zurich. This work was supported by the Swiss National Science Foundation (SNF), the University Research Priority Program (URPP) ‘Translational Cancer Research’ Biobank, and the Swiss Cancer League. Work in the Nicolis laboratory was supported by AIRC and Telethon.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Schaefer, S., Segalada, C., Cheng, P. et al. Sox2 is dispensable for primary melanoma and metastasis formation. Oncogene 36, 4516–4524 (2017). https://doi.org/10.1038/onc.2017.55
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2017.55
This article is cited by
-
SOX2 as a novel contributor of oxidative metabolism in melanoma cells
Cell Communication and Signaling (2018)
-
Sox2 is not required for melanomagenesis, melanoma growth and melanoma metastasis in vivo
Oncogene (2017)